Key Insights
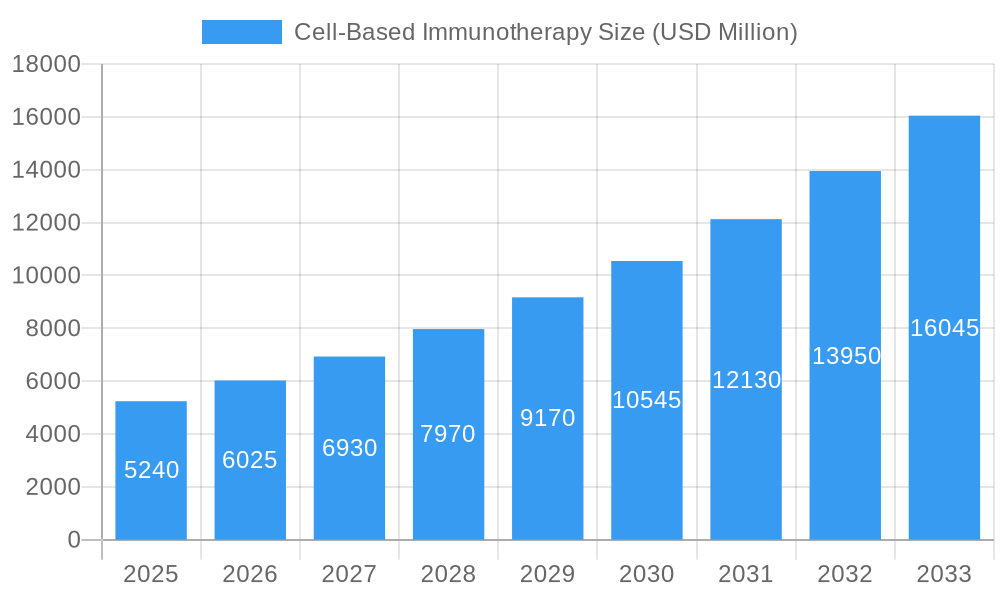
The global Cell-Based Immunotherapy market is poised for substantial growth, projected to reach a market size of $5.24 billion in 2025, with an impressive Compound Annual Growth Rate (CAGR) of 14.98% through 2033. This robust expansion is primarily fueled by the increasing prevalence of chronic diseases, including cancer and autoimmune disorders, which are driving demand for innovative treatment modalities. The escalating investments in research and development by leading pharmaceutical and biotechnology companies are further propelling the market forward, fostering the development of novel cellular therapies. Furthermore, a growing awareness among patients and healthcare providers about the efficacy and potential of immunotherapy is contributing to its wider adoption, particularly in advanced healthcare systems. The market is also benefiting from favorable regulatory landscapes in key regions, which are streamlining the approval processes for these groundbreaking treatments.
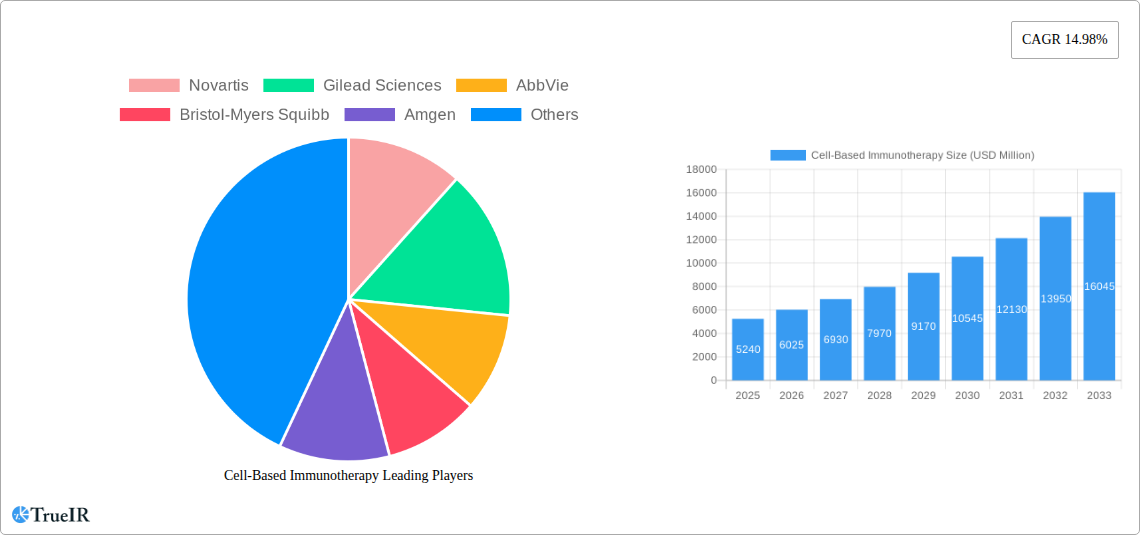
Cell-Based Immunotherapy Market Size (In Billion)

The market's trajectory is also influenced by significant trends such as the advancement of CAR T-cell therapy, which has demonstrated remarkable success in treating certain hematological malignancies, and the growing interest in autologous cellular immunotherapy as a personalized treatment approach. While the market is experiencing strong growth, certain restraints, such as the high cost of these advanced therapies and the complex manufacturing processes, could pose challenges. However, ongoing efforts to improve manufacturing efficiency and reduce costs are expected to mitigate these limitations over the forecast period. Geographically, North America and Europe are anticipated to lead the market due to well-established healthcare infrastructures and high healthcare expenditure, while the Asia Pacific region is expected to witness the fastest growth driven by increasing healthcare investments and a rising patient pool.
Cell-Based Immunotherapy Company Market Share

Cell-Based Immunotherapy Market Report: Driving Cancer Treatment Innovation
This comprehensive report delves into the dynamic global Cell-Based Immunotherapy market, projecting a market size of xxx billion by 2033, with a robust Compound Annual Growth Rate (CAGR) of xx% during the forecast period of 2025–2033. Fueled by advancements in oncology and a growing demand for personalized treatment approaches, cell-based immunotherapies are revolutionizing cancer care. The report offers deep insights into market structure, trends, dominant segments, product analysis, key drivers, challenges, competitive landscape, and future outlook, providing critical intelligence for stakeholders in the biotechnology, pharmaceutical, and healthcare industries. The study covers the historical period from 2019 to 2024, with a base year of 2025, and an estimated year also of 2025.
Cell-Based Immunotherapy Market Structure & Competitive Landscape
The global cell-based immunotherapy market is characterized by a moderate to high concentration, with a few key players holding significant market share. Innovation drivers are primarily focused on enhancing efficacy, improving safety profiles, and expanding the applicability of existing therapies to a broader range of cancers. Regulatory bodies play a crucial role, with evolving guidelines for novel cell therapies impacting market entry and development timelines. Product substitutes, while emerging, are largely nascent, with conventional treatments like chemotherapy and radiation therapy still prevalent. However, the personalized nature and targeted efficacy of cell-based immunotherapies are increasingly differentiating them. End-user segmentation reveals a strong reliance on Hospitals, followed by Clinics and Ambulatory Surgical Centers, with significant growth potential in Others, encompassing specialized research institutions and patient advocacy groups. Mergers and Acquisitions (M&A) activity is a notable trend, with companies acquiring innovative technologies and pipelines to strengthen their portfolios. In the historical period (2019-2024), there were an estimated xx M&A deals with a cumulative value of xx billion. Looking ahead, the market is expected to see further consolidation as companies seek to capture market share and leverage economies of scale.
Cell-Based Immunotherapy Market Trends & Opportunities
The cell-based immunotherapy market is experiencing exponential growth, driven by a paradigm shift in cancer treatment strategies. The market size, projected to reach xxx billion by 2033, reflects the increasing adoption of these advanced therapies. Technological advancements are at the forefront, with continuous innovation in areas like CAR T-cell engineering, gene editing techniques (e.g., CRISPR), and manufacturing processes for autologous and allogeneic cell therapies. The development of next-generation CAR T-cells with improved persistence, reduced on-target, off-tumor toxicities, and applicability to solid tumors presents significant opportunities. Furthermore, the rise of dendritic cell-based vaccine therapies is offering novel approaches to stimulate endogenous immune responses against cancer.
Consumer preferences are increasingly leaning towards personalized and less toxic treatment options, making cell-based immunotherapies highly attractive. Patient outcomes data demonstrating significant remission rates and improved quality of life are fueling this demand. The competitive dynamics are intensifying, with both established pharmaceutical giants and nimble biotechnology startups vying for market dominance. Strategic collaborations, partnerships, and licensing agreements are becoming commonplace as companies seek to accelerate research and development and expand their market reach.
Market penetration rates are steadily increasing, particularly for approved CAR T-cell therapies in hematological malignancies. The expansion of indications to include more cancer types and earlier lines of treatment is a key growth catalyst. The development of off-the-shelf allogeneic cell therapies promises to address scalability and accessibility challenges, further driving market adoption. Opportunities also lie in exploring combination therapies, integrating cell-based immunotherapies with other treatment modalities to achieve synergistic effects. The increasing investment in R&D, coupled with supportive regulatory frameworks for innovative therapies, underscores the robust growth trajectory of the cell-based immunotherapy market. The market is projected to grow at a CAGR of xx% from 2025 to 2033.
Dominant Markets & Segments in Cell-Based Immunotherapy
The North America region currently dominates the global cell-based immunotherapy market, driven by substantial investments in research and development, a robust presence of leading pharmaceutical and biotechnology companies, and favorable reimbursement policies for advanced therapies. Within North America, the United States stands out as the leading country, boasting a high number of clinical trials, approved cell therapies, and advanced healthcare infrastructure.
Among the application segments, Hospitals are the dominant channel, as they possess the specialized infrastructure, trained personnel, and multidisciplinary teams required for the administration and management of complex cell-based immunotherapies. The intricate nature of these treatments, often involving intensive patient monitoring and management of potential side effects, necessitates the resources available in hospital settings.
In terms of therapeutic types, Chimeric Antigen Receptor (CAR) T-Cell Therapy holds a significant market share. This dominance is attributed to its remarkable success in treating certain hematological malignancies, leading to high remission rates and improved patient survival. The ongoing development of new CAR T-cell targets and the expansion of their application to various blood cancers continue to fuel this segment's growth. Autologous Cellular Immunotherapy also commands a substantial market presence, as it leverages a patient's own immune cells, theoretically minimizing rejection risks. However, the logistical complexities and manufacturing timelines associated with autologous therapies present challenges. Dendritic Cell-based Vaccine Therapy is an emerging and promising segment, showing potential in stimulating endogenous anti-tumor immunity, and its market share is expected to grow as research and clinical applications expand. Key growth drivers across these dominant markets include:
- Advanced Research Infrastructure: Significant funding allocated to cancer research institutions and biotech hubs.
- Supportive Regulatory Frameworks: Expedited review pathways and incentives for innovative therapies.
- High Healthcare Expenditure: Greater capacity for adopting expensive, cutting-edge treatments.
- Increasing Cancer Incidence: A growing patient pool requiring novel therapeutic solutions.
- Technological Advancements: Continuous innovation in cell engineering and manufacturing.
Cell-Based Immunotherapy Product Analysis
Cell-based immunotherapies are characterized by groundbreaking product innovations focused on harnessing the power of the patient's immune system to combat cancer. Key advancements include the development of CAR T-cell therapies with enhanced target recognition, improved persistence, and reduced cytokine release syndrome (CRS) and neurotoxicity. Products are increasingly designed for specific cancer types and genetic mutations, offering highly personalized treatment. For instance, autologous therapies like Kymriah and Yescarta have demonstrated significant efficacy in hematological cancers, while ongoing research targets solid tumors. Dendritic cell-based vaccines are emerging as promising alternatives, aiming to stimulate a broad anti-tumor immune response. The competitive advantage of these products lies in their potential for durable remissions and improved quality of life compared to traditional therapies, albeit often at a higher initial cost.
Key Drivers, Barriers & Challenges in Cell-Based Immunotherapy
Key Drivers:
The cell-based immunotherapy market is propelled by several potent forces. Technological advancements in genetic engineering, cell culture, and manufacturing are continuously improving the efficacy and safety of these therapies. The growing burden of cancer globally creates a constant demand for more effective treatments. Furthermore, increasing research and development investments by pharmaceutical and biotechnology companies, coupled with supportive regulatory pathways for novel therapies, accelerate innovation and market entry. The proven clinical success of existing cell therapies, particularly CAR T-cells, is a significant driver, inspiring further research and investor confidence.
Barriers & Challenges:
Despite its promise, the cell-based immunotherapy market faces significant hurdles. High manufacturing costs and complex production processes contribute to the exorbitant pricing of these therapies, limiting accessibility for many patients and healthcare systems. Regulatory complexities and lengthy approval timelines for novel cell-based products, especially for personalized treatments, pose significant challenges. Supply chain issues, including the sourcing of raw materials and specialized logistics for cell transport, can impact scalability. Limited manufacturing capacity and the need for specialized treatment centers and trained personnel create bottlenecks. Potential for adverse events, such as cytokine release syndrome and neurotoxicity, requires stringent patient monitoring and management protocols. Competition from conventional therapies and emerging treatment modalities also presents a challenge. The market size impacted by these challenges is estimated to be xx billion in forgone revenue.
Growth Drivers in the Cell-Based Immunotherapy Market
The cell-based immunotherapy market is experiencing robust growth fueled by a confluence of factors. Technological innovation remains paramount, with advancements in gene editing, synthetic biology, and novel cell engineering techniques continuously enhancing the potency and safety of these therapies. The increasing global cancer burden necessitates the development of more effective and personalized treatment options, driving demand. Supportive regulatory environments in key markets, such as expedited review pathways and incentives for orphan drugs, are accelerating the approval and commercialization of new therapies. Furthermore, growing investments in research and development by both established pharmaceutical giants and agile biotechnology firms are fueling the pipeline of promising cell-based immunotherapies. The proven efficacy of existing therapies, particularly CAR T-cells in certain hematological malignancies, is generating significant clinical and commercial momentum.
Challenges Impacting Cell-Based Immunotherapy Growth
The rapid growth of the cell-based immunotherapy market is not without its impediments. High manufacturing costs and the complexity of producing personalized cell therapies contribute to exorbitant treatment prices, posing significant accessibility challenges for a substantial patient population and straining healthcare budgets. Navigating complex regulatory pathways and achieving timely approvals for novel cell therapies, especially those with intricate manufacturing processes, remains a significant hurdle. Supply chain disruptions, from the procurement of specialized reagents to the logistics of transporting autologous cells, can impact the timely delivery of treatment. Limited manufacturing capacity and the scarcity of highly trained personnel capable of administering and managing these advanced therapies can create bottlenecks. Furthermore, the potential for severe adverse events, such as cytokine release syndrome and neurotoxicity, necessitates extensive patient monitoring and specialized care infrastructure, adding to the overall cost and complexity of treatment.
Key Players Shaping the Cell-Based Immunotherapy Market
- Novartis
- Gilead Sciences
- AbbVie
- Bristol-Myers Squibb
- Amgen
- AstraZeneca
- Biogen
- Boehringer Ingelheim
- Eli Lilly and Company
- GSK
- Pfizer
- Roche
- Takara Bio
- Bausch Health
- Apac Biotech
- Dendreon Pharmaceuticals (SanPower)
- JW CreaGene
- Green Cross Corp
Significant Cell-Based Immunotherapy Industry Milestones
- 2019: FDA approval of Yescarta (axicabtagene ciloleucel) for relapsed/refractory follicular lymphoma.
- 2020: EMA approval of Kymriah (tisagenlecleucel) for relapsed/refractory pediatric acute lymphoblastic leukemia.
- 2021: significant increase in CAR T-cell therapy approvals for hematological malignancies globally, reaching an estimated xx new approvals.
- 2022: Advancement of CAR T-cell therapies into earlier lines of treatment for certain blood cancers.
- 2023: Increased focus on developing CAR T-cell therapies for solid tumors, with several promising clinical trial results reported.
- 2024: Emergence of allogeneic CAR T-cell therapies in late-stage clinical trials, promising "off-the-shelf" solutions.
Future Outlook for Cell-Based Immunotherapy Market
The future outlook for the cell-based immunotherapy market is exceptionally bright, with continued exponential growth anticipated. Key growth catalysts include the expansion of CAR T-cell therapies to a wider array of hematological and solid tumors, coupled with the increasing development of allogeneic (off-the-shelf) cell therapies to address scalability and cost challenges. The integration of novel gene-editing technologies and sophisticated cell engineering techniques will further enhance therapeutic efficacy and safety. Strategic collaborations between pharmaceutical giants and specialized biotech firms will continue to drive innovation and accelerate pipeline development. The market is poised to witness a significant increase in patient access as manufacturing processes mature and reimbursement landscapes evolve, ultimately transforming cancer treatment paradigms and offering hope to millions worldwide. The projected market size by 2033 is expected to reach xxx billion.
Cell-Based Immunotherapy Segmentation
-
1. Application
- 1.1. Hospitals
- 1.2. Clinics
- 1.3. Ambulatory Surgical Centers
- 1.4. Others
-
2. Types
- 2.1. Autologous Cellular Immunotherapy
- 2.2. Chimeric Antigen Receptor (CAR) T-Cell Therapy
- 2.3. Dendritic Cell-based Vaccine Therapy
Cell-Based Immunotherapy Segmentation By Geography
-
1. North America
- 1.1. United States
- 1.2. Canada
- 1.3. Mexico
-
2. South America
- 2.1. Brazil
- 2.2. Argentina
- 2.3. Rest of South America
-
3. Europe
- 3.1. United Kingdom
- 3.2. Germany
- 3.3. France
- 3.4. Italy
- 3.5. Spain
- 3.6. Russia
- 3.7. Benelux
- 3.8. Nordics
- 3.9. Rest of Europe
-
4. Middle East & Africa
- 4.1. Turkey
- 4.2. Israel
- 4.3. GCC
- 4.4. North Africa
- 4.5. South Africa
- 4.6. Rest of Middle East & Africa
-
5. Asia Pacific
- 5.1. China
- 5.2. India
- 5.3. Japan
- 5.4. South Korea
- 5.5. ASEAN
- 5.6. Oceania
- 5.7. Rest of Asia Pacific
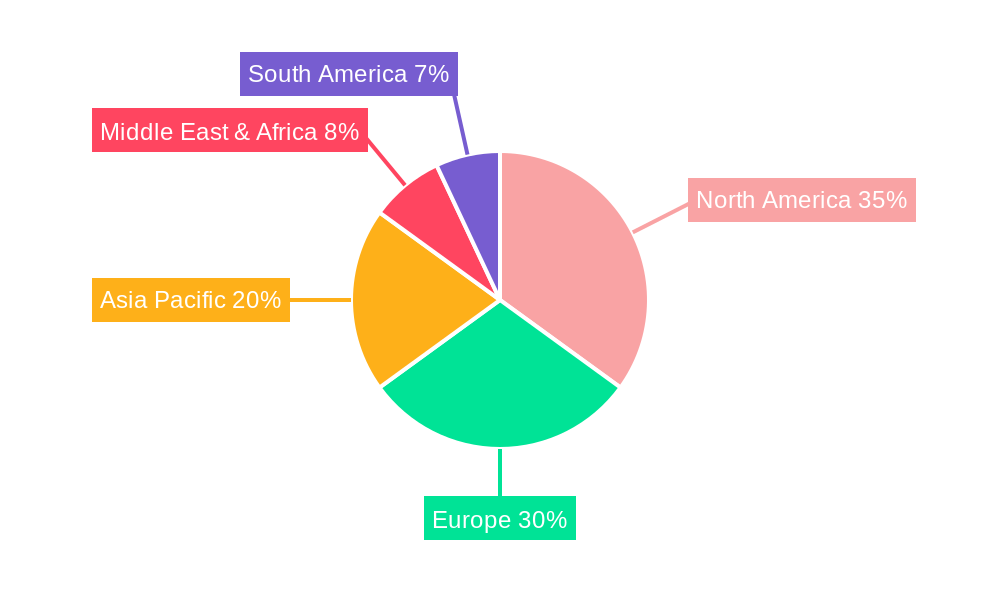
Cell-Based Immunotherapy Regional Market Share

Geographic Coverage of Cell-Based Immunotherapy
Cell-Based Immunotherapy REPORT HIGHLIGHTS
| Aspects | Details |
|---|---|
| Study Period | 2020-2034 |
| Base Year | 2025 |
| Estimated Year | 2026 |
| Forecast Period | 2026-2034 |
| Historical Period | 2020-2025 |
| Growth Rate | CAGR of 14.98% from 2020-2034 |
| Segmentation |
|
Table of Contents
- 1. Introduction
- 1.1. Research Scope
- 1.2. Market Segmentation
- 1.3. Research Methodology
- 1.4. Definitions and Assumptions
- 2. Executive Summary
- 2.1. Introduction
- 3. Market Dynamics
- 3.1. Introduction
- 3.2. Market Drivers
- 3.3. Market Restrains
- 3.4. Market Trends
- 4. Market Factor Analysis
- 4.1. Porters Five Forces
- 4.2. Supply/Value Chain
- 4.3. PESTEL analysis
- 4.4. Market Entropy
- 4.5. Patent/Trademark Analysis
- 5. Global Cell-Based Immunotherapy Analysis, Insights and Forecast, 2020-2032
- 5.1. Market Analysis, Insights and Forecast - by Application
- 5.1.1. Hospitals
- 5.1.2. Clinics
- 5.1.3. Ambulatory Surgical Centers
- 5.1.4. Others
- 5.2. Market Analysis, Insights and Forecast - by Types
- 5.2.1. Autologous Cellular Immunotherapy
- 5.2.2. Chimeric Antigen Receptor (CAR) T-Cell Therapy
- 5.2.3. Dendritic Cell-based Vaccine Therapy
- 5.3. Market Analysis, Insights and Forecast - by Region
- 5.3.1. North America
- 5.3.2. South America
- 5.3.3. Europe
- 5.3.4. Middle East & Africa
- 5.3.5. Asia Pacific
- 5.1. Market Analysis, Insights and Forecast - by Application
- 6. North America Cell-Based Immunotherapy Analysis, Insights and Forecast, 2020-2032
- 6.1. Market Analysis, Insights and Forecast - by Application
- 6.1.1. Hospitals
- 6.1.2. Clinics
- 6.1.3. Ambulatory Surgical Centers
- 6.1.4. Others
- 6.2. Market Analysis, Insights and Forecast - by Types
- 6.2.1. Autologous Cellular Immunotherapy
- 6.2.2. Chimeric Antigen Receptor (CAR) T-Cell Therapy
- 6.2.3. Dendritic Cell-based Vaccine Therapy
- 6.1. Market Analysis, Insights and Forecast - by Application
- 7. South America Cell-Based Immunotherapy Analysis, Insights and Forecast, 2020-2032
- 7.1. Market Analysis, Insights and Forecast - by Application
- 7.1.1. Hospitals
- 7.1.2. Clinics
- 7.1.3. Ambulatory Surgical Centers
- 7.1.4. Others
- 7.2. Market Analysis, Insights and Forecast - by Types
- 7.2.1. Autologous Cellular Immunotherapy
- 7.2.2. Chimeric Antigen Receptor (CAR) T-Cell Therapy
- 7.2.3. Dendritic Cell-based Vaccine Therapy
- 7.1. Market Analysis, Insights and Forecast - by Application
- 8. Europe Cell-Based Immunotherapy Analysis, Insights and Forecast, 2020-2032
- 8.1. Market Analysis, Insights and Forecast - by Application
- 8.1.1. Hospitals
- 8.1.2. Clinics
- 8.1.3. Ambulatory Surgical Centers
- 8.1.4. Others
- 8.2. Market Analysis, Insights and Forecast - by Types
- 8.2.1. Autologous Cellular Immunotherapy
- 8.2.2. Chimeric Antigen Receptor (CAR) T-Cell Therapy
- 8.2.3. Dendritic Cell-based Vaccine Therapy
- 8.1. Market Analysis, Insights and Forecast - by Application
- 9. Middle East & Africa Cell-Based Immunotherapy Analysis, Insights and Forecast, 2020-2032
- 9.1. Market Analysis, Insights and Forecast - by Application
- 9.1.1. Hospitals
- 9.1.2. Clinics
- 9.1.3. Ambulatory Surgical Centers
- 9.1.4. Others
- 9.2. Market Analysis, Insights and Forecast - by Types
- 9.2.1. Autologous Cellular Immunotherapy
- 9.2.2. Chimeric Antigen Receptor (CAR) T-Cell Therapy
- 9.2.3. Dendritic Cell-based Vaccine Therapy
- 9.1. Market Analysis, Insights and Forecast - by Application
- 10. Asia Pacific Cell-Based Immunotherapy Analysis, Insights and Forecast, 2020-2032
- 10.1. Market Analysis, Insights and Forecast - by Application
- 10.1.1. Hospitals
- 10.1.2. Clinics
- 10.1.3. Ambulatory Surgical Centers
- 10.1.4. Others
- 10.2. Market Analysis, Insights and Forecast - by Types
- 10.2.1. Autologous Cellular Immunotherapy
- 10.2.2. Chimeric Antigen Receptor (CAR) T-Cell Therapy
- 10.2.3. Dendritic Cell-based Vaccine Therapy
- 10.1. Market Analysis, Insights and Forecast - by Application
- 11. Competitive Analysis
- 11.1. Global Market Share Analysis 2025
- 11.2. Company Profiles
- 11.2.1 Novartis
- 11.2.1.1. Overview
- 11.2.1.2. Products
- 11.2.1.3. SWOT Analysis
- 11.2.1.4. Recent Developments
- 11.2.1.5. Financials (Based on Availability)
- 11.2.2 Gilead Sciences
- 11.2.2.1. Overview
- 11.2.2.2. Products
- 11.2.2.3. SWOT Analysis
- 11.2.2.4. Recent Developments
- 11.2.2.5. Financials (Based on Availability)
- 11.2.3 AbbVie
- 11.2.3.1. Overview
- 11.2.3.2. Products
- 11.2.3.3. SWOT Analysis
- 11.2.3.4. Recent Developments
- 11.2.3.5. Financials (Based on Availability)
- 11.2.4 Bristol-Myers Squibb
- 11.2.4.1. Overview
- 11.2.4.2. Products
- 11.2.4.3. SWOT Analysis
- 11.2.4.4. Recent Developments
- 11.2.4.5. Financials (Based on Availability)
- 11.2.5 Amgen
- 11.2.5.1. Overview
- 11.2.5.2. Products
- 11.2.5.3. SWOT Analysis
- 11.2.5.4. Recent Developments
- 11.2.5.5. Financials (Based on Availability)
- 11.2.6 AstraZeneca
- 11.2.6.1. Overview
- 11.2.6.2. Products
- 11.2.6.3. SWOT Analysis
- 11.2.6.4. Recent Developments
- 11.2.6.5. Financials (Based on Availability)
- 11.2.7 Biogen
- 11.2.7.1. Overview
- 11.2.7.2. Products
- 11.2.7.3. SWOT Analysis
- 11.2.7.4. Recent Developments
- 11.2.7.5. Financials (Based on Availability)
- 11.2.8 Boehringer Ingelheim
- 11.2.8.1. Overview
- 11.2.8.2. Products
- 11.2.8.3. SWOT Analysis
- 11.2.8.4. Recent Developments
- 11.2.8.5. Financials (Based on Availability)
- 11.2.9 Eli Lilly and Company
- 11.2.9.1. Overview
- 11.2.9.2. Products
- 11.2.9.3. SWOT Analysis
- 11.2.9.4. Recent Developments
- 11.2.9.5. Financials (Based on Availability)
- 11.2.10 GSK
- 11.2.10.1. Overview
- 11.2.10.2. Products
- 11.2.10.3. SWOT Analysis
- 11.2.10.4. Recent Developments
- 11.2.10.5. Financials (Based on Availability)
- 11.2.11 Pfizer
- 11.2.11.1. Overview
- 11.2.11.2. Products
- 11.2.11.3. SWOT Analysis
- 11.2.11.4. Recent Developments
- 11.2.11.5. Financials (Based on Availability)
- 11.2.12 Roche
- 11.2.12.1. Overview
- 11.2.12.2. Products
- 11.2.12.3. SWOT Analysis
- 11.2.12.4. Recent Developments
- 11.2.12.5. Financials (Based on Availability)
- 11.2.13 Takara Bio
- 11.2.13.1. Overview
- 11.2.13.2. Products
- 11.2.13.3. SWOT Analysis
- 11.2.13.4. Recent Developments
- 11.2.13.5. Financials (Based on Availability)
- 11.2.14 Bausch Health
- 11.2.14.1. Overview
- 11.2.14.2. Products
- 11.2.14.3. SWOT Analysis
- 11.2.14.4. Recent Developments
- 11.2.14.5. Financials (Based on Availability)
- 11.2.15 Apac Biotech
- 11.2.15.1. Overview
- 11.2.15.2. Products
- 11.2.15.3. SWOT Analysis
- 11.2.15.4. Recent Developments
- 11.2.15.5. Financials (Based on Availability)
- 11.2.16 Dendreon Pharmaceuticals (SanPower)
- 11.2.16.1. Overview
- 11.2.16.2. Products
- 11.2.16.3. SWOT Analysis
- 11.2.16.4. Recent Developments
- 11.2.16.5. Financials (Based on Availability)
- 11.2.17 JW CreaGene
- 11.2.17.1. Overview
- 11.2.17.2. Products
- 11.2.17.3. SWOT Analysis
- 11.2.17.4. Recent Developments
- 11.2.17.5. Financials (Based on Availability)
- 11.2.18 Green Cross Corp
- 11.2.18.1. Overview
- 11.2.18.2. Products
- 11.2.18.3. SWOT Analysis
- 11.2.18.4. Recent Developments
- 11.2.18.5. Financials (Based on Availability)
- 11.2.1 Novartis
List of Figures
- Figure 1: Global Cell-Based Immunotherapy Revenue Breakdown (undefined, %) by Region 2025 & 2033
- Figure 2: North America Cell-Based Immunotherapy Revenue (undefined), by Application 2025 & 2033
- Figure 3: North America Cell-Based Immunotherapy Revenue Share (%), by Application 2025 & 2033
- Figure 4: North America Cell-Based Immunotherapy Revenue (undefined), by Types 2025 & 2033
- Figure 5: North America Cell-Based Immunotherapy Revenue Share (%), by Types 2025 & 2033
- Figure 6: North America Cell-Based Immunotherapy Revenue (undefined), by Country 2025 & 2033
- Figure 7: North America Cell-Based Immunotherapy Revenue Share (%), by Country 2025 & 2033
- Figure 8: South America Cell-Based Immunotherapy Revenue (undefined), by Application 2025 & 2033
- Figure 9: South America Cell-Based Immunotherapy Revenue Share (%), by Application 2025 & 2033
- Figure 10: South America Cell-Based Immunotherapy Revenue (undefined), by Types 2025 & 2033
- Figure 11: South America Cell-Based Immunotherapy Revenue Share (%), by Types 2025 & 2033
- Figure 12: South America Cell-Based Immunotherapy Revenue (undefined), by Country 2025 & 2033
- Figure 13: South America Cell-Based Immunotherapy Revenue Share (%), by Country 2025 & 2033
- Figure 14: Europe Cell-Based Immunotherapy Revenue (undefined), by Application 2025 & 2033
- Figure 15: Europe Cell-Based Immunotherapy Revenue Share (%), by Application 2025 & 2033
- Figure 16: Europe Cell-Based Immunotherapy Revenue (undefined), by Types 2025 & 2033
- Figure 17: Europe Cell-Based Immunotherapy Revenue Share (%), by Types 2025 & 2033
- Figure 18: Europe Cell-Based Immunotherapy Revenue (undefined), by Country 2025 & 2033
- Figure 19: Europe Cell-Based Immunotherapy Revenue Share (%), by Country 2025 & 2033
- Figure 20: Middle East & Africa Cell-Based Immunotherapy Revenue (undefined), by Application 2025 & 2033
- Figure 21: Middle East & Africa Cell-Based Immunotherapy Revenue Share (%), by Application 2025 & 2033
- Figure 22: Middle East & Africa Cell-Based Immunotherapy Revenue (undefined), by Types 2025 & 2033
- Figure 23: Middle East & Africa Cell-Based Immunotherapy Revenue Share (%), by Types 2025 & 2033
- Figure 24: Middle East & Africa Cell-Based Immunotherapy Revenue (undefined), by Country 2025 & 2033
- Figure 25: Middle East & Africa Cell-Based Immunotherapy Revenue Share (%), by Country 2025 & 2033
- Figure 26: Asia Pacific Cell-Based Immunotherapy Revenue (undefined), by Application 2025 & 2033
- Figure 27: Asia Pacific Cell-Based Immunotherapy Revenue Share (%), by Application 2025 & 2033
- Figure 28: Asia Pacific Cell-Based Immunotherapy Revenue (undefined), by Types 2025 & 2033
- Figure 29: Asia Pacific Cell-Based Immunotherapy Revenue Share (%), by Types 2025 & 2033
- Figure 30: Asia Pacific Cell-Based Immunotherapy Revenue (undefined), by Country 2025 & 2033
- Figure 31: Asia Pacific Cell-Based Immunotherapy Revenue Share (%), by Country 2025 & 2033
List of Tables
- Table 1: Global Cell-Based Immunotherapy Revenue undefined Forecast, by Application 2020 & 2033
- Table 2: Global Cell-Based Immunotherapy Revenue undefined Forecast, by Types 2020 & 2033
- Table 3: Global Cell-Based Immunotherapy Revenue undefined Forecast, by Region 2020 & 2033
- Table 4: Global Cell-Based Immunotherapy Revenue undefined Forecast, by Application 2020 & 2033
- Table 5: Global Cell-Based Immunotherapy Revenue undefined Forecast, by Types 2020 & 2033
- Table 6: Global Cell-Based Immunotherapy Revenue undefined Forecast, by Country 2020 & 2033
- Table 7: United States Cell-Based Immunotherapy Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 8: Canada Cell-Based Immunotherapy Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 9: Mexico Cell-Based Immunotherapy Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 10: Global Cell-Based Immunotherapy Revenue undefined Forecast, by Application 2020 & 2033
- Table 11: Global Cell-Based Immunotherapy Revenue undefined Forecast, by Types 2020 & 2033
- Table 12: Global Cell-Based Immunotherapy Revenue undefined Forecast, by Country 2020 & 2033
- Table 13: Brazil Cell-Based Immunotherapy Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 14: Argentina Cell-Based Immunotherapy Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 15: Rest of South America Cell-Based Immunotherapy Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 16: Global Cell-Based Immunotherapy Revenue undefined Forecast, by Application 2020 & 2033
- Table 17: Global Cell-Based Immunotherapy Revenue undefined Forecast, by Types 2020 & 2033
- Table 18: Global Cell-Based Immunotherapy Revenue undefined Forecast, by Country 2020 & 2033
- Table 19: United Kingdom Cell-Based Immunotherapy Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 20: Germany Cell-Based Immunotherapy Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 21: France Cell-Based Immunotherapy Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 22: Italy Cell-Based Immunotherapy Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 23: Spain Cell-Based Immunotherapy Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 24: Russia Cell-Based Immunotherapy Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 25: Benelux Cell-Based Immunotherapy Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 26: Nordics Cell-Based Immunotherapy Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 27: Rest of Europe Cell-Based Immunotherapy Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 28: Global Cell-Based Immunotherapy Revenue undefined Forecast, by Application 2020 & 2033
- Table 29: Global Cell-Based Immunotherapy Revenue undefined Forecast, by Types 2020 & 2033
- Table 30: Global Cell-Based Immunotherapy Revenue undefined Forecast, by Country 2020 & 2033
- Table 31: Turkey Cell-Based Immunotherapy Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 32: Israel Cell-Based Immunotherapy Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 33: GCC Cell-Based Immunotherapy Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 34: North Africa Cell-Based Immunotherapy Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 35: South Africa Cell-Based Immunotherapy Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 36: Rest of Middle East & Africa Cell-Based Immunotherapy Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 37: Global Cell-Based Immunotherapy Revenue undefined Forecast, by Application 2020 & 2033
- Table 38: Global Cell-Based Immunotherapy Revenue undefined Forecast, by Types 2020 & 2033
- Table 39: Global Cell-Based Immunotherapy Revenue undefined Forecast, by Country 2020 & 2033
- Table 40: China Cell-Based Immunotherapy Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 41: India Cell-Based Immunotherapy Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 42: Japan Cell-Based Immunotherapy Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 43: South Korea Cell-Based Immunotherapy Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 44: ASEAN Cell-Based Immunotherapy Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 45: Oceania Cell-Based Immunotherapy Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 46: Rest of Asia Pacific Cell-Based Immunotherapy Revenue (undefined) Forecast, by Application 2020 & 2033
Frequently Asked Questions
1. What is the projected Compound Annual Growth Rate (CAGR) of the Cell-Based Immunotherapy?
The projected CAGR is approximately 14.98%.
2. Which companies are prominent players in the Cell-Based Immunotherapy?
Key companies in the market include Novartis, Gilead Sciences, AbbVie, Bristol-Myers Squibb, Amgen, AstraZeneca, Biogen, Boehringer Ingelheim, Eli Lilly and Company, GSK, Pfizer, Roche, Takara Bio, Bausch Health, Apac Biotech, Dendreon Pharmaceuticals (SanPower), JW CreaGene, Green Cross Corp.
3. What are the main segments of the Cell-Based Immunotherapy?
The market segments include Application, Types.
4. Can you provide details about the market size?
The market size is estimated to be USD XXX N/A as of 2022.
5. What are some drivers contributing to market growth?
N/A
6. What are the notable trends driving market growth?
N/A
7. Are there any restraints impacting market growth?
N/A
8. Can you provide examples of recent developments in the market?
N/A
9. What pricing options are available for accessing the report?
Pricing options include single-user, multi-user, and enterprise licenses priced at USD 2900.00, USD 4350.00, and USD 5800.00 respectively.
10. Is the market size provided in terms of value or volume?
The market size is provided in terms of value, measured in N/A.
11. Are there any specific market keywords associated with the report?
Yes, the market keyword associated with the report is "Cell-Based Immunotherapy," which aids in identifying and referencing the specific market segment covered.
12. How do I determine which pricing option suits my needs best?
The pricing options vary based on user requirements and access needs. Individual users may opt for single-user licenses, while businesses requiring broader access may choose multi-user or enterprise licenses for cost-effective access to the report.
13. Are there any additional resources or data provided in the Cell-Based Immunotherapy report?
While the report offers comprehensive insights, it's advisable to review the specific contents or supplementary materials provided to ascertain if additional resources or data are available.
14. How can I stay updated on further developments or reports in the Cell-Based Immunotherapy?
To stay informed about further developments, trends, and reports in the Cell-Based Immunotherapy, consider subscribing to industry newsletters, following relevant companies and organizations, or regularly checking reputable industry news sources and publications.
Methodology
Step 1 - Identification of Relevant Samples Size from Population Database



Step 2 - Approaches for Defining Global Market Size (Value, Volume* & Price*)

Note*: In applicable scenarios
Step 3 - Data Sources
Primary Research
- Web Analytics
- Survey Reports
- Research Institute
- Latest Research Reports
- Opinion Leaders
Secondary Research
- Annual Reports
- White Paper
- Latest Press Release
- Industry Association
- Paid Database
- Investor Presentations

Step 4 - Data Triangulation
Involves using different sources of information in order to increase the validity of a study
These sources are likely to be stakeholders in a program - participants, other researchers, program staff, other community members, and so on.
Then we put all data in single framework & apply various statistical tools to find out the dynamic on the market.
During the analysis stage, feedback from the stakeholder groups would be compared to determine areas of agreement as well as areas of divergence


