Key Insights
The Medical Nitinol Braiding Solutions market is poised for significant expansion, projected to reach $500 million in 2025 and grow at a robust 8% CAGR through 2033. This impressive growth is primarily fueled by the increasing prevalence of cardiovascular diseases and orthopedic conditions, driving the demand for advanced minimally invasive surgical procedures. The unique properties of Nitinol, such as superelasticity and shape memory, make it an ideal material for crafting intricate medical devices like stents, guidewires, and vascular grafts, all of which fall under the dominant "Stents and Vascular Devices" segment. The "Cardiovascular Field" application is expected to lead the market, owing to the continuous innovation in interventional cardiology and the rising number of procedures performed globally. Furthermore, the growing adoption of Nitinol braiding solutions in dental and other specialized medical applications, including neurovascular and urological interventions, will contribute to sustained market momentum.
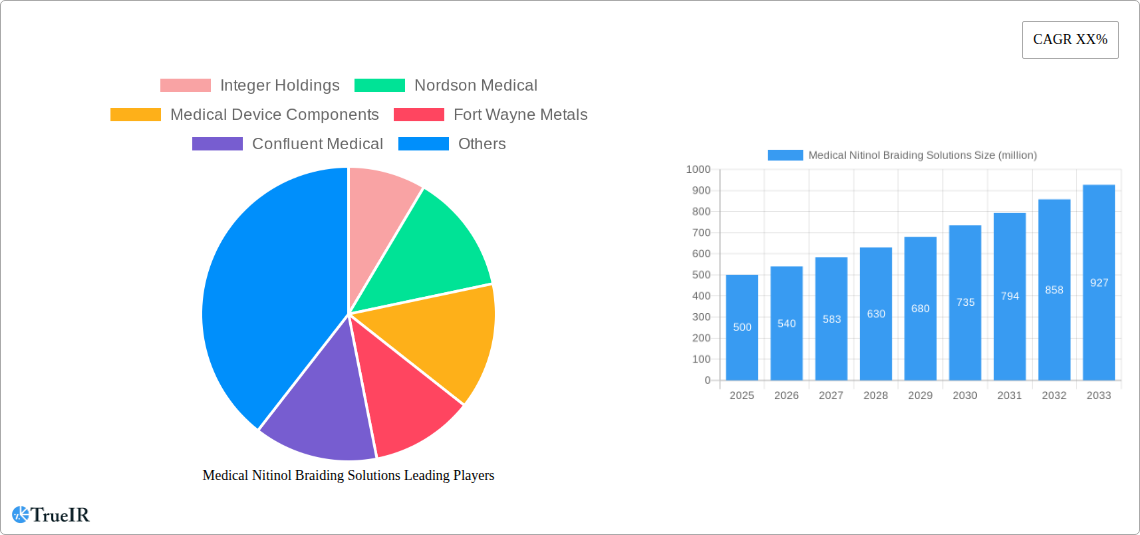
Medical Nitinol Braiding Solutions Market Size (In Million)

Geographically, North America is anticipated to maintain its leading position in the Medical Nitinol Braiding Solutions market, driven by advanced healthcare infrastructure, high patient spending, and early adoption of innovative medical technologies. Europe follows closely, with strong research and development capabilities and a growing demand for sophisticated medical devices. The Asia Pacific region, however, presents the most substantial growth opportunity, propelled by an expanding patient pool, increasing healthcare expenditure, and a rapidly developing medical device manufacturing sector, particularly in China and India. Key market restraints include the high cost of Nitinol material and complex manufacturing processes, alongside stringent regulatory approvals for new medical devices. Nevertheless, ongoing technological advancements in braiding techniques and material processing are expected to mitigate these challenges, paving the way for broader market penetration and continued innovation in medical device design.
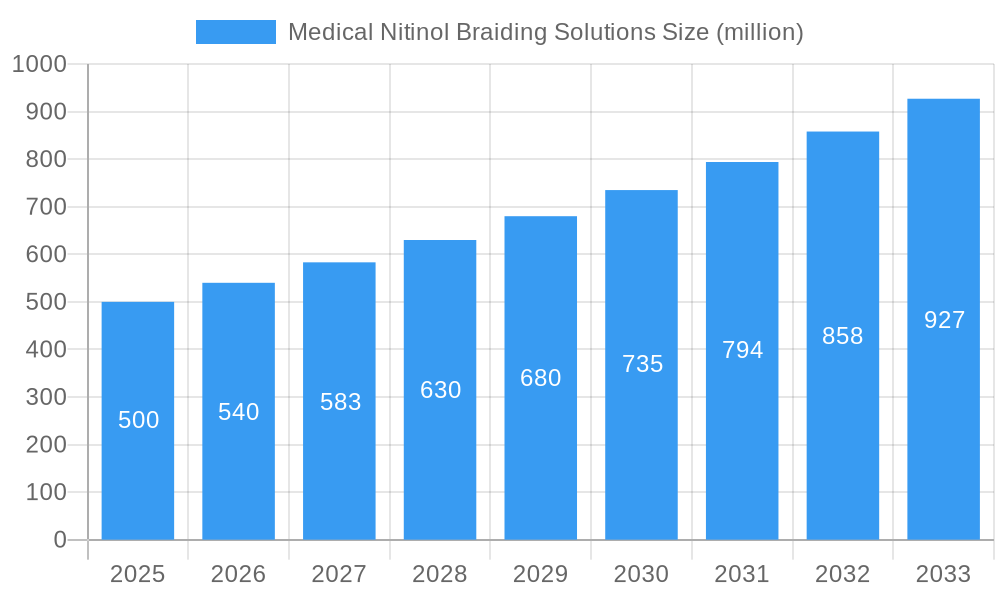
Medical Nitinol Braiding Solutions Company Market Share

Medical Nitinol Braiding Solutions Market Structure & Competitive Landscape
The medical nitinol braiding solutions market exhibits a moderately concentrated structure, driven by a blend of established players and emerging innovators. Key companies like Integer Holdings, Nordson Medical, and Medical Device Components hold significant market share due to their extensive product portfolios and robust R&D capabilities. Innovation is primarily fueled by advancements in nitinol material science and braiding technologies, enabling the creation of more intricate and patient-specific devices. Regulatory frameworks, particularly FDA and CE marking approvals, significantly influence market entry and product development, acting as both a barrier and a quality assurance mechanism. Product substitutes, while present in some lower-end applications, struggle to match nitinol's unique superelastic and shape memory properties for critical medical interventions. End-user segmentation is dominated by the cardiovascular field, followed by orthopedics and dentistry, with "Others" encompassing neurovascular and other specialized applications. Mergers and acquisitions (M&A) are a notable trend, with companies strategically acquiring innovative technologies and expanding their geographical reach. For instance, in the historical period (2019-2024), an estimated 15 significant M&A activities were recorded, consolidating expertise and market presence. Concentration ratios are estimated to be around 65% for the top five players.
Medical Nitinol Braiding Solutions Market Trends & Opportunities
The global medical nitinol braiding solutions market is poised for substantial expansion, projected to grow at a Compound Annual Growth Rate (CAGR) of approximately 9.50% from its base year of 2025 through 2033. This robust growth trajectory is underpinned by a confluence of favorable market trends and burgeoning opportunities. The increasing prevalence of chronic diseases, such as cardiovascular disorders and orthopedic conditions, is a primary catalyst, driving the demand for minimally invasive medical devices that leverage nitinol's unique properties for enhanced patient outcomes. Technological advancements in braiding techniques and nitinol processing are enabling the development of smaller, more flexible, and highly precise medical implants and instruments. For example, advancements in laser braiding and advanced computational modeling are allowing for unprecedented control over braid architecture, leading to devices with superior biomechanical performance.
Consumer preferences are shifting towards less invasive procedures, directly benefiting the adoption of nitinol-based braided solutions that facilitate shorter recovery times and reduced patient discomfort. This trend is particularly evident in the cardiovascular segment, where nitinol stents and vascular grafts are replacing traditional open surgical interventions. The competitive landscape is characterized by intense innovation and strategic partnerships. Companies are investing heavily in R&D to develop novel nitinol alloys and braiding configurations optimized for specific anatomical locations and therapeutic interventions. Market penetration rates for advanced nitinol braided devices are expected to surge, particularly in developed economies with advanced healthcare infrastructure and a high propensity for adopting cutting-edge medical technologies. Emerging markets also present significant untapped potential, driven by improving healthcare access and a growing demand for advanced medical treatments. The market size is estimated to grow from approximately $5,500 million in the base year 2025 to an estimated $11,000 million by the end of the forecast period in 2033.
Furthermore, the increasing focus on personalized medicine is creating opportunities for customized nitinol braiding solutions tailored to individual patient anatomy and therapeutic needs. This could involve patient-specific stent designs or complex braided structures for highly intricate surgical procedures. The integration of artificial intelligence and machine learning in device design and manufacturing is also expected to streamline the development process and improve the quality and consistency of nitinol braided products.
Dominant Markets & Segments in Medical Nitinol Braiding Solutions
The Cardiovascular Field stands as the dominant application segment within the medical nitinol braiding solutions market, driven by the escalating global burden of cardiovascular diseases. This segment is projected to continue its leadership throughout the study period (2019-2033).
- Cardiovascular Field:
- Stents and Vascular Devices: This sub-segment is the primary growth engine, fueled by the widespread use of nitinol stents for treating peripheral artery disease, coronary artery disease, and aortic aneurysms. The demand for minimally invasive vascular repair and treatment solutions is insatiable.
- Guidewires: Advanced nitinol braided guidewires offer superior torqueability, flexibility, and kink resistance, making them indispensable for complex interventional cardiology procedures. Their precision allows for navigation through tortuous vascular pathways.
- Growth Drivers:
- Aging Population: An increasing elderly population globally directly correlates with a higher incidence of cardiovascular ailments.
- Lifestyle Changes: Sedentary lifestyles, unhealthy diets, and rising obesity rates contribute to the growing prevalence of cardiovascular conditions.
- Technological Advancements: Continuous innovation in nitinol braiding techniques allows for the development of smaller, more deliverable, and highly effective vascular devices.
- Reimbursement Policies: Favorable reimbursement policies for minimally invasive procedures further encourage the adoption of nitinol-based cardiovascular treatments.
The Orthopedic segment is the second-largest contributor, with applications in fracture fixation, joint replacement, and spinal implants. Nitinol's shape memory properties are ideal for creating self-expanding or self-adjusting orthopedic devices.
- Orthopedic:
- Implants: Nitinol-based orthopedic implants offer biocompatibility, corrosion resistance, and excellent mechanical properties, making them suitable for long-term implantation.
- Growth Drivers:
- Increasing Sports Injuries: A rise in participation in sports and recreational activities leads to a higher incidence of orthopedic injuries.
- Osteoporosis: The growing prevalence of osteoporosis, particularly in post-menopausal women, necessitates advanced fixation devices.
- Demand for Revision Surgeries: Wear and tear on existing implants create a demand for revision orthopedic procedures where nitinol can play a crucial role.
The Dental segment, though smaller, is experiencing steady growth driven by applications in orthodontics and endodontics. Nitinol wires in braces offer continuous, gentle force, and nitinol endodontic files provide enhanced flexibility and resistance to fracture.
- Dental:
- Orthodontic Wires: Nitinol's superelasticity allows for precise and comfortable tooth movement.
- Endodontic Files: Superior flexibility and shape memory reduce the risk of instrument fracture during root canal treatments.
- Growth Drivers:
- Cosmetic Dentistry Trends: Growing awareness and demand for aesthetic dental treatments.
- Advanced Orthodontic Solutions: Patient preference for faster and more comfortable orthodontic treatments.
The "Others" segment, encompassing neurovascular devices (e.g., coils for aneurysm treatment), gastrointestinal devices, and other specialized surgical instruments, represents a growing area of opportunity due to the expansion of interventional procedures across various medical disciplines.
Medical Nitinol Braiding Solutions Product Analysis
Medical nitinol braiding solutions are characterized by remarkable product innovation driven by the material's unique superelasticity and shape memory properties. Braided structures allow for enhanced flexibility, kink resistance, and radial force, making them ideal for complex minimally invasive devices such as vascular stents, guidewires, and neurovascular coils. Companies are developing intricate braiding patterns and utilizing advanced manufacturing techniques like laser cutting and electrochemical polishing to create high-performance devices with precise control over mechanical characteristics. The competitive advantage lies in the ability to tailor device designs for specific anatomical regions and therapeutic needs, leading to improved patient outcomes and procedural efficiency. Technological advancements are enabling the creation of bioresorbable nitinol alloys and drug-eluting braided devices, further expanding their therapeutic potential.
Key Drivers, Barriers & Challenges in Medical Nitinol Braiding Solutions
Growth Drivers:
- Technological Innovation: Continuous advancements in nitinol processing and braiding techniques are creating novel, high-performance medical devices.
- Rising Prevalence of Chronic Diseases: Increasing incidence of cardiovascular, orthopedic, and other conditions drives demand for minimally invasive solutions.
- Growing Demand for Minimally Invasive Procedures: Patient and physician preference for less invasive interventions favors nitinol-braided devices.
- Favorable Reimbursement Policies: Supportive reimbursement structures for advanced medical procedures encourage adoption.
Challenges Impacting Medical Nitinol Braiding Solutions Growth:
- High Cost of Raw Materials: Nitinol, particularly high-purity grades, can be expensive, impacting the overall cost of braided devices.
- Stringent Regulatory Approvals: Navigating complex regulatory pathways for medical devices can be time-consuming and costly.
- Technical Expertise Required: Manufacturing intricate nitinol braids demands specialized machinery and highly skilled labor.
- Competition from Alternative Materials: While nitinol offers unique advantages, alternative materials may be cost-effective in certain applications.
Growth Drivers in the Medical Nitinol Braiding Solutions Market
The medical nitinol braiding solutions market is propelled by significant growth drivers, prominently featuring technological innovation in material science and manufacturing processes. Advancements in braiding techniques enable the creation of increasingly sophisticated and precisely engineered medical devices with enhanced flexibility and superior performance. The escalating global burden of chronic diseases, particularly cardiovascular ailments and orthopedic disorders, directly translates into a higher demand for minimally invasive treatment options that nitinol braiding excels at providing. Furthermore, a pronounced shift in patient and clinician preference towards minimally invasive procedures, driven by their benefits of reduced recovery times and improved patient outcomes, strongly favors the adoption of these advanced braided solutions. The presence of supportive reimbursement policies for these innovative medical interventions further bolsters market growth by ensuring accessibility and affordability.
Challenges Impacting Medical Nitinol Braiding Solutions Growth
Despite its promising growth trajectory, the medical nitinol braiding solutions market faces several critical challenges that impact its expansion. The inherent high cost of premium nitinol alloys and the specialized, capital-intensive manufacturing processes required for braiding contribute to elevated production costs, which can translate into higher device prices. Navigating the stringent and evolving regulatory landscapes of different countries for medical device approvals presents a significant hurdle, often demanding extensive clinical trials and documentation, thereby increasing time-to-market and R&D expenditures. The specialized technical expertise required for designing and manufacturing complex nitinol braids also poses a challenge, limiting the number of capable manufacturers and potentially creating supply chain bottlenecks. Moreover, while nitinol offers distinct advantages, it faces competition from alternative materials in certain applications, particularly where cost is a paramount consideration, necessitating continuous innovation to maintain its competitive edge.
Key Players Shaping the Medical Nitinol Braiding Solutions Market
- Integer Holdings
- Nordson Medical
- Medical Device Components
- Fort Wayne Metals
- Confluent Medical
- Admedes
- KOS
- Custom Wire Technologies
- Alleima
- Resonetics
- Ingpuls
- Wytech Industries
- AccuPath Group
- KT Medical
- Peiertech
Significant Medical Nitinol Braiding Solutions Industry Milestones
- 2019 (Q1): Launch of next-generation braided nitinol stent with enhanced deliverability and radial strength.
- 2020 (Q3): Acquisition of a leading nitinol wire manufacturer by a major medical device company to secure supply chain.
- 2021 (Q2): Introduction of advanced computational modeling software for optimizing nitinol braid designs.
- 2022 (Q4): FDA approval for a novel braided nitinol guidewire for complex neurovascular interventions.
- 2023 (Q1): Development of bioresorbable nitinol alloys for temporary implant applications.
- 2023 (Q3): Strategic partnership formed between a nitinol supplier and a braiding technology provider to accelerate innovation.
- 2024 (Q1): CE marking obtained for a new line of braided nitinol vascular grafts.
- 2024 (Q3): Significant investment announced in R&D for advanced nitinol braiding for orthopedic applications.
Future Outlook for Medical Nitinol Braiding Solutions Market
The future outlook for the medical nitinol braiding solutions market is exceptionally bright, driven by ongoing technological advancements and an ever-increasing demand for sophisticated, minimally invasive medical devices. Strategic opportunities lie in the expansion of applications into emerging fields like neuromodulation and advanced drug delivery systems, where nitinol's unique properties can be leveraged for highly precise and controlled interventions. The market will likely witness further consolidation through strategic acquisitions and collaborations, fostering innovation and streamlining supply chains. The increasing emphasis on personalized medicine will also create a surge in demand for customized nitinol braiding solutions tailored to individual patient anatomies and therapeutic needs. With an estimated market size projected to reach approximately $11,000 million by 2033, the market is poised for sustained and robust growth, offering significant potential for stakeholders across the value chain.
Medical Nitinol Braiding Solutions Segmentation
-
1. Application
- 1.1. Cardiovascular Field
- 1.2. Orthopedic
- 1.3. Dental
- 1.4. Others
-
2. Types
- 2.1. Stents and Vascular Devices
- 2.2. Guidewires
- 2.3. Others
Medical Nitinol Braiding Solutions Segmentation By Geography
-
1. North America
- 1.1. United States
- 1.2. Canada
- 1.3. Mexico
-
2. South America
- 2.1. Brazil
- 2.2. Argentina
- 2.3. Rest of South America
-
3. Europe
- 3.1. United Kingdom
- 3.2. Germany
- 3.3. France
- 3.4. Italy
- 3.5. Spain
- 3.6. Russia
- 3.7. Benelux
- 3.8. Nordics
- 3.9. Rest of Europe
-
4. Middle East & Africa
- 4.1. Turkey
- 4.2. Israel
- 4.3. GCC
- 4.4. North Africa
- 4.5. South Africa
- 4.6. Rest of Middle East & Africa
-
5. Asia Pacific
- 5.1. China
- 5.2. India
- 5.3. Japan
- 5.4. South Korea
- 5.5. ASEAN
- 5.6. Oceania
- 5.7. Rest of Asia Pacific
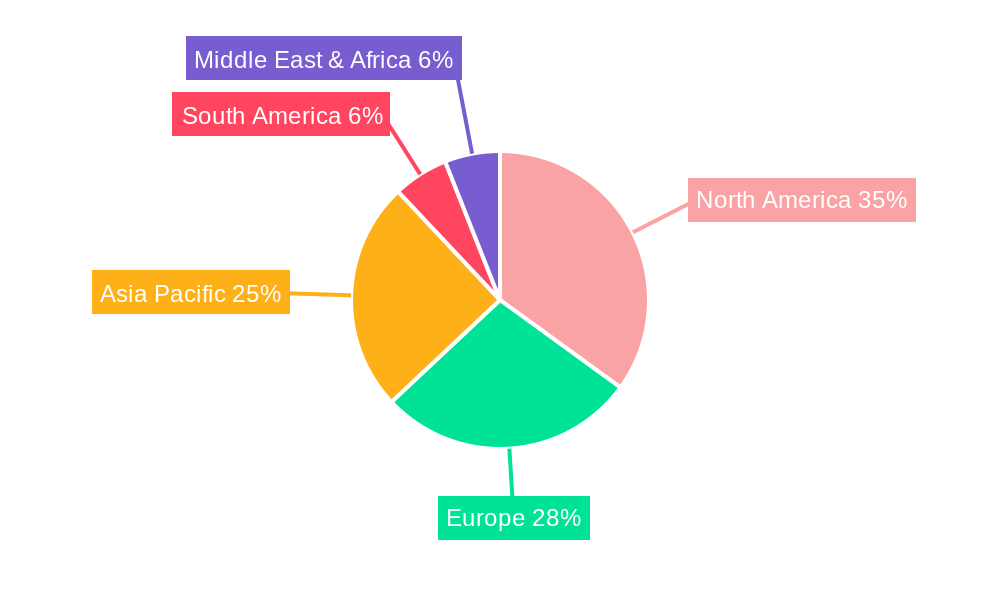
Medical Nitinol Braiding Solutions Regional Market Share

Geographic Coverage of Medical Nitinol Braiding Solutions
Medical Nitinol Braiding Solutions REPORT HIGHLIGHTS
| Aspects | Details |
|---|---|
| Study Period | 2020-2034 |
| Base Year | 2025 |
| Estimated Year | 2026 |
| Forecast Period | 2026-2034 |
| Historical Period | 2020-2025 |
| Growth Rate | CAGR of 8% from 2020-2034 |
| Segmentation |
|
Table of Contents
- 1. Introduction
- 1.1. Research Scope
- 1.2. Market Segmentation
- 1.3. Research Methodology
- 1.4. Definitions and Assumptions
- 2. Executive Summary
- 2.1. Introduction
- 3. Market Dynamics
- 3.1. Introduction
- 3.2. Market Drivers
- 3.3. Market Restrains
- 3.4. Market Trends
- 4. Market Factor Analysis
- 4.1. Porters Five Forces
- 4.2. Supply/Value Chain
- 4.3. PESTEL analysis
- 4.4. Market Entropy
- 4.5. Patent/Trademark Analysis
- 5. Global Medical Nitinol Braiding Solutions Analysis, Insights and Forecast, 2020-2032
- 5.1. Market Analysis, Insights and Forecast - by Application
- 5.1.1. Cardiovascular Field
- 5.1.2. Orthopedic
- 5.1.3. Dental
- 5.1.4. Others
- 5.2. Market Analysis, Insights and Forecast - by Types
- 5.2.1. Stents and Vascular Devices
- 5.2.2. Guidewires
- 5.2.3. Others
- 5.3. Market Analysis, Insights and Forecast - by Region
- 5.3.1. North America
- 5.3.2. South America
- 5.3.3. Europe
- 5.3.4. Middle East & Africa
- 5.3.5. Asia Pacific
- 5.1. Market Analysis, Insights and Forecast - by Application
- 6. North America Medical Nitinol Braiding Solutions Analysis, Insights and Forecast, 2020-2032
- 6.1. Market Analysis, Insights and Forecast - by Application
- 6.1.1. Cardiovascular Field
- 6.1.2. Orthopedic
- 6.1.3. Dental
- 6.1.4. Others
- 6.2. Market Analysis, Insights and Forecast - by Types
- 6.2.1. Stents and Vascular Devices
- 6.2.2. Guidewires
- 6.2.3. Others
- 6.1. Market Analysis, Insights and Forecast - by Application
- 7. South America Medical Nitinol Braiding Solutions Analysis, Insights and Forecast, 2020-2032
- 7.1. Market Analysis, Insights and Forecast - by Application
- 7.1.1. Cardiovascular Field
- 7.1.2. Orthopedic
- 7.1.3. Dental
- 7.1.4. Others
- 7.2. Market Analysis, Insights and Forecast - by Types
- 7.2.1. Stents and Vascular Devices
- 7.2.2. Guidewires
- 7.2.3. Others
- 7.1. Market Analysis, Insights and Forecast - by Application
- 8. Europe Medical Nitinol Braiding Solutions Analysis, Insights and Forecast, 2020-2032
- 8.1. Market Analysis, Insights and Forecast - by Application
- 8.1.1. Cardiovascular Field
- 8.1.2. Orthopedic
- 8.1.3. Dental
- 8.1.4. Others
- 8.2. Market Analysis, Insights and Forecast - by Types
- 8.2.1. Stents and Vascular Devices
- 8.2.2. Guidewires
- 8.2.3. Others
- 8.1. Market Analysis, Insights and Forecast - by Application
- 9. Middle East & Africa Medical Nitinol Braiding Solutions Analysis, Insights and Forecast, 2020-2032
- 9.1. Market Analysis, Insights and Forecast - by Application
- 9.1.1. Cardiovascular Field
- 9.1.2. Orthopedic
- 9.1.3. Dental
- 9.1.4. Others
- 9.2. Market Analysis, Insights and Forecast - by Types
- 9.2.1. Stents and Vascular Devices
- 9.2.2. Guidewires
- 9.2.3. Others
- 9.1. Market Analysis, Insights and Forecast - by Application
- 10. Asia Pacific Medical Nitinol Braiding Solutions Analysis, Insights and Forecast, 2020-2032
- 10.1. Market Analysis, Insights and Forecast - by Application
- 10.1.1. Cardiovascular Field
- 10.1.2. Orthopedic
- 10.1.3. Dental
- 10.1.4. Others
- 10.2. Market Analysis, Insights and Forecast - by Types
- 10.2.1. Stents and Vascular Devices
- 10.2.2. Guidewires
- 10.2.3. Others
- 10.1. Market Analysis, Insights and Forecast - by Application
- 11. Competitive Analysis
- 11.1. Global Market Share Analysis 2025
- 11.2. Company Profiles
- 11.2.1 Integer Holdings
- 11.2.1.1. Overview
- 11.2.1.2. Products
- 11.2.1.3. SWOT Analysis
- 11.2.1.4. Recent Developments
- 11.2.1.5. Financials (Based on Availability)
- 11.2.2 Nordson Medical
- 11.2.2.1. Overview
- 11.2.2.2. Products
- 11.2.2.3. SWOT Analysis
- 11.2.2.4. Recent Developments
- 11.2.2.5. Financials (Based on Availability)
- 11.2.3 Medical Device Components
- 11.2.3.1. Overview
- 11.2.3.2. Products
- 11.2.3.3. SWOT Analysis
- 11.2.3.4. Recent Developments
- 11.2.3.5. Financials (Based on Availability)
- 11.2.4 Fort Wayne Metals
- 11.2.4.1. Overview
- 11.2.4.2. Products
- 11.2.4.3. SWOT Analysis
- 11.2.4.4. Recent Developments
- 11.2.4.5. Financials (Based on Availability)
- 11.2.5 Confluent Medical
- 11.2.5.1. Overview
- 11.2.5.2. Products
- 11.2.5.3. SWOT Analysis
- 11.2.5.4. Recent Developments
- 11.2.5.5. Financials (Based on Availability)
- 11.2.6 Admedes
- 11.2.6.1. Overview
- 11.2.6.2. Products
- 11.2.6.3. SWOT Analysis
- 11.2.6.4. Recent Developments
- 11.2.6.5. Financials (Based on Availability)
- 11.2.7 KOS
- 11.2.7.1. Overview
- 11.2.7.2. Products
- 11.2.7.3. SWOT Analysis
- 11.2.7.4. Recent Developments
- 11.2.7.5. Financials (Based on Availability)
- 11.2.8 Custom Wire technologies
- 11.2.8.1. Overview
- 11.2.8.2. Products
- 11.2.8.3. SWOT Analysis
- 11.2.8.4. Recent Developments
- 11.2.8.5. Financials (Based on Availability)
- 11.2.9 Alleima
- 11.2.9.1. Overview
- 11.2.9.2. Products
- 11.2.9.3. SWOT Analysis
- 11.2.9.4. Recent Developments
- 11.2.9.5. Financials (Based on Availability)
- 11.2.10 Resonetics
- 11.2.10.1. Overview
- 11.2.10.2. Products
- 11.2.10.3. SWOT Analysis
- 11.2.10.4. Recent Developments
- 11.2.10.5. Financials (Based on Availability)
- 11.2.11 Ingpuls
- 11.2.11.1. Overview
- 11.2.11.2. Products
- 11.2.11.3. SWOT Analysis
- 11.2.11.4. Recent Developments
- 11.2.11.5. Financials (Based on Availability)
- 11.2.12 Wytech Industries
- 11.2.12.1. Overview
- 11.2.12.2. Products
- 11.2.12.3. SWOT Analysis
- 11.2.12.4. Recent Developments
- 11.2.12.5. Financials (Based on Availability)
- 11.2.13 AccuPath Group
- 11.2.13.1. Overview
- 11.2.13.2. Products
- 11.2.13.3. SWOT Analysis
- 11.2.13.4. Recent Developments
- 11.2.13.5. Financials (Based on Availability)
- 11.2.14 KT Medical
- 11.2.14.1. Overview
- 11.2.14.2. Products
- 11.2.14.3. SWOT Analysis
- 11.2.14.4. Recent Developments
- 11.2.14.5. Financials (Based on Availability)
- 11.2.15 Peiertech
- 11.2.15.1. Overview
- 11.2.15.2. Products
- 11.2.15.3. SWOT Analysis
- 11.2.15.4. Recent Developments
- 11.2.15.5. Financials (Based on Availability)
- 11.2.1 Integer Holdings
List of Figures
- Figure 1: Global Medical Nitinol Braiding Solutions Revenue Breakdown (undefined, %) by Region 2025 & 2033
- Figure 2: North America Medical Nitinol Braiding Solutions Revenue (undefined), by Application 2025 & 2033
- Figure 3: North America Medical Nitinol Braiding Solutions Revenue Share (%), by Application 2025 & 2033
- Figure 4: North America Medical Nitinol Braiding Solutions Revenue (undefined), by Types 2025 & 2033
- Figure 5: North America Medical Nitinol Braiding Solutions Revenue Share (%), by Types 2025 & 2033
- Figure 6: North America Medical Nitinol Braiding Solutions Revenue (undefined), by Country 2025 & 2033
- Figure 7: North America Medical Nitinol Braiding Solutions Revenue Share (%), by Country 2025 & 2033
- Figure 8: South America Medical Nitinol Braiding Solutions Revenue (undefined), by Application 2025 & 2033
- Figure 9: South America Medical Nitinol Braiding Solutions Revenue Share (%), by Application 2025 & 2033
- Figure 10: South America Medical Nitinol Braiding Solutions Revenue (undefined), by Types 2025 & 2033
- Figure 11: South America Medical Nitinol Braiding Solutions Revenue Share (%), by Types 2025 & 2033
- Figure 12: South America Medical Nitinol Braiding Solutions Revenue (undefined), by Country 2025 & 2033
- Figure 13: South America Medical Nitinol Braiding Solutions Revenue Share (%), by Country 2025 & 2033
- Figure 14: Europe Medical Nitinol Braiding Solutions Revenue (undefined), by Application 2025 & 2033
- Figure 15: Europe Medical Nitinol Braiding Solutions Revenue Share (%), by Application 2025 & 2033
- Figure 16: Europe Medical Nitinol Braiding Solutions Revenue (undefined), by Types 2025 & 2033
- Figure 17: Europe Medical Nitinol Braiding Solutions Revenue Share (%), by Types 2025 & 2033
- Figure 18: Europe Medical Nitinol Braiding Solutions Revenue (undefined), by Country 2025 & 2033
- Figure 19: Europe Medical Nitinol Braiding Solutions Revenue Share (%), by Country 2025 & 2033
- Figure 20: Middle East & Africa Medical Nitinol Braiding Solutions Revenue (undefined), by Application 2025 & 2033
- Figure 21: Middle East & Africa Medical Nitinol Braiding Solutions Revenue Share (%), by Application 2025 & 2033
- Figure 22: Middle East & Africa Medical Nitinol Braiding Solutions Revenue (undefined), by Types 2025 & 2033
- Figure 23: Middle East & Africa Medical Nitinol Braiding Solutions Revenue Share (%), by Types 2025 & 2033
- Figure 24: Middle East & Africa Medical Nitinol Braiding Solutions Revenue (undefined), by Country 2025 & 2033
- Figure 25: Middle East & Africa Medical Nitinol Braiding Solutions Revenue Share (%), by Country 2025 & 2033
- Figure 26: Asia Pacific Medical Nitinol Braiding Solutions Revenue (undefined), by Application 2025 & 2033
- Figure 27: Asia Pacific Medical Nitinol Braiding Solutions Revenue Share (%), by Application 2025 & 2033
- Figure 28: Asia Pacific Medical Nitinol Braiding Solutions Revenue (undefined), by Types 2025 & 2033
- Figure 29: Asia Pacific Medical Nitinol Braiding Solutions Revenue Share (%), by Types 2025 & 2033
- Figure 30: Asia Pacific Medical Nitinol Braiding Solutions Revenue (undefined), by Country 2025 & 2033
- Figure 31: Asia Pacific Medical Nitinol Braiding Solutions Revenue Share (%), by Country 2025 & 2033
List of Tables
- Table 1: Global Medical Nitinol Braiding Solutions Revenue undefined Forecast, by Application 2020 & 2033
- Table 2: Global Medical Nitinol Braiding Solutions Revenue undefined Forecast, by Types 2020 & 2033
- Table 3: Global Medical Nitinol Braiding Solutions Revenue undefined Forecast, by Region 2020 & 2033
- Table 4: Global Medical Nitinol Braiding Solutions Revenue undefined Forecast, by Application 2020 & 2033
- Table 5: Global Medical Nitinol Braiding Solutions Revenue undefined Forecast, by Types 2020 & 2033
- Table 6: Global Medical Nitinol Braiding Solutions Revenue undefined Forecast, by Country 2020 & 2033
- Table 7: United States Medical Nitinol Braiding Solutions Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 8: Canada Medical Nitinol Braiding Solutions Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 9: Mexico Medical Nitinol Braiding Solutions Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 10: Global Medical Nitinol Braiding Solutions Revenue undefined Forecast, by Application 2020 & 2033
- Table 11: Global Medical Nitinol Braiding Solutions Revenue undefined Forecast, by Types 2020 & 2033
- Table 12: Global Medical Nitinol Braiding Solutions Revenue undefined Forecast, by Country 2020 & 2033
- Table 13: Brazil Medical Nitinol Braiding Solutions Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 14: Argentina Medical Nitinol Braiding Solutions Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 15: Rest of South America Medical Nitinol Braiding Solutions Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 16: Global Medical Nitinol Braiding Solutions Revenue undefined Forecast, by Application 2020 & 2033
- Table 17: Global Medical Nitinol Braiding Solutions Revenue undefined Forecast, by Types 2020 & 2033
- Table 18: Global Medical Nitinol Braiding Solutions Revenue undefined Forecast, by Country 2020 & 2033
- Table 19: United Kingdom Medical Nitinol Braiding Solutions Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 20: Germany Medical Nitinol Braiding Solutions Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 21: France Medical Nitinol Braiding Solutions Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 22: Italy Medical Nitinol Braiding Solutions Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 23: Spain Medical Nitinol Braiding Solutions Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 24: Russia Medical Nitinol Braiding Solutions Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 25: Benelux Medical Nitinol Braiding Solutions Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 26: Nordics Medical Nitinol Braiding Solutions Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 27: Rest of Europe Medical Nitinol Braiding Solutions Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 28: Global Medical Nitinol Braiding Solutions Revenue undefined Forecast, by Application 2020 & 2033
- Table 29: Global Medical Nitinol Braiding Solutions Revenue undefined Forecast, by Types 2020 & 2033
- Table 30: Global Medical Nitinol Braiding Solutions Revenue undefined Forecast, by Country 2020 & 2033
- Table 31: Turkey Medical Nitinol Braiding Solutions Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 32: Israel Medical Nitinol Braiding Solutions Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 33: GCC Medical Nitinol Braiding Solutions Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 34: North Africa Medical Nitinol Braiding Solutions Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 35: South Africa Medical Nitinol Braiding Solutions Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 36: Rest of Middle East & Africa Medical Nitinol Braiding Solutions Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 37: Global Medical Nitinol Braiding Solutions Revenue undefined Forecast, by Application 2020 & 2033
- Table 38: Global Medical Nitinol Braiding Solutions Revenue undefined Forecast, by Types 2020 & 2033
- Table 39: Global Medical Nitinol Braiding Solutions Revenue undefined Forecast, by Country 2020 & 2033
- Table 40: China Medical Nitinol Braiding Solutions Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 41: India Medical Nitinol Braiding Solutions Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 42: Japan Medical Nitinol Braiding Solutions Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 43: South Korea Medical Nitinol Braiding Solutions Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 44: ASEAN Medical Nitinol Braiding Solutions Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 45: Oceania Medical Nitinol Braiding Solutions Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 46: Rest of Asia Pacific Medical Nitinol Braiding Solutions Revenue (undefined) Forecast, by Application 2020 & 2033
Frequently Asked Questions
1. What is the projected Compound Annual Growth Rate (CAGR) of the Medical Nitinol Braiding Solutions?
The projected CAGR is approximately 8%.
2. Which companies are prominent players in the Medical Nitinol Braiding Solutions?
Key companies in the market include Integer Holdings, Nordson Medical, Medical Device Components, Fort Wayne Metals, Confluent Medical, Admedes, KOS, Custom Wire technologies, Alleima, Resonetics, Ingpuls, Wytech Industries, AccuPath Group, KT Medical, Peiertech.
3. What are the main segments of the Medical Nitinol Braiding Solutions?
The market segments include Application, Types.
4. Can you provide details about the market size?
The market size is estimated to be USD XXX N/A as of 2022.
5. What are some drivers contributing to market growth?
N/A
6. What are the notable trends driving market growth?
N/A
7. Are there any restraints impacting market growth?
N/A
8. Can you provide examples of recent developments in the market?
N/A
9. What pricing options are available for accessing the report?
Pricing options include single-user, multi-user, and enterprise licenses priced at USD 3950.00, USD 5925.00, and USD 7900.00 respectively.
10. Is the market size provided in terms of value or volume?
The market size is provided in terms of value, measured in N/A.
11. Are there any specific market keywords associated with the report?
Yes, the market keyword associated with the report is "Medical Nitinol Braiding Solutions," which aids in identifying and referencing the specific market segment covered.
12. How do I determine which pricing option suits my needs best?
The pricing options vary based on user requirements and access needs. Individual users may opt for single-user licenses, while businesses requiring broader access may choose multi-user or enterprise licenses for cost-effective access to the report.
13. Are there any additional resources or data provided in the Medical Nitinol Braiding Solutions report?
While the report offers comprehensive insights, it's advisable to review the specific contents or supplementary materials provided to ascertain if additional resources or data are available.
14. How can I stay updated on further developments or reports in the Medical Nitinol Braiding Solutions?
To stay informed about further developments, trends, and reports in the Medical Nitinol Braiding Solutions, consider subscribing to industry newsletters, following relevant companies and organizations, or regularly checking reputable industry news sources and publications.
Methodology
Step 1 - Identification of Relevant Samples Size from Population Database



Step 2 - Approaches for Defining Global Market Size (Value, Volume* & Price*)

Note*: In applicable scenarios
Step 3 - Data Sources
Primary Research
- Web Analytics
- Survey Reports
- Research Institute
- Latest Research Reports
- Opinion Leaders
Secondary Research
- Annual Reports
- White Paper
- Latest Press Release
- Industry Association
- Paid Database
- Investor Presentations

Step 4 - Data Triangulation
Involves using different sources of information in order to increase the validity of a study
These sources are likely to be stakeholders in a program - participants, other researchers, program staff, other community members, and so on.
Then we put all data in single framework & apply various statistical tools to find out the dynamic on the market.
During the analysis stage, feedback from the stakeholder groups would be compared to determine areas of agreement as well as areas of divergence


