Key Insights
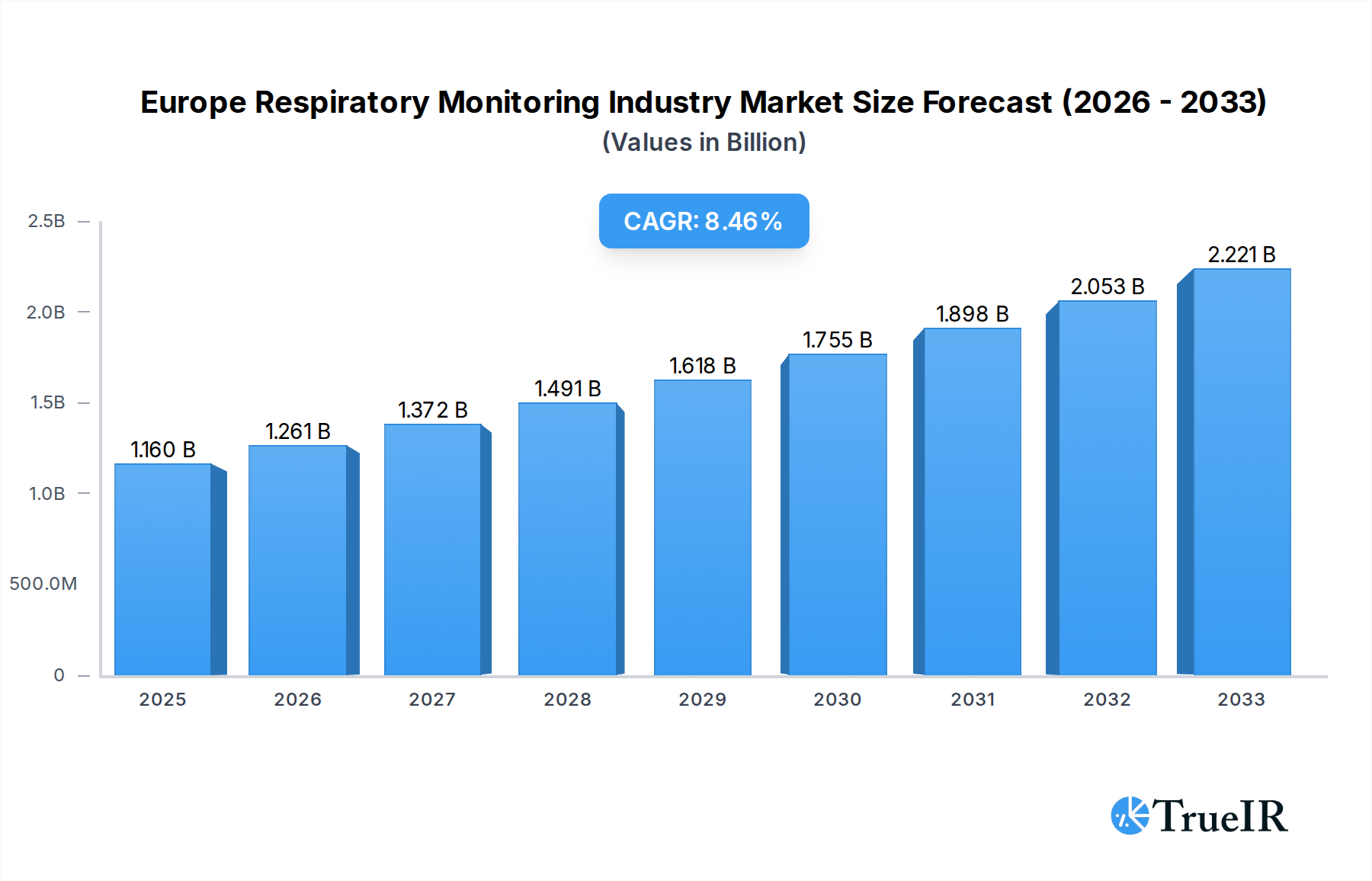
The European Respiratory Monitoring Industry is poised for significant expansion, driven by an increasing prevalence of respiratory diseases, an aging population, and advancements in diagnostic technologies. The market was valued at $1.16 billion in the base year of 2025 and is projected to grow at a robust Compound Annual Growth Rate (CAGR) of 8.60% during the forecast period of 2025-2033. This growth trajectory is fueled by a heightened awareness of respiratory health, leading to increased demand for early detection and continuous monitoring of conditions like COPD, asthma, and sleep apnea. Furthermore, the integration of connected devices and digital health solutions is enhancing patient outcomes and healthcare efficiency, acting as a substantial catalyst for market expansion. The growing adoption of portable and user-friendly respiratory monitoring devices, coupled with supportive regulatory frameworks and reimbursement policies, further underpins this optimistic market outlook.
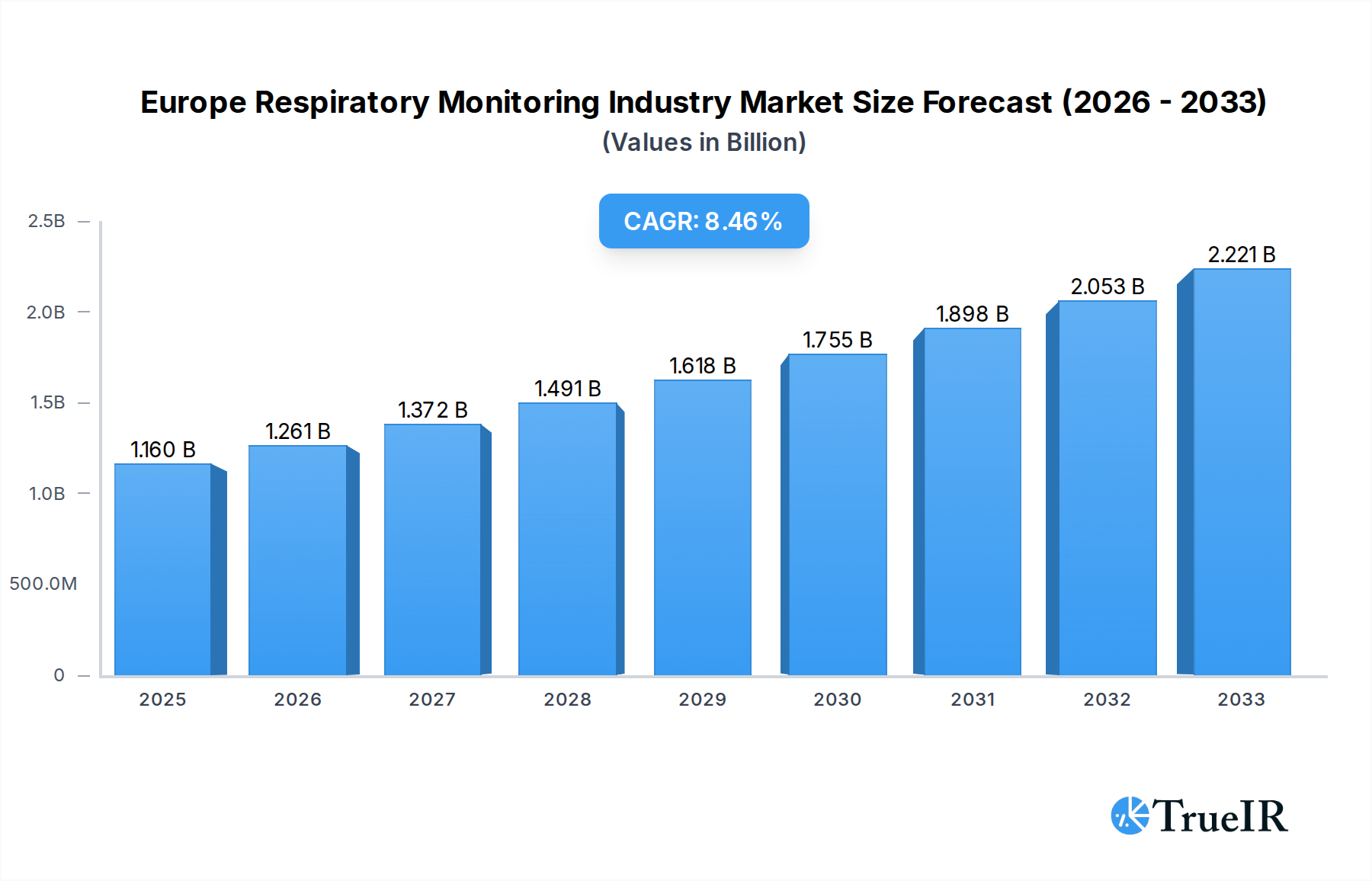
Europe Respiratory Monitoring Industry Market Size (In Billion)

The market segmentation reveals a diverse landscape, with Spirometers and Pulse Oximeters being key device types currently leading in adoption, reflecting their established roles in diagnosing and managing chronic respiratory conditions. However, the increasing focus on sleep disorders is driving considerable growth in Sleep Test Devices. Major industry players like Medtronic, GE Healthcare, and Koninklijke Philips N.V. are actively investing in research and development to innovate their product portfolios and expand their market reach across key European regions, including Germany, the United Kingdom, and France, which represent substantial market shares. While the market benefits from strong demand drivers, potential restraints such as high initial device costs for advanced technologies and the need for skilled healthcare professionals to interpret complex data could present challenges. Nevertheless, the overarching trend towards proactive respiratory care and technological innovation ensures a dynamic and expanding market in the coming years.
Europe Respiratory Monitoring Industry Company Market Share

This comprehensive report delves into the dynamic Europe respiratory monitoring market, a critical segment of the healthcare industry experiencing rapid technological advancements and increasing demand. Analyzing the respiratory monitoring devices market from 2019 to 2033, with a base year of 2025 and a forecast period of 2025–2033, this study provides unparalleled insights into market structure, competitive landscape, key trends, and future opportunities. Leveraging high-volume keywords such as respiratory diagnostics, pulmonary function testing, sleep apnea devices, and lung function monitors, this report is meticulously designed for industry stakeholders seeking to understand the intricate workings and growth trajectory of the European medical device market focused on respiratory health.
The Europe respiratory monitoring industry encompasses a wide array of essential diagnostic and therapeutic tools designed to assess, manage, and treat a spectrum of respiratory conditions. As the prevalence of chronic respiratory diseases like COPD, asthma, and sleep apnea continues to rise across the continent, the demand for advanced and accessible respiratory monitoring solutions is escalating. This report addresses the critical need for a thorough understanding of the market's current state and future potential, providing actionable intelligence for manufacturers, distributors, healthcare providers, and investors.
Europe Respiratory Monitoring Industry Market Structure & Competitive Landscape
The Europe respiratory monitoring market exhibits a moderately consolidated structure, with leading players like Medtronic, GE Healthcare, and Koninklijke Philips N.V. holding significant market share. Innovation is a key driver, fueled by continuous research and development in areas such as miniaturization, wireless connectivity, and AI-powered diagnostics. Regulatory frameworks, including those set by the European Medicines Agency (EMA), play a crucial role in shaping product approvals and market access. While a few dominant players exist, the market also features a growing number of specialized manufacturers, particularly in segments like spirometry and portable monitoring solutions. Product substitutes are limited due to the specialized nature of respiratory monitoring, but advancements in telemedicine and remote patient monitoring are creating new avenues for device utilization. End-user segmentation is diverse, encompassing hospitals, clinics, home healthcare settings, and diagnostic laboratories. Merger and acquisition (M&A) activities are observed as companies seek to expand their product portfolios, gain market access, and achieve economies of scale. For instance, the historical period from 2019–2024 saw an estimated xx M&A transactions aimed at consolidating market positions and acquiring innovative technologies. The market concentration is estimated to be in the moderate to high range, with the top 5 companies accounting for approximately 60% of the market value in the base year of 2025.
Europe Respiratory Monitoring Industry Market Trends & Opportunities
The Europe respiratory monitoring market is poised for significant expansion, driven by a confluence of demographic shifts, technological advancements, and evolving healthcare paradigms. The market size is projected to grow from approximately XX Million in the base year of 2025 to over XX Million by 2033, exhibiting a Compound Annual Growth Rate (CAGR) of approximately X.XX%. This growth is underpinned by the increasing prevalence of respiratory diseases, including asthma, Chronic Obstructive Pulmonary Disease (COPD), and sleep disorders like sleep apnea, across European nations. An aging population and rising air pollution levels further exacerbate the burden of these conditions, creating a sustained demand for effective diagnostic and monitoring tools.
Technological innovation is a primary catalyst, with a discernible shift towards connected and portable devices that enable remote patient monitoring and telehealth solutions. The integration of artificial intelligence (AI) and machine learning (ML) into respiratory monitoring systems is enhancing diagnostic accuracy, predicting disease progression, and personalizing treatment plans. For example, the development of smart spirometers and wearable pulse oximeters is empowering patients to actively participate in their care and allowing healthcare providers to intervene proactively. Consumer preferences are increasingly leaning towards user-friendly, non-invasive, and home-based monitoring solutions, driving the adoption of devices like home sleep apnea test devices and personal spirometers. The COVID-19 pandemic has also accelerated the adoption of remote monitoring technologies, highlighting their efficacy in managing patients outside traditional clinical settings.
The competitive landscape is dynamic, with established players continually innovating and new entrants bringing disruptive technologies to the market. Opportunities abound for companies that can offer integrated solutions, leverage data analytics for predictive insights, and provide cost-effective alternatives that improve patient outcomes. The focus is shifting from purely diagnostic tools to comprehensive management platforms. The market penetration rate for advanced respiratory monitoring devices in home care settings is expected to rise from approximately XX% in 2025 to over XX% by 2033, reflecting the growing acceptance of these technologies by both patients and healthcare providers. Furthermore, the increasing focus on preventative healthcare and early disease detection is creating significant growth opportunities for diagnostic spirometers and gas analyzers used in regular health check-ups. The market is also witnessing a growing demand for specialized devices catering to specific patient populations, such as pediatric respiratory monitors and devices for individuals with neuromuscular disorders. The expansion of reimbursement policies for remote patient monitoring and telehealth services across European countries is further bolstering the growth prospects of this market.
Dominant Markets & Segments in Europe Respiratory Monitoring Industry
The Europe respiratory monitoring industry is characterized by the significant dominance of specific regions and device segments, driven by varying healthcare infrastructure, disease prevalence, and regulatory landscapes. Germany and the United Kingdom consistently emerge as leading markets due to their advanced healthcare systems, high disposable incomes, and proactive adoption of medical technologies. France and Italy also represent substantial markets, with increasing investment in respiratory health management.
Within the Type of Device segmentation, Spirometers currently hold a dominant position, driven by their foundational role in diagnosing and managing common respiratory conditions such as asthma and COPD. The demand for advanced spirometers, including portable and connected models, is robust. However, Sleep Test Devices are experiencing the fastest growth rate. This surge is directly attributable to the escalating prevalence of sleep apnea across Europe and increasing awareness among both patients and healthcare professionals. The accessibility and increasing accuracy of home-based sleep testing devices are significant growth drivers in this segment.
Pulse Oximeters also represent a substantial segment, particularly given their widespread use in clinical settings and the growing adoption of home-based monitoring, amplified by events like the COVID-19 pandemic. Gas Analyzers are crucial for specialized diagnostic procedures and are widely used in pulmonary function laboratories and research institutions. Peak Flow Meters, while a more traditional device, continue to find application in the routine management of asthma, especially among pediatric populations. Capnographs, essential for critical care and anesthesia, maintain a steady demand in hospital settings. The "Others" category, which includes devices like respiratory muscle testers and plethysmography systems, caters to niche applications and specialized diagnostic needs.
Key growth drivers for dominant segments include:
Spirometers:
- High prevalence of COPD and asthma across Europe.
- Increasing focus on early diagnosis and preventative healthcare.
- Technological advancements leading to more portable and user-friendly devices.
- Reimbursement policies supporting pulmonary function testing.
Sleep Test Devices:
- Rising incidence of undiagnosed sleep apnea.
- Growing awareness of the health implications of sleep disorders.
- Technological advancements in home sleep apnea testing (HSAT).
- Demand for discreet and convenient diagnostic solutions.
- Integration with telehealth platforms for remote diagnosis and management.
Pulse Oximeters:
- Continued need in critical care and post-operative monitoring.
- Increased adoption in home healthcare and chronic disease management.
- Growing demand for wearable and continuous monitoring solutions.
- Public health initiatives promoting oxygen saturation monitoring.
The market dominance is further amplified by robust healthcare spending in key European countries, government initiatives promoting respiratory health awareness and screening programs, and a well-established network of medical device distributors and healthcare providers. The increasing integration of these devices with electronic health records (EHRs) and telehealth platforms is also playing a pivotal role in their market penetration and growth.
Europe Respiratory Monitoring Industry Product Analysis
The Europe respiratory monitoring industry is characterized by continuous product innovation aimed at enhancing diagnostic accuracy, portability, and patient comfort. Spirometers are evolving with integrated software for intuitive data analysis and reporting, alongside wireless connectivity options. Peak flow meters are becoming more digital and user-friendly, often incorporating motivational feedback mechanisms. Sleep test devices are transitioning from complex polysomnography (PSG) to more accessible home sleep apnea testing (HSAT) devices, emphasizing ease of use and data integration with cloud platforms for remote analysis. Gas analyzers are seeing improvements in sensitivity and speed. Pulse oximeters are increasingly integrated into wearables, offering continuous, non-invasive monitoring. Capnographs are becoming more compact and adaptable for various clinical settings, including emergency transport. These advancements cater to the growing demand for patient-centric solutions and remote patient monitoring, offering competitive advantages through improved clinical utility and user experience.
Key Drivers, Barriers & Challenges in Europe Respiratory Monitoring Industry
Key Drivers: The Europe respiratory monitoring industry is propelled by several key factors. Technologically, advancements in miniaturization, wireless connectivity, and AI are enabling more sophisticated and portable devices. Economically, an aging population and increasing healthcare expenditure across Europe fuel demand. Policy-driven factors, such as government initiatives promoting early diagnosis and management of respiratory diseases, alongside favorable reimbursement policies for remote patient monitoring, are significant growth catalysts. The rising awareness of respiratory health and the increasing burden of chronic respiratory diseases like COPD and asthma also contribute significantly.
Barriers & Challenges: Despite strong growth prospects, the industry faces challenges. Regulatory hurdles, including complex CE marking processes for medical devices, can lead to extended market entry timelines. Supply chain disruptions, as experienced globally, can impact the availability and cost of essential components. Competitive pressures from both established players and emerging innovators necessitate continuous investment in R&D. Furthermore, the cost of advanced respiratory monitoring devices can be a barrier to adoption in resource-constrained healthcare settings, and ensuring data security and privacy in connected devices remains a critical concern. The challenge of educating patients and healthcare professionals on the proper use and interpretation of data from these devices also needs to be addressed.
Growth Drivers in the Europe Respiratory Monitoring Industry Market
The Europe respiratory monitoring industry market is experiencing robust growth driven by several compelling factors. Technological innovation is paramount, with the development of connected spirometers, wearable pulse oximeters, and AI-powered diagnostic tools enhancing accuracy and accessibility. Economically, the rising prevalence of chronic respiratory diseases such as COPD and asthma, coupled with an aging population, creates a sustained and expanding market for monitoring solutions. Regulatory advancements, including supportive reimbursement policies for remote patient monitoring and telehealth services, are further accelerating adoption. Public health initiatives focused on early detection and preventative care also play a crucial role in driving market expansion. The increasing demand for home-based monitoring solutions, facilitated by user-friendly devices, is another significant growth catalyst.
Challenges Impacting Europe Respiratory Monitoring Industry Growth
Several factors pose challenges to the Europe respiratory monitoring industry growth. Regulatory complexities and the stringent approval processes for medical devices across different European Union member states can create significant delays and add to development costs. Supply chain issues, including the availability and cost of raw materials and electronic components, can impact production volumes and pricing strategies. Intense competitive pressures from both established global players and agile regional manufacturers necessitate continuous innovation and cost optimization. Furthermore, the high cost of advanced diagnostic equipment can be a barrier to adoption in some healthcare systems, particularly in Eastern Europe. Ensuring data security and patient privacy in the era of connected devices also presents an ongoing challenge.
Key Players Shaping the Europe Respiratory Monitoring Industry Market
- Maquet (Getinge AB)
- Vitalograph Ltd
- Medtronic
- GE Healthcare
- Koninklijke Philips N V
- COSMED srl
- Resmed
- Siemens Healthcare GmbH
- Koko PFT
- Draegerwerk AG
Significant Europe Respiratory Monitoring Industry Industry Milestones
- March 2022: KoKo, LLC, a United Kingdom-based manufacturer of spirometers, signed a three-year partnership with MAGNET GROUP. This partnership allows MAGNET GROUP to leverage KoKo's high-quality lung function diagnostics and software for spirometry and other devices.
- March 2021: Vyaire Medical launched AioCare, a mobile spirometry system, in more than 15 countries across Europe, Australia, and the Middle East, enhancing the accessibility of pulmonary function testing.
Future Outlook for Europe Respiratory Monitoring Industry Market
The future outlook for the Europe respiratory monitoring industry is exceptionally promising, fueled by ongoing technological advancements and a growing global emphasis on proactive respiratory health management. The continued development of AI-integrated diagnostics, miniaturized wearable devices, and advanced remote patient monitoring platforms will redefine patient care. Opportunities lie in expanding the reach of these technologies into underserved markets and further integrating them with telemedicine infrastructure. The market is expected to witness a significant surge in demand for personalized respiratory care solutions, driven by an increasing understanding of individual patient needs and disease trajectories. Strategic collaborations between device manufacturers, healthcare providers, and research institutions will be crucial for unlocking the full potential of this dynamic market, driving innovation and improving patient outcomes across Europe. The projected market growth from 2025 to 2033 indicates a sustained upward trajectory, solidifying the importance of respiratory monitoring in modern healthcare.
Europe Respiratory Monitoring Industry Segmentation
-
1. Type of Device
- 1.1. Spirometers
- 1.2. Peak Flow Meters
- 1.3. Sleep Test Devices
- 1.4. Gas Analyzers
- 1.5. Pulse Oximeters
- 1.6. Capnographs
- 1.7. Others
Europe Respiratory Monitoring Industry Segmentation By Geography
- 1. Germany
- 2. United Kingdom
- 3. France
- 4. Italy
- 5. Spain
- 6. Rest of Europe
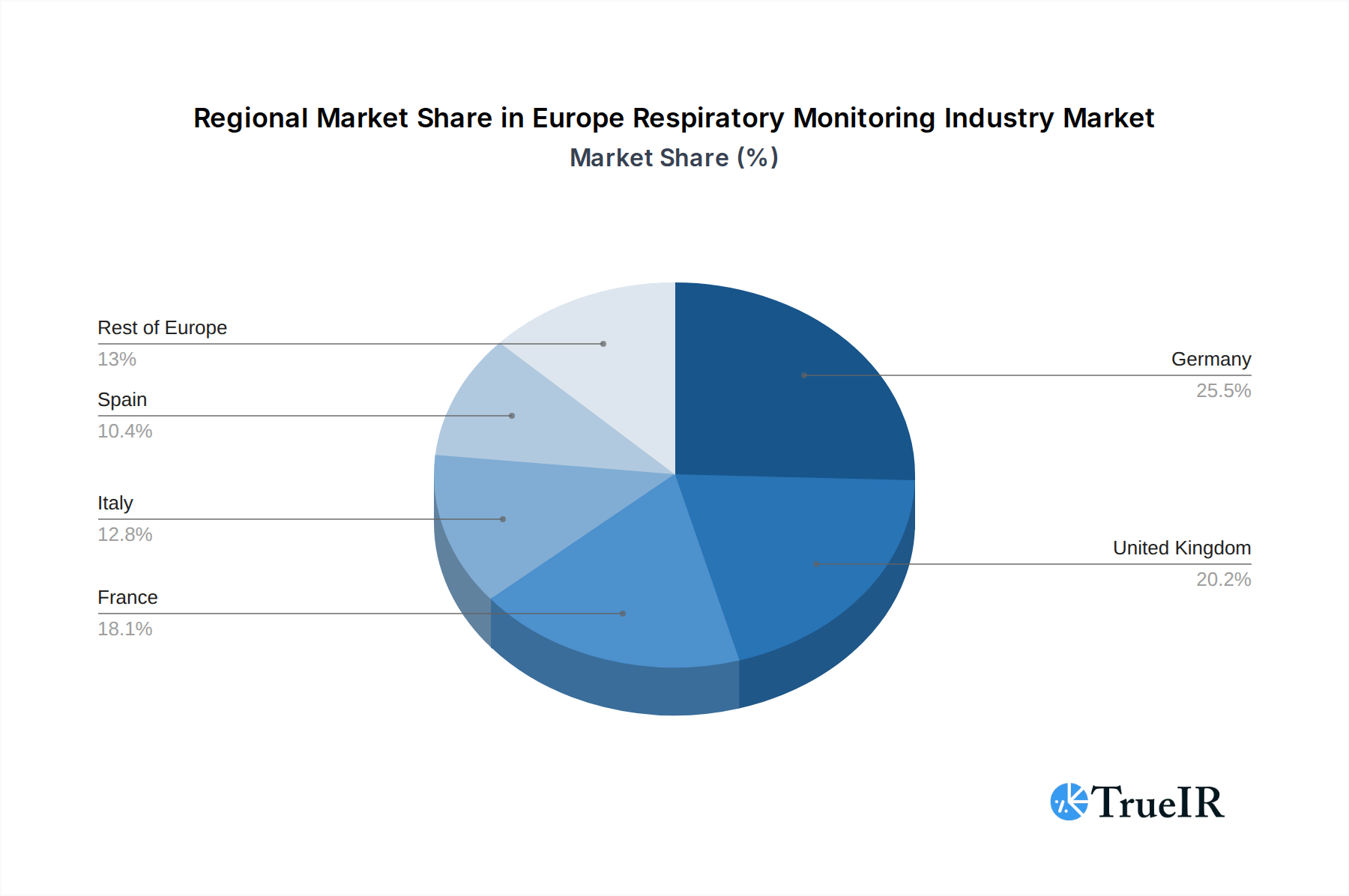
Europe Respiratory Monitoring Industry Regional Market Share

Geographic Coverage of Europe Respiratory Monitoring Industry
Europe Respiratory Monitoring Industry REPORT HIGHLIGHTS
| Aspects | Details |
|---|---|
| Study Period | 2020-2034 |
| Base Year | 2025 |
| Estimated Year | 2026 |
| Forecast Period | 2026-2034 |
| Historical Period | 2020-2025 |
| Growth Rate | CAGR of 8.60% from 2020-2034 |
| Segmentation |
|
Table of Contents
- 1. Introduction
- 1.1. Research Scope
- 1.2. Market Segmentation
- 1.3. Research Objective
- 1.4. Definitions and Assumptions
- 2. Executive Summary
- 2.1. Market Snapshot
- 3. Market Dynamics
- 3.1. Market Drivers
- 3.2. Market Restrains
- 3.3. Market Trends
- 3.4. Market Opportunities
- 4. Market Factor Analysis
- 4.1. Porters Five Forces
- 4.1.1. Bargaining Power of Suppliers
- 4.1.2. Bargaining Power of Buyers
- 4.1.3. Threat of New Entrants
- 4.1.4. Threat of Substitutes
- 4.1.5. Competitive Rivalry
- 4.2. PESTEL analysis
- 4.3. BCG Analysis
- 4.3.1. Stars (High Growth, High Market Share)
- 4.3.2. Cash Cows (Low Growth, High Market Share)
- 4.3.3. Question Mark (High Growth, Low Market Share)
- 4.3.4. Dogs (Low Growth, Low Market Share)
- 4.4. Ansoff Matrix Analysis
- 4.5. Supply Chain Analysis
- 4.6. Regulatory Landscape
- 4.7. Current Market Potential and Opportunity Assessment (TAM–SAM–SOM Framework)
- 4.8. TIR Analyst Note
- 4.1. Porters Five Forces
- 5. Market Analysis, Insights and Forecast 2021-2033
- 5.1. Market Analysis, Insights and Forecast - by Type of Device
- 5.1.1. Spirometers
- 5.1.2. Peak Flow Meters
- 5.1.3. Sleep Test Devices
- 5.1.4. Gas Analyzers
- 5.1.5. Pulse Oximeters
- 5.1.6. Capnographs
- 5.1.7. Others
- 5.2. Market Analysis, Insights and Forecast - by Region
- 5.2.1. Germany
- 5.2.2. United Kingdom
- 5.2.3. France
- 5.2.4. Italy
- 5.2.5. Spain
- 5.2.6. Rest of Europe
- 5.1. Market Analysis, Insights and Forecast - by Type of Device
- 6. Europe Respiratory Monitoring Industry Analysis, Insights and Forecast, 2021-2033
- 6.1. Market Analysis, Insights and Forecast - by Type of Device
- 6.1.1. Spirometers
- 6.1.2. Peak Flow Meters
- 6.1.3. Sleep Test Devices
- 6.1.4. Gas Analyzers
- 6.1.5. Pulse Oximeters
- 6.1.6. Capnographs
- 6.1.7. Others
- 6.1. Market Analysis, Insights and Forecast - by Type of Device
- 7. Germany Europe Respiratory Monitoring Industry Analysis, Insights and Forecast, 2020-2032
- 7.1. Market Analysis, Insights and Forecast - by Type of Device
- 7.1.1. Spirometers
- 7.1.2. Peak Flow Meters
- 7.1.3. Sleep Test Devices
- 7.1.4. Gas Analyzers
- 7.1.5. Pulse Oximeters
- 7.1.6. Capnographs
- 7.1.7. Others
- 7.1. Market Analysis, Insights and Forecast - by Type of Device
- 8. United Kingdom Europe Respiratory Monitoring Industry Analysis, Insights and Forecast, 2020-2032
- 8.1. Market Analysis, Insights and Forecast - by Type of Device
- 8.1.1. Spirometers
- 8.1.2. Peak Flow Meters
- 8.1.3. Sleep Test Devices
- 8.1.4. Gas Analyzers
- 8.1.5. Pulse Oximeters
- 8.1.6. Capnographs
- 8.1.7. Others
- 8.1. Market Analysis, Insights and Forecast - by Type of Device
- 9. France Europe Respiratory Monitoring Industry Analysis, Insights and Forecast, 2020-2032
- 9.1. Market Analysis, Insights and Forecast - by Type of Device
- 9.1.1. Spirometers
- 9.1.2. Peak Flow Meters
- 9.1.3. Sleep Test Devices
- 9.1.4. Gas Analyzers
- 9.1.5. Pulse Oximeters
- 9.1.6. Capnographs
- 9.1.7. Others
- 9.1. Market Analysis, Insights and Forecast - by Type of Device
- 10. Italy Europe Respiratory Monitoring Industry Analysis, Insights and Forecast, 2020-2032
- 10.1. Market Analysis, Insights and Forecast - by Type of Device
- 10.1.1. Spirometers
- 10.1.2. Peak Flow Meters
- 10.1.3. Sleep Test Devices
- 10.1.4. Gas Analyzers
- 10.1.5. Pulse Oximeters
- 10.1.6. Capnographs
- 10.1.7. Others
- 10.1. Market Analysis, Insights and Forecast - by Type of Device
- 11. Spain Europe Respiratory Monitoring Industry Analysis, Insights and Forecast, 2020-2032
- 11.1. Market Analysis, Insights and Forecast - by Type of Device
- 11.1.1. Spirometers
- 11.1.2. Peak Flow Meters
- 11.1.3. Sleep Test Devices
- 11.1.4. Gas Analyzers
- 11.1.5. Pulse Oximeters
- 11.1.6. Capnographs
- 11.1.7. Others
- 11.1. Market Analysis, Insights and Forecast - by Type of Device
- 12. Rest of Europe Europe Respiratory Monitoring Industry Analysis, Insights and Forecast, 2020-2032
- 12.1. Market Analysis, Insights and Forecast - by Type of Device
- 12.1.1. Spirometers
- 12.1.2. Peak Flow Meters
- 12.1.3. Sleep Test Devices
- 12.1.4. Gas Analyzers
- 12.1.5. Pulse Oximeters
- 12.1.6. Capnographs
- 12.1.7. Others
- 12.1. Market Analysis, Insights and Forecast - by Type of Device
- 13. Competitive Analysis
- 13.1. Company Profiles
- 13.1.1 Maquet (Getinge AB)
- 13.1.1.1. Company Overview
- 13.1.1.2. Products
- 13.1.1.3. Company Financials
- 13.1.1.4. SWOT Analysis
- 13.1.2 Vitalograph Ltd
- 13.1.2.1. Company Overview
- 13.1.2.2. Products
- 13.1.2.3. Company Financials
- 13.1.2.4. SWOT Analysis
- 13.1.3 Medtronic
- 13.1.3.1. Company Overview
- 13.1.3.2. Products
- 13.1.3.3. Company Financials
- 13.1.3.4. SWOT Analysis
- 13.1.4 GE Healthcare
- 13.1.4.1. Company Overview
- 13.1.4.2. Products
- 13.1.4.3. Company Financials
- 13.1.4.4. SWOT Analysis
- 13.1.5 Koninklijke Philips N V
- 13.1.5.1. Company Overview
- 13.1.5.2. Products
- 13.1.5.3. Company Financials
- 13.1.5.4. SWOT Analysis
- 13.1.6 COSMED srl
- 13.1.6.1. Company Overview
- 13.1.6.2. Products
- 13.1.6.3. Company Financials
- 13.1.6.4. SWOT Analysis
- 13.1.7 Resmed
- 13.1.7.1. Company Overview
- 13.1.7.2. Products
- 13.1.7.3. Company Financials
- 13.1.7.4. SWOT Analysis
- 13.1.8 Siemens Healthcare GmbH
- 13.1.8.1. Company Overview
- 13.1.8.2. Products
- 13.1.8.3. Company Financials
- 13.1.8.4. SWOT Analysis
- 13.1.9 Koko PFT
- 13.1.9.1. Company Overview
- 13.1.9.2. Products
- 13.1.9.3. Company Financials
- 13.1.9.4. SWOT Analysis
- 13.1.10 Draegerwerk AG
- 13.1.10.1. Company Overview
- 13.1.10.2. Products
- 13.1.10.3. Company Financials
- 13.1.10.4. SWOT Analysis
- 13.1.1 Maquet (Getinge AB)
- 13.2. Market Entropy
- 13.2.1 Company's Key Areas Served
- 13.2.2 Recent Developments
- 13.3. Company Market Share Analysis 2025
- 13.3.1 Top 5 Companies Market Share Analysis
- 13.3.2 Top 3 Companies Market Share Analysis
- 13.4. List of Potential Customers
- 14. Research Methodology
List of Figures
- Figure 1: Europe Respiratory Monitoring Industry Revenue Breakdown (Million, %) by Product 2025 & 2033
- Figure 2: Europe Respiratory Monitoring Industry Share (%) by Company 2025
List of Tables
- Table 1: Europe Respiratory Monitoring Industry Revenue Million Forecast, by Type of Device 2020 & 2033
- Table 2: Europe Respiratory Monitoring Industry Revenue Million Forecast, by Region 2020 & 2033
- Table 3: Europe Respiratory Monitoring Industry Revenue Million Forecast, by Type of Device 2020 & 2033
- Table 4: Europe Respiratory Monitoring Industry Revenue Million Forecast, by Country 2020 & 2033
- Table 5: Europe Respiratory Monitoring Industry Revenue Million Forecast, by Type of Device 2020 & 2033
- Table 6: Europe Respiratory Monitoring Industry Revenue Million Forecast, by Country 2020 & 2033
- Table 7: Europe Respiratory Monitoring Industry Revenue Million Forecast, by Type of Device 2020 & 2033
- Table 8: Europe Respiratory Monitoring Industry Revenue Million Forecast, by Country 2020 & 2033
- Table 9: Europe Respiratory Monitoring Industry Revenue Million Forecast, by Type of Device 2020 & 2033
- Table 10: Europe Respiratory Monitoring Industry Revenue Million Forecast, by Country 2020 & 2033
- Table 11: Europe Respiratory Monitoring Industry Revenue Million Forecast, by Type of Device 2020 & 2033
- Table 12: Europe Respiratory Monitoring Industry Revenue Million Forecast, by Country 2020 & 2033
- Table 13: Europe Respiratory Monitoring Industry Revenue Million Forecast, by Type of Device 2020 & 2033
- Table 14: Europe Respiratory Monitoring Industry Revenue Million Forecast, by Country 2020 & 2033
Frequently Asked Questions
1. What is the projected Compound Annual Growth Rate (CAGR) of the Europe Respiratory Monitoring Industry?
The projected CAGR is approximately 8.60%.
2. Which companies are prominent players in the Europe Respiratory Monitoring Industry?
Key companies in the market include Maquet (Getinge AB), Vitalograph Ltd, Medtronic, GE Healthcare, Koninklijke Philips N V, COSMED srl, Resmed, Siemens Healthcare GmbH, Koko PFT, Draegerwerk AG.
3. What are the main segments of the Europe Respiratory Monitoring Industry?
The market segments include Type of Device.
4. Can you provide details about the market size?
The market size is estimated to be USD 1.16 Million as of 2022.
5. What are some drivers contributing to market growth?
Increasing Prevalence of Respiratory Diseases; Technological Advancements; Growing Geriatric Population.
6. What are the notable trends driving market growth?
Peak Flow Meters are Expected to Register Significant Growth over the Forecast Period.
7. Are there any restraints impacting market growth?
Reimbursement Concerns; High Cost Associated with the Devices.
8. Can you provide examples of recent developments in the market?
In March 2022, KoKo, LLC, a United Kingdom-based manufacturer of spirometers, signed a three-year partnership with MAGNET GROUP. This partnership would allow MAGNET GROUP to take advantage of the high-quality testing capabilities and features of KoKo's lung function diagnostics and software for spirometry and other devices.
9. What pricing options are available for accessing the report?
Pricing options include single-user, multi-user, and enterprise licenses priced at USD 4750, USD 5250, and USD 8750 respectively.
10. Is the market size provided in terms of value or volume?
The market size is provided in terms of value, measured in Million.
11. Are there any specific market keywords associated with the report?
Yes, the market keyword associated with the report is "Europe Respiratory Monitoring Industry," which aids in identifying and referencing the specific market segment covered.
12. How do I determine which pricing option suits my needs best?
The pricing options vary based on user requirements and access needs. Individual users may opt for single-user licenses, while businesses requiring broader access may choose multi-user or enterprise licenses for cost-effective access to the report.
13. Are there any additional resources or data provided in the Europe Respiratory Monitoring Industry report?
While the report offers comprehensive insights, it's advisable to review the specific contents or supplementary materials provided to ascertain if additional resources or data are available.
14. How can I stay updated on further developments or reports in the Europe Respiratory Monitoring Industry?
To stay informed about further developments, trends, and reports in the Europe Respiratory Monitoring Industry, consider subscribing to industry newsletters, following relevant companies and organizations, or regularly checking reputable industry news sources and publications.
Methodology
Step 1 - Identification of Relevant Samples Size from Population Database



Step 2 - Approaches for Defining Global Market Size (Value, Volume* & Price*)

Note*: In applicable scenarios
Step 3 - Data Sources
Primary Research
- Web Analytics
- Survey Reports
- Research Institute
- Latest Research Reports
- Opinion Leaders
Secondary Research
- Annual Reports
- White Paper
- Latest Press Release
- Industry Association
- Paid Database
- Investor Presentations

Step 4 - Data Triangulation
Involves using different sources of information in order to increase the validity of a study
These sources are likely to be stakeholders in a program - participants, other researchers, program staff, other community members, and so on.
Then we put all data in single framework & apply various statistical tools to find out the dynamic on the market.
During the analysis stage, feedback from the stakeholder groups would be compared to determine areas of agreement as well as areas of divergence


