Key Insights
The global flu vaccine market is poised for significant expansion, driven by increasing awareness of influenza's health implications and the continuous development of more effective vaccine technologies. The market is projected to reach $1.52 million by 2025 and grow at a Compound Annual Growth Rate (CAGR) of 6.95% through 2033. This robust growth is fueled by several key factors, including the rising prevalence of influenza strains necessitating annual vaccinations, government initiatives promoting widespread immunization, and advancements in drug delivery systems. Microneedle patch technology, encompassing both solid and hollow microneedle types, is emerging as a transformative segment, offering potentially less painful and more efficient administration compared to traditional injection methods. This innovation is expected to be a major catalyst for market penetration, particularly for trivalent and quadrivalent flu vaccines.
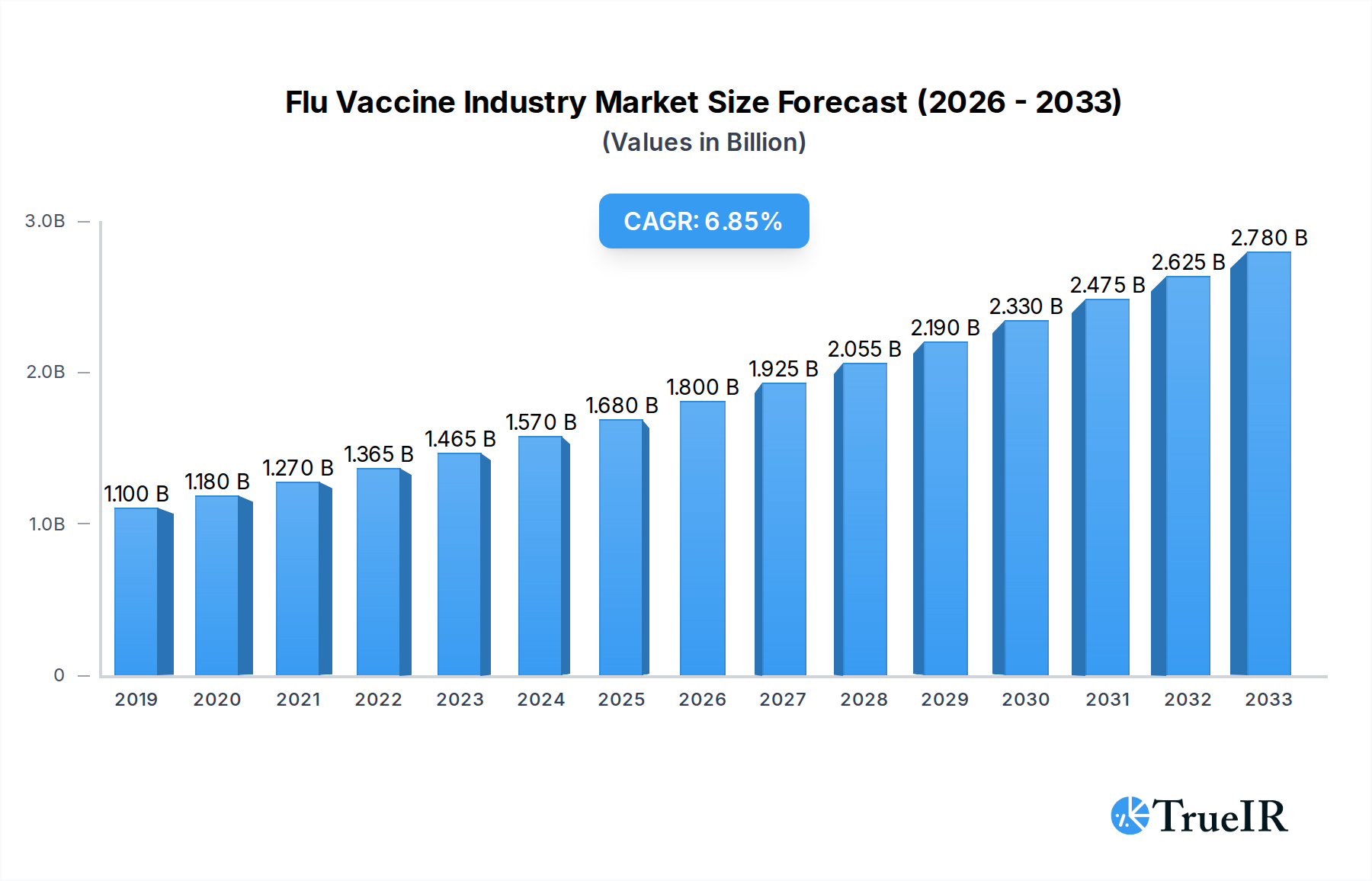
Flu Vaccine Industry Market Size (In Billion)

The competitive landscape is characterized by established pharmaceutical giants and innovative biotech firms vying for market share. Key players like Becton Dickinson and Company, CosMED Pharmaceuticals Co Ltd, and Vaxess Technologies are investing heavily in research and development to enhance vaccine efficacy and delivery mechanisms. Emerging trends include a focus on universal flu vaccines that offer broader and longer-lasting protection, as well as the increasing demand for flu vaccines in emerging economies within the Asia Pacific and Rest of the World regions. While restraints such as stringent regulatory approvals and the cost of developing novel technologies exist, the overarching trend points towards a dynamic and growing market, with North America and Europe currently leading in adoption, and Asia Pacific showing substantial growth potential due to its large population and increasing healthcare expenditure.
Flu Vaccine Industry Company Market Share

This comprehensive report provides an in-depth analysis of the global Flu Vaccine Industry, offering critical insights into market structure, competitive landscape, prevailing trends, dominant segments, and future outlook. Covering the study period from 2019 to 2033, with a base and estimated year of 2025, this report is an essential resource for stakeholders seeking to understand the evolving dynamics of this vital healthcare sector. The analysis leverages high-volume keywords such as "flu vaccine market," "influenza vaccine," "vaccination trends," "biotechnology," and "pharmaceutical industry" to enhance SEO visibility and reach a broad industry audience.
Flu Vaccine Industry Market Structure & Competitive Landscape
The Flu Vaccine Industry exhibits a moderately concentrated market structure, characterized by a blend of established pharmaceutical giants and innovative biotechnology firms. Key innovation drivers stem from advancements in vaccine development technologies, including novel delivery systems and improved antigen production methods. Regulatory impacts are significant, with stringent approval processes by bodies like the FDA and EMA shaping product lifecycles and market entry. Product substitutes exist, primarily in the form of other preventative health measures, though direct vaccine competition remains intense. End-user segmentation spans individual consumers, healthcare providers, government health organizations, and large employers. Mergers and acquisitions (M&A) are pivotal in consolidating market share and expanding R&D capabilities. In the historical period (2019-2024), an estimated XX M&A deals were completed, reflecting a market consolidation trend. Concentration ratios for the top five players are estimated at approximately XX%, indicating a significant, but not fully monopolized, market. Strategic partnerships are also crucial for R&D, manufacturing, and distribution.
Flu Vaccine Industry Market Trends & Opportunities
The global Flu Vaccine Industry is poised for substantial growth, projected to expand from an estimated XX million in the base year 2025 to XX million by the forecast year 2033, exhibiting a Compound Annual Growth Rate (CAGR) of approximately XX%. This growth is fueled by an increasing global focus on preventative healthcare, rising awareness of the severity of influenza, and government-backed vaccination programs aimed at reducing public health burdens. Technological shifts are a major trend, with the development of more effective and user-friendly vaccine delivery methods gaining traction. The success of mRNA vaccine technologies in other fields is also influencing R&D in influenza vaccines, promising faster development cycles and potentially broader protection. Consumer preferences are evolving towards convenience and efficacy, driving demand for novel administration routes and vaccines offering longer-lasting immunity. Competitive dynamics are intensifying, with companies investing heavily in R&D to secure market share through superior product offerings. The increasing prevalence of influenza strains and the need for annual vaccinations ensure a consistent and growing market demand. Opportunities lie in addressing unmet needs in specific demographics, such as the elderly and children, and in developing vaccines with broader spectrum protection against circulating strains. The rise of personalized medicine and the exploration of universal flu vaccines also represent significant future opportunities. Furthermore, emerging markets present a vast untapped potential for market penetration as healthcare infrastructure and public health initiatives expand.
Dominant Markets & Segments in Flu Vaccine Industry
The Quadrivalent Flu Vaccine segment is expected to dominate the Flu Vaccine Industry, driven by its broader protection against four influenza strains compared to its trivalent counterpart. In terms of Product Type, Solid Microneedle patches are emerging as a significant growth area, offering potential for improved patient compliance and administration without the need for refrigeration. The leading geographic region for flu vaccine consumption is anticipated to be North America, owing to high healthcare expenditure, well-established vaccination programs, and strong public awareness.
- Key Growth Drivers for Quadrivalent Flu Vaccines:
- Superior protection against a wider range of influenza strains.
- Increased recommendation by public health organizations globally.
- Growing demand from individuals seeking enhanced preventative measures.
- Key Growth Drivers for Solid Microneedle Delivery:
- Potential for self-administration, reducing healthcare provider burden.
- Improved vaccine stability and reduced cold chain requirements.
- Enhanced patient comfort and reduced needle-phobia.
- Key Growth Drivers for North America:
- Mandatory vaccination policies for healthcare workers and specific populations.
- High disposable incomes enabling greater healthcare spending.
- Robust research and development infrastructure for new vaccine technologies.
- Government initiatives to promote widespread vaccination coverage.
Detailed analysis of market dominance reveals that while established players hold significant sway in the traditional injectable vaccine market, the burgeoning microneedle segment presents an opportunity for innovative companies to capture market share through first-mover advantage and superior technological offerings. The increasing burden of influenza, coupled with the economic impact of outbreaks, continues to propel demand for effective vaccines across all segments.
Flu Vaccine Industry Product Analysis
Product innovations in the Flu Vaccine Industry are primarily focused on enhancing efficacy, improving patient convenience, and expanding the spectrum of protection. The development of solid microneedle patches, as exemplified by companies like Micron Biomedical, Inc. and Vaxess Technologies, Inc., represents a significant technological advancement, offering the potential for self-administration and improved vaccine stability. These innovations aim to overcome traditional barriers to vaccination, such as needle phobia and the need for cold chain logistics. Competitive advantages are being carved out through patented delivery systems, novel antigen formulations that elicit broader immune responses, and manufacturing processes that allow for rapid scale-up. The market fit for these advanced products lies in their ability to increase vaccination rates, particularly in underserved populations and in pandemic preparedness scenarios.
Key Drivers, Barriers & Challenges in Flu Vaccine Industry
Key Drivers: The Flu Vaccine Industry is propelled by several critical factors. Technological advancements in vaccine development, such as mRNA platforms and novel delivery systems like microneedles, are enabling the creation of more effective and convenient vaccines. Economic drivers include the significant healthcare costs associated with influenza-related illnesses, making preventative vaccination a cost-effective solution for governments and individuals. Policy-driven factors, such as government mandates and public health campaigns promoting vaccination, play a crucial role in driving demand and market penetration.
Key Barriers & Challenges: Supply chain disruptions, particularly for specialized raw materials and manufacturing capacity, can pose significant challenges, as can the complex and lengthy regulatory approval processes for new vaccines. Competitive pressures from established players and the emergence of new technologies require continuous innovation. Furthermore, vaccine hesitancy and misinformation can impact public acceptance and uptake, creating a barrier to achieving optimal vaccination rates. The annualized nature of flu vaccine development, requiring constant adaptation to evolving strains, presents a unique challenge.
Growth Drivers in the Flu Vaccine Industry Market
The Flu Vaccine Industry's growth is significantly propelled by an increasing global emphasis on public health and preventative care. Technological innovation is a primary catalyst, with breakthroughs in vaccine platforms like mRNA and advancements in delivery systems such as microneedle patches offering enhanced efficacy and user convenience. Economic factors, including the substantial healthcare expenditures related to influenza management, make vaccination a cost-effective strategy for governments and payers. Policy support, manifested through public health initiatives, vaccination mandates for specific groups, and funding for research and development, further fuels market expansion. The continuous evolution of influenza strains necessitates annual vaccine updates, ensuring a sustained demand cycle.
Challenges Impacting Flu Vaccine Industry Growth
The Flu Vaccine Industry faces several hurdles that can impede its growth trajectory. Regulatory complexities and the rigorous approval processes for new vaccines can lead to extended development timelines and increased costs. Supply chain vulnerabilities, including the availability of raw materials and the capacity for large-scale manufacturing, can create bottlenecks. Intense competition among established pharmaceutical companies and emerging biotechnology firms necessitates significant R&D investment and strategic market positioning. Vaccine hesitancy, fueled by misinformation and a lack of trust, remains a persistent challenge in achieving widespread vaccine uptake.
Key Players Shaping the Flu Vaccine Industry Market
- Becton Dickinson and Company
- CosMED Pharmaceuticals Co Ltd
- TSRL Inc
- Microdermics
- Vaxess Technologies
- NanoPass Technologies Limited
- Debiotech S A
- FluGen Inc
Significant Flu Vaccine Industry Industry Milestones
- November 2022: Micron Biomedical, Inc. secured USD 14 million in Series A financing, bolstering commercial manufacturing development and a strong partnership with LTS Lohmann. This funding supports Micron's pipeline of vaccine and drug products.
- April 2022: Vaxess Technologies, Inc. announced the production of the first GMP batch of MIMIX Technology Vaccine Patches, marking a significant step towards a Phase I seasonal influenza clinical trial for their innovative, shelf-stable vaccine delivery system.
Future Outlook for Flu Vaccine Industry Market
The future outlook for the Flu Vaccine Industry is exceptionally bright, driven by ongoing innovation and a growing global commitment to disease prevention. The market is expected to witness continued expansion fueled by the demand for more effective and convenient vaccination methods, particularly the adoption of microneedle technologies for improved accessibility and stability. Strategic partnerships between research institutions, pharmaceutical companies, and government agencies will accelerate the development of next-generation vaccines, including those offering broader protection or targeting novel influenza strains. Investment in pandemic preparedness infrastructure and the continuous evolution of influenza strains will ensure a sustained demand for flu vaccines, positioning the industry for robust and long-term growth.
Flu Vaccine Industry Segmentation
-
1. Product Type
- 1.1. Solid Microneedle
- 1.2. Hollow Microneedle
-
2. Vaccine Type
- 2.1. Trivalent Flu Vaccine
- 2.2. Quadrivalent Flu Vaccine
Flu Vaccine Industry Segmentation By Geography
-
1. North America
- 1.1. United States
- 1.2. Canada
- 1.3. Mexico
-
2. Europe
- 2.1. Germany
- 2.2. United Kingdom
- 2.3. France
- 2.4. Italy
- 2.5. Spain
- 2.6. Rest of Europe
-
3. Asia Pacific
- 3.1. China
- 3.2. Japan
- 3.3. India
- 3.4. Australia
- 3.5. South Korea
- 3.6. Rest of Asia Pacific
- 4. Rest of the World
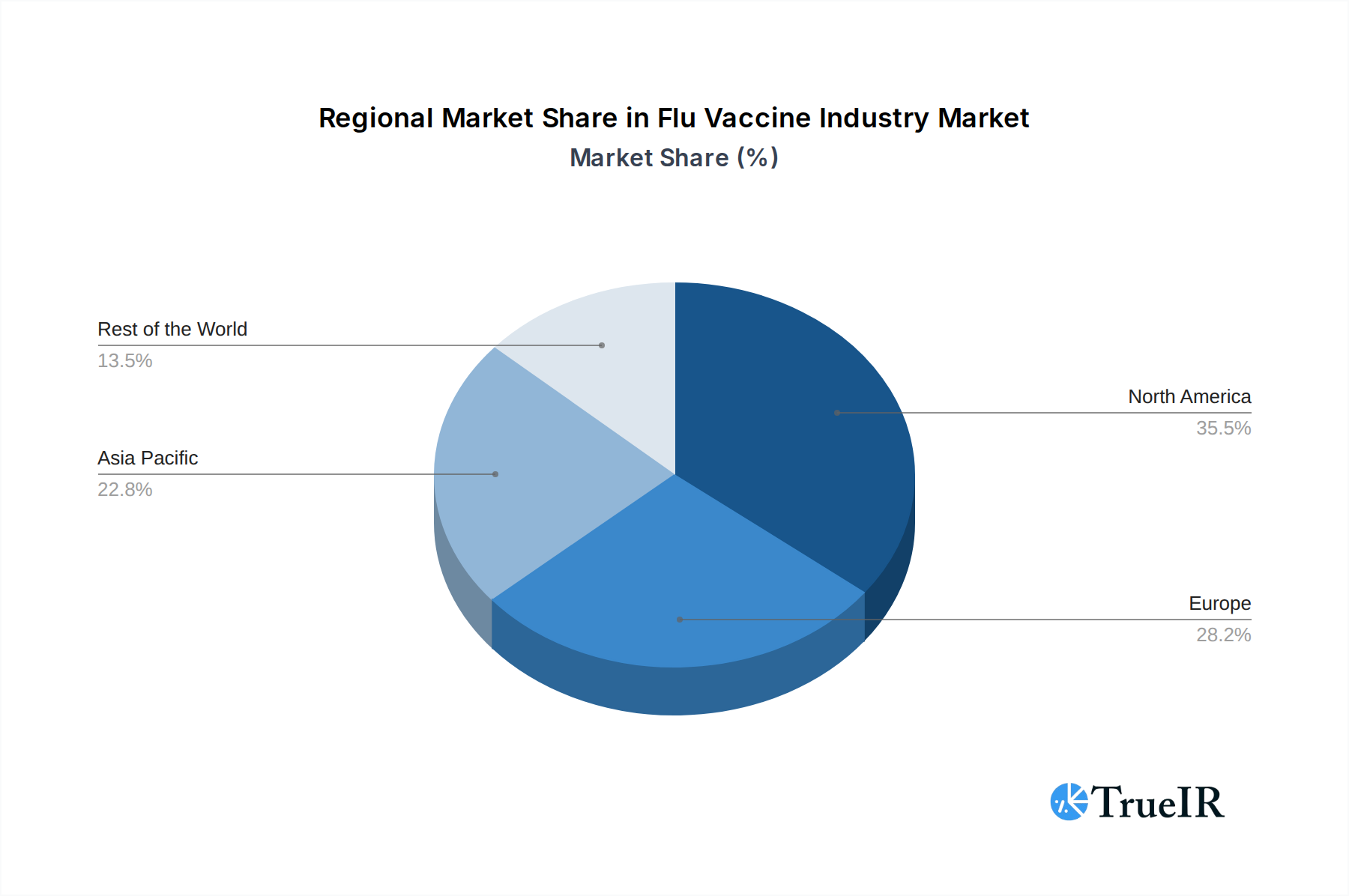
Flu Vaccine Industry Regional Market Share

Geographic Coverage of Flu Vaccine Industry
Flu Vaccine Industry REPORT HIGHLIGHTS
| Aspects | Details |
|---|---|
| Study Period | 2020-2034 |
| Base Year | 2025 |
| Estimated Year | 2026 |
| Forecast Period | 2026-2034 |
| Historical Period | 2020-2025 |
| Growth Rate | CAGR of 6.95% from 2020-2034 |
| Segmentation |
|
Table of Contents
- 1. Introduction
- 1.1. Research Scope
- 1.2. Market Segmentation
- 1.3. Research Objective
- 1.4. Definitions and Assumptions
- 2. Executive Summary
- 2.1. Market Snapshot
- 3. Market Dynamics
- 3.1. Market Drivers
- 3.2. Market Restrains
- 3.3. Market Trends
- 3.4. Market Opportunities
- 4. Market Factor Analysis
- 4.1. Porters Five Forces
- 4.1.1. Bargaining Power of Suppliers
- 4.1.2. Bargaining Power of Buyers
- 4.1.3. Threat of New Entrants
- 4.1.4. Threat of Substitutes
- 4.1.5. Competitive Rivalry
- 4.2. PESTEL analysis
- 4.3. BCG Analysis
- 4.3.1. Stars (High Growth, High Market Share)
- 4.3.2. Cash Cows (Low Growth, High Market Share)
- 4.3.3. Question Mark (High Growth, Low Market Share)
- 4.3.4. Dogs (Low Growth, Low Market Share)
- 4.4. Ansoff Matrix Analysis
- 4.5. Supply Chain Analysis
- 4.6. Regulatory Landscape
- 4.7. Current Market Potential and Opportunity Assessment (TAM–SAM–SOM Framework)
- 4.8. TIR Analyst Note
- 4.1. Porters Five Forces
- 5. Market Analysis, Insights and Forecast 2021-2033
- 5.1. Market Analysis, Insights and Forecast - by Product Type
- 5.1.1. Solid Microneedle
- 5.1.2. Hollow Microneedle
- 5.2. Market Analysis, Insights and Forecast - by Vaccine Type
- 5.2.1. Trivalent Flu Vaccine
- 5.2.2. Quadrivalent Flu Vaccine
- 5.3. Market Analysis, Insights and Forecast - by Region
- 5.3.1. North America
- 5.3.2. Europe
- 5.3.3. Asia Pacific
- 5.3.4. Rest of the World
- 5.1. Market Analysis, Insights and Forecast - by Product Type
- 6. Global Flu Vaccine Industry Analysis, Insights and Forecast, 2021-2033
- 6.1. Market Analysis, Insights and Forecast - by Product Type
- 6.1.1. Solid Microneedle
- 6.1.2. Hollow Microneedle
- 6.2. Market Analysis, Insights and Forecast - by Vaccine Type
- 6.2.1. Trivalent Flu Vaccine
- 6.2.2. Quadrivalent Flu Vaccine
- 6.1. Market Analysis, Insights and Forecast - by Product Type
- 7. North America Flu Vaccine Industry Analysis, Insights and Forecast, 2020-2032
- 7.1. Market Analysis, Insights and Forecast - by Product Type
- 7.1.1. Solid Microneedle
- 7.1.2. Hollow Microneedle
- 7.2. Market Analysis, Insights and Forecast - by Vaccine Type
- 7.2.1. Trivalent Flu Vaccine
- 7.2.2. Quadrivalent Flu Vaccine
- 7.1. Market Analysis, Insights and Forecast - by Product Type
- 8. Europe Flu Vaccine Industry Analysis, Insights and Forecast, 2020-2032
- 8.1. Market Analysis, Insights and Forecast - by Product Type
- 8.1.1. Solid Microneedle
- 8.1.2. Hollow Microneedle
- 8.2. Market Analysis, Insights and Forecast - by Vaccine Type
- 8.2.1. Trivalent Flu Vaccine
- 8.2.2. Quadrivalent Flu Vaccine
- 8.1. Market Analysis, Insights and Forecast - by Product Type
- 9. Asia Pacific Flu Vaccine Industry Analysis, Insights and Forecast, 2020-2032
- 9.1. Market Analysis, Insights and Forecast - by Product Type
- 9.1.1. Solid Microneedle
- 9.1.2. Hollow Microneedle
- 9.2. Market Analysis, Insights and Forecast - by Vaccine Type
- 9.2.1. Trivalent Flu Vaccine
- 9.2.2. Quadrivalent Flu Vaccine
- 9.1. Market Analysis, Insights and Forecast - by Product Type
- 10. Rest of the World Flu Vaccine Industry Analysis, Insights and Forecast, 2020-2032
- 10.1. Market Analysis, Insights and Forecast - by Product Type
- 10.1.1. Solid Microneedle
- 10.1.2. Hollow Microneedle
- 10.2. Market Analysis, Insights and Forecast - by Vaccine Type
- 10.2.1. Trivalent Flu Vaccine
- 10.2.2. Quadrivalent Flu Vaccine
- 10.1. Market Analysis, Insights and Forecast - by Product Type
- 11. Competitive Analysis
- 11.1. Company Profiles
- 11.1.1 Becton Dickinson and Company
- 11.1.1.1. Company Overview
- 11.1.1.2. Products
- 11.1.1.3. Company Financials
- 11.1.1.4. SWOT Analysis
- 11.1.2 CosMED Pharmaceuticals Co Ltd
- 11.1.2.1. Company Overview
- 11.1.2.2. Products
- 11.1.2.3. Company Financials
- 11.1.2.4. SWOT Analysis
- 11.1.3 TSRL Inc
- 11.1.3.1. Company Overview
- 11.1.3.2. Products
- 11.1.3.3. Company Financials
- 11.1.3.4. SWOT Analysis
- 11.1.4 Microdermics
- 11.1.4.1. Company Overview
- 11.1.4.2. Products
- 11.1.4.3. Company Financials
- 11.1.4.4. SWOT Analysis
- 11.1.5 Vaxess Technologies
- 11.1.5.1. Company Overview
- 11.1.5.2. Products
- 11.1.5.3. Company Financials
- 11.1.5.4. SWOT Analysis
- 11.1.6 NanoPass Technologies Limited
- 11.1.6.1. Company Overview
- 11.1.6.2. Products
- 11.1.6.3. Company Financials
- 11.1.6.4. SWOT Analysis
- 11.1.7 Debiotech S A
- 11.1.7.1. Company Overview
- 11.1.7.2. Products
- 11.1.7.3. Company Financials
- 11.1.7.4. SWOT Analysis
- 11.1.8 FluGen Inc
- 11.1.8.1. Company Overview
- 11.1.8.2. Products
- 11.1.8.3. Company Financials
- 11.1.8.4. SWOT Analysis
- 11.1.1 Becton Dickinson and Company
- 11.2. Market Entropy
- 11.2.1 Company's Key Areas Served
- 11.2.2 Recent Developments
- 11.3. Company Market Share Analysis 2025
- 11.3.1 Top 5 Companies Market Share Analysis
- 11.3.2 Top 3 Companies Market Share Analysis
- 11.4. List of Potential Customers
- 12. Research Methodology
List of Figures
- Figure 1: Global Flu Vaccine Industry Revenue Breakdown (Million, %) by Region 2025 & 2033
- Figure 2: North America Flu Vaccine Industry Revenue (Million), by Product Type 2025 & 2033
- Figure 3: North America Flu Vaccine Industry Revenue Share (%), by Product Type 2025 & 2033
- Figure 4: North America Flu Vaccine Industry Revenue (Million), by Vaccine Type 2025 & 2033
- Figure 5: North America Flu Vaccine Industry Revenue Share (%), by Vaccine Type 2025 & 2033
- Figure 6: North America Flu Vaccine Industry Revenue (Million), by Country 2025 & 2033
- Figure 7: North America Flu Vaccine Industry Revenue Share (%), by Country 2025 & 2033
- Figure 8: Europe Flu Vaccine Industry Revenue (Million), by Product Type 2025 & 2033
- Figure 9: Europe Flu Vaccine Industry Revenue Share (%), by Product Type 2025 & 2033
- Figure 10: Europe Flu Vaccine Industry Revenue (Million), by Vaccine Type 2025 & 2033
- Figure 11: Europe Flu Vaccine Industry Revenue Share (%), by Vaccine Type 2025 & 2033
- Figure 12: Europe Flu Vaccine Industry Revenue (Million), by Country 2025 & 2033
- Figure 13: Europe Flu Vaccine Industry Revenue Share (%), by Country 2025 & 2033
- Figure 14: Asia Pacific Flu Vaccine Industry Revenue (Million), by Product Type 2025 & 2033
- Figure 15: Asia Pacific Flu Vaccine Industry Revenue Share (%), by Product Type 2025 & 2033
- Figure 16: Asia Pacific Flu Vaccine Industry Revenue (Million), by Vaccine Type 2025 & 2033
- Figure 17: Asia Pacific Flu Vaccine Industry Revenue Share (%), by Vaccine Type 2025 & 2033
- Figure 18: Asia Pacific Flu Vaccine Industry Revenue (Million), by Country 2025 & 2033
- Figure 19: Asia Pacific Flu Vaccine Industry Revenue Share (%), by Country 2025 & 2033
- Figure 20: Rest of the World Flu Vaccine Industry Revenue (Million), by Product Type 2025 & 2033
- Figure 21: Rest of the World Flu Vaccine Industry Revenue Share (%), by Product Type 2025 & 2033
- Figure 22: Rest of the World Flu Vaccine Industry Revenue (Million), by Vaccine Type 2025 & 2033
- Figure 23: Rest of the World Flu Vaccine Industry Revenue Share (%), by Vaccine Type 2025 & 2033
- Figure 24: Rest of the World Flu Vaccine Industry Revenue (Million), by Country 2025 & 2033
- Figure 25: Rest of the World Flu Vaccine Industry Revenue Share (%), by Country 2025 & 2033
List of Tables
- Table 1: Global Flu Vaccine Industry Revenue Million Forecast, by Product Type 2020 & 2033
- Table 2: Global Flu Vaccine Industry Revenue Million Forecast, by Vaccine Type 2020 & 2033
- Table 3: Global Flu Vaccine Industry Revenue Million Forecast, by Region 2020 & 2033
- Table 4: Global Flu Vaccine Industry Revenue Million Forecast, by Product Type 2020 & 2033
- Table 5: Global Flu Vaccine Industry Revenue Million Forecast, by Vaccine Type 2020 & 2033
- Table 6: Global Flu Vaccine Industry Revenue Million Forecast, by Country 2020 & 2033
- Table 7: United States Flu Vaccine Industry Revenue (Million) Forecast, by Application 2020 & 2033
- Table 8: Canada Flu Vaccine Industry Revenue (Million) Forecast, by Application 2020 & 2033
- Table 9: Mexico Flu Vaccine Industry Revenue (Million) Forecast, by Application 2020 & 2033
- Table 10: Global Flu Vaccine Industry Revenue Million Forecast, by Product Type 2020 & 2033
- Table 11: Global Flu Vaccine Industry Revenue Million Forecast, by Vaccine Type 2020 & 2033
- Table 12: Global Flu Vaccine Industry Revenue Million Forecast, by Country 2020 & 2033
- Table 13: Germany Flu Vaccine Industry Revenue (Million) Forecast, by Application 2020 & 2033
- Table 14: United Kingdom Flu Vaccine Industry Revenue (Million) Forecast, by Application 2020 & 2033
- Table 15: France Flu Vaccine Industry Revenue (Million) Forecast, by Application 2020 & 2033
- Table 16: Italy Flu Vaccine Industry Revenue (Million) Forecast, by Application 2020 & 2033
- Table 17: Spain Flu Vaccine Industry Revenue (Million) Forecast, by Application 2020 & 2033
- Table 18: Rest of Europe Flu Vaccine Industry Revenue (Million) Forecast, by Application 2020 & 2033
- Table 19: Global Flu Vaccine Industry Revenue Million Forecast, by Product Type 2020 & 2033
- Table 20: Global Flu Vaccine Industry Revenue Million Forecast, by Vaccine Type 2020 & 2033
- Table 21: Global Flu Vaccine Industry Revenue Million Forecast, by Country 2020 & 2033
- Table 22: China Flu Vaccine Industry Revenue (Million) Forecast, by Application 2020 & 2033
- Table 23: Japan Flu Vaccine Industry Revenue (Million) Forecast, by Application 2020 & 2033
- Table 24: India Flu Vaccine Industry Revenue (Million) Forecast, by Application 2020 & 2033
- Table 25: Australia Flu Vaccine Industry Revenue (Million) Forecast, by Application 2020 & 2033
- Table 26: South Korea Flu Vaccine Industry Revenue (Million) Forecast, by Application 2020 & 2033
- Table 27: Rest of Asia Pacific Flu Vaccine Industry Revenue (Million) Forecast, by Application 2020 & 2033
- Table 28: Global Flu Vaccine Industry Revenue Million Forecast, by Product Type 2020 & 2033
- Table 29: Global Flu Vaccine Industry Revenue Million Forecast, by Vaccine Type 2020 & 2033
- Table 30: Global Flu Vaccine Industry Revenue Million Forecast, by Country 2020 & 2033
Frequently Asked Questions
1. What is the projected Compound Annual Growth Rate (CAGR) of the Flu Vaccine Industry?
The projected CAGR is approximately 6.95%.
2. Which companies are prominent players in the Flu Vaccine Industry?
Key companies in the market include Becton Dickinson and Company, CosMED Pharmaceuticals Co Ltd, TSRL Inc, Microdermics, Vaxess Technologies, NanoPass Technologies Limited, Debiotech S A, FluGen Inc.
3. What are the main segments of the Flu Vaccine Industry?
The market segments include Product Type, Vaccine Type.
4. Can you provide details about the market size?
The market size is estimated to be USD 1.52 Million as of 2022.
5. What are some drivers contributing to market growth?
Increasing Prevalence of Influenza and Viral Infections; Key Players Focusing on Research and Development of Micro-Needle Flu Vaccines.
6. What are the notable trends driving market growth?
Quadrivalent Flu Vaccine Segment is Expected to Projected Significant Growth Over the Forecast Period.
7. Are there any restraints impacting market growth?
Complications and Risks Associated with Microneedles.
8. Can you provide examples of recent developments in the market?
In November 2022, Micron Biomedical, Inc. secured USD 14 million in Series A financing to support the company's commercial manufacturing development and establish a strong partnership between Micron and LTS Lohmann. Micron's development pipeline includes vaccine and drug products partnered with pharma companies, foundations, and government agencies.
9. What pricing options are available for accessing the report?
Pricing options include single-user, multi-user, and enterprise licenses priced at USD 4750, USD 5250, and USD 8750 respectively.
10. Is the market size provided in terms of value or volume?
The market size is provided in terms of value, measured in Million.
11. Are there any specific market keywords associated with the report?
Yes, the market keyword associated with the report is "Flu Vaccine Industry," which aids in identifying and referencing the specific market segment covered.
12. How do I determine which pricing option suits my needs best?
The pricing options vary based on user requirements and access needs. Individual users may opt for single-user licenses, while businesses requiring broader access may choose multi-user or enterprise licenses for cost-effective access to the report.
13. Are there any additional resources or data provided in the Flu Vaccine Industry report?
While the report offers comprehensive insights, it's advisable to review the specific contents or supplementary materials provided to ascertain if additional resources or data are available.
14. How can I stay updated on further developments or reports in the Flu Vaccine Industry?
To stay informed about further developments, trends, and reports in the Flu Vaccine Industry, consider subscribing to industry newsletters, following relevant companies and organizations, or regularly checking reputable industry news sources and publications.
Methodology
Step 1 - Identification of Relevant Samples Size from Population Database



Step 2 - Approaches for Defining Global Market Size (Value, Volume* & Price*)

Note*: In applicable scenarios
Step 3 - Data Sources
Primary Research
- Web Analytics
- Survey Reports
- Research Institute
- Latest Research Reports
- Opinion Leaders
Secondary Research
- Annual Reports
- White Paper
- Latest Press Release
- Industry Association
- Paid Database
- Investor Presentations

Step 4 - Data Triangulation
Involves using different sources of information in order to increase the validity of a study
These sources are likely to be stakeholders in a program - participants, other researchers, program staff, other community members, and so on.
Then we put all data in single framework & apply various statistical tools to find out the dynamic on the market.
During the analysis stage, feedback from the stakeholder groups would be compared to determine areas of agreement as well as areas of divergence


