Key Insights
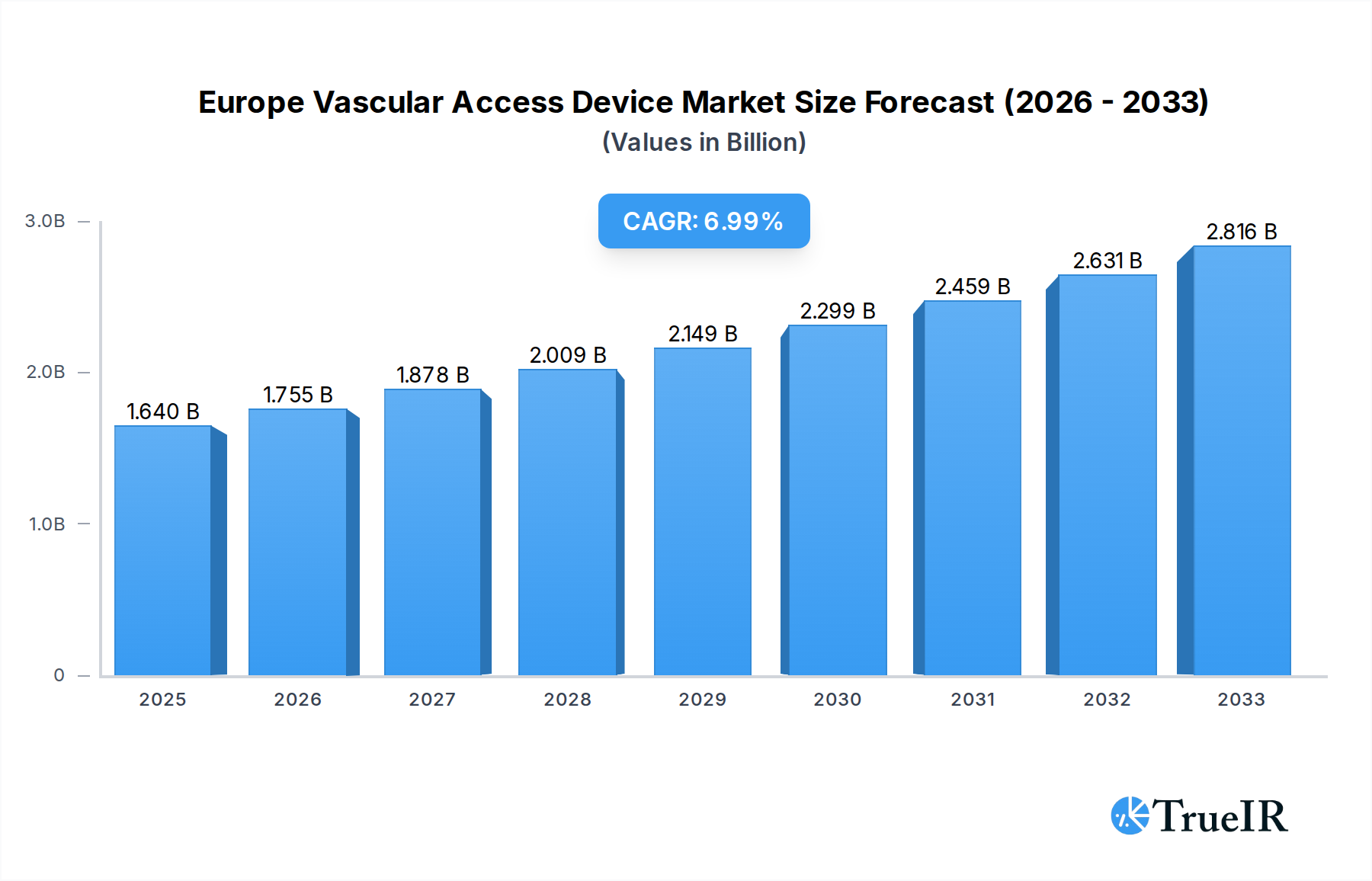
The European vascular access device market is poised for substantial growth, projected to reach $1.64 billion in 2025 with a robust CAGR of 6.91% through 2033. This expansion is driven by an increasing prevalence of chronic diseases, a growing aging population, and advancements in medical technology, all contributing to a higher demand for effective vascular access solutions. The market’s dynamics are shaped by a rising need for both central and peripheral vascular access devices, catering to diverse clinical applications such as drug administration, fluid and nutrition delivery, and diagnostic procedures. Hospitals and clinics represent the dominant end-user segment, reflecting the critical role these devices play in inpatient care and medical interventions. Continuous innovation in catheter materials, design, and safety features, alongside a focus on reducing complications like infections and thrombotic events, are key trends fueling market expansion.
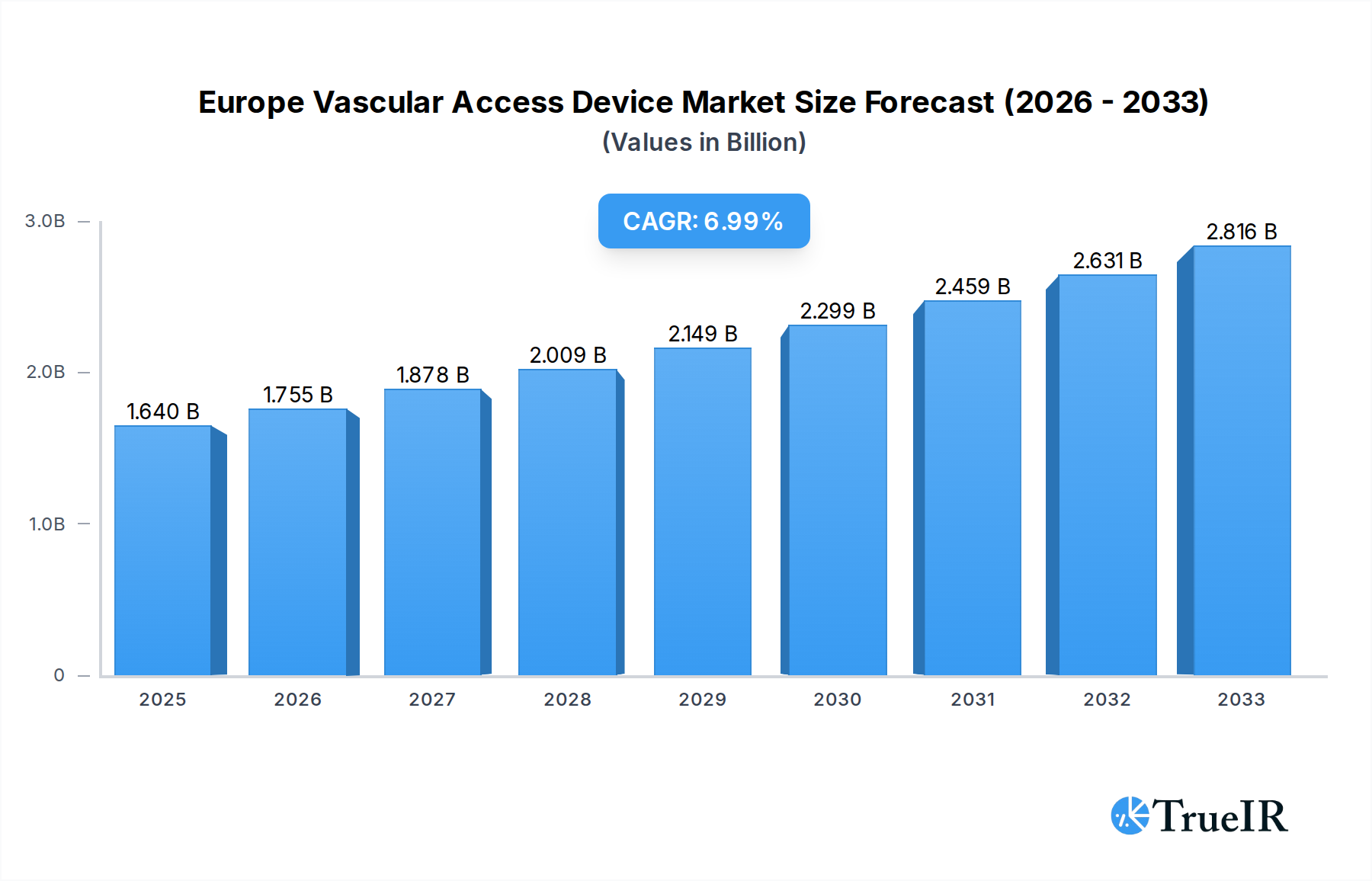
Europe Vascular Access Device Market Market Size (In Billion)

The market's growth is further bolstered by strategic initiatives from leading players and an increasing emphasis on minimally invasive procedures. However, challenges such as stringent regulatory frameworks and the risk of healthcare-associated infections necessitate a continued focus on product safety and efficacy. The competitive landscape is characterized by the presence of established global manufacturers like Becton Dickinson and Company, Medtronic, and 3M, who are actively engaged in research and development to introduce novel products and expand their market reach. The projected expansion underscores the indispensable nature of vascular access devices in modern healthcare delivery across Europe, making it a dynamic and evolving sector within the medical devices industry.
Europe Vascular Access Device Market Company Market Share

This in-depth report offers a detailed examination of the Europe vascular access device market, providing critical insights for stakeholders navigating this dynamic sector. Spanning the historical period of 2019-2024, the base year of 2025, and a comprehensive forecast period of 2025-2033, this study delves into market structure, key trends, dominant segments, product innovations, growth drivers, challenges, and the competitive landscape. Leveraging high-volume keywords such as "vascular access devices Europe," "central venous catheters market," "peripheral IV catheters," "drug administration devices," and "hospital medical devices," this report is optimized for search engines and designed to engage industry professionals, researchers, and investors. The market is projected to reach a valuation exceeding $5.0 billion by 2033, exhibiting a Compound Annual Growth Rate (CAGR) of approximately 6.8% during the forecast period.
Europe Vascular Access Device Market Market Structure & Competitive Landscape
The Europe vascular access device market is characterized by a moderate to high level of concentration, with key players like Becton Dickinson and Company, Nipro Medical Corporation, ICU Medical Inc., Edwards Lifesciences Corporation, Medtronic, Fresenius Medical Care AG & Co KGaA, 3M, AngioDynamics, Terumo Medical Corporation, Teleflex Incorporated, and B Braun SE holding significant market shares. Innovation is a key driver, fueled by the constant demand for safer, more effective, and patient-friendly vascular access solutions, particularly in the realm of infection prevention and advanced catheter technologies. Regulatory frameworks, while ensuring patient safety, also present a substantial barrier to entry and influence product development cycles. Product substitutes, such as alternative drug delivery methods, exist but are often limited by specific clinical indications. End-user segmentation within hospitals and clinics remains dominant, with increasing penetration in diagnostic centers and other specialized healthcare settings. Mergers and acquisitions (M&A) play a crucial role in market consolidation and expansion, with an estimated 15-20 M&A activities recorded during the historical period, aimed at broadening product portfolios and geographic reach. The market exhibits a trend towards specialized devices catering to critical care, oncology, and long-term therapy needs.
Europe Vascular Access Device Market Market Trends & Opportunities
The European vascular access device market is on a robust growth trajectory, projected to expand significantly in value from approximately $3.5 billion in 2025 to over $5.0 billion by 2033, reflecting a steady CAGR of around 6.8%. This expansion is underpinned by a confluence of technological advancements, evolving patient demographics, and increasing healthcare expenditures across the continent. A prominent trend is the rising demand for antimicrobial-coated catheters and advanced technologies aimed at reducing catheter-related bloodstream infections (CRBSIs), a persistent challenge in healthcare settings. This aligns with growing patient preferences for minimally invasive procedures and improved comfort, driving the adoption of sophisticated PICC lines and midline catheters. The increasing prevalence of chronic diseases, such as cardiovascular conditions, cancer, and diabetes, necessitates long-term and frequent vascular access for drug administration, fluid management, and nutritional support, thereby fueling market growth. Furthermore, advancements in materials science are leading to the development of more biocompatible and durable vascular access devices, enhancing patient outcomes and reducing complications. The integration of smart technologies and connectivity in vascular access devices, although in its nascent stages, presents a significant future opportunity, enabling remote patient monitoring and improved data management for healthcare providers. The shift towards home healthcare and ambulatory surgery centers also creates new avenues for the adoption of peripheral vascular access devices. Competitive dynamics are intensifying, with established players investing heavily in R&D to introduce innovative products and secure market dominance. Opportunities abound for companies focusing on specialized applications, such as oncology and critical care, and those capable of navigating complex regulatory landscapes while offering cost-effective solutions.
Dominant Markets & Segments in Europe Vascular Access Device Market
The Central Vascular Access Devices segment is poised to be the largest contributor to the Europe vascular access device market revenue, estimated to account for over 60% of the total market value in 2025, projected to reach approximately $2.1 billion. Within this segment, Peripherally Inserted Central Catheters (PICCs) are experiencing substantial growth due to their ease of insertion, reduced risk of complications compared to traditional central lines, and suitability for long-term therapies. The demand for PICCs is driven by the increasing incidence of cancer and the associated chemotherapy regimens, as well as the growing need for parenteral nutrition.
Peripheral Vascular Access Devices are also witnessing significant expansion, driven by the high volume of routine procedures and the increasing preference for minimally invasive interventions. Peripheral Catheters, used for short-term venous access, will continue to dominate this sub-segment.
By Application, the Administration of Drugs segment is the largest and fastest-growing, expected to capture over 45% of the market share in 2025, valued at around $1.6 billion. This growth is propelled by the escalating use of intravenous medications for chronic disease management, pain management, and critical care interventions. The Administration of Fluid and Nutrition segment also represents a substantial market, particularly in hospital settings catering to patients with malnourishment or those requiring extensive hydration.
Geographically, Germany and the United Kingdom are expected to remain the dominant markets within Europe, collectively holding over 40% of the market share in 2025. This dominance is attributed to their well-established healthcare infrastructures, high healthcare spending, advanced medical technologies, and favorable reimbursement policies. Strong growth is also anticipated in France and Italy, driven by increasing healthcare investments and a growing patient pool requiring vascular access.
The Hospital/Clinic end-user segment will continue to be the primary revenue generator, accounting for an estimated 75% of the market in 2025. This is due to the concentration of complex procedures and inpatient care within these settings. However, the Diagnostic Centers segment is expected to exhibit a higher CAGR during the forecast period, driven by the increasing outpatient diagnostic procedures and specialized testing requiring intravenous access.
Europe Vascular Access Device Market Product Analysis
Product innovation in the Europe vascular access device market is primarily focused on enhancing patient safety and improving clinical outcomes. Key advancements include the development of novel antimicrobial coatings and materials that significantly reduce the risk of catheter-related infections. Technologies such as integrated needle protection systems and improved catheter tip designs are minimizing accidental needlesticks and tissue trauma. Furthermore, the market is witnessing the emergence of smart catheters with embedded sensors for real-time monitoring of flow rates and pressure, offering enhanced therapeutic precision. These innovations are driven by a growing demand for minimally invasive, efficient, and patient-centric vascular access solutions across various clinical applications, from short-term peripheral access to long-term central venous access.
Key Drivers, Barriers & Challenges in Europe Vascular Access Device Market
Key Drivers: The Europe vascular access device market is propelled by a surge in the prevalence of chronic diseases like cancer and cardiovascular ailments, necessitating frequent and long-term intravenous therapies. Technological advancements, including the development of antimicrobial coatings and sophisticated catheter designs, are crucial in reducing infection rates and improving patient safety. Growing healthcare expenditure across Europe, coupled with an aging population, further fuels demand. Government initiatives promoting early diagnosis and treatment of diseases also contribute to market expansion.
Barriers & Challenges: Stringent regulatory approval processes by agencies like the EMA can be time-consuming and costly, acting as a significant barrier to market entry for new products. Issues related to supply chain disruptions, as observed in recent global events, can impact product availability and lead to increased costs. Intense price competition among market players, especially for standard peripheral catheters, puts pressure on profit margins. Additionally, the risk of healthcare-associated infections (HAIs), despite technological advancements, remains a persistent challenge requiring continuous vigilance and adherence to best practices. The increasing adoption of alternative drug delivery methods for certain therapies can also pose a restraint.
Growth Drivers in the Europe Vascular Access Device Market Market
The Europe vascular access device market is experiencing robust growth fueled by several key factors. The escalating incidence of chronic diseases, including cancer, diabetes, and cardiovascular disorders, directly translates to a higher demand for vascular access for drug administration, chemotherapy, and nutritional support. Technological innovations are paramount, with the development of advanced antimicrobial-coated catheters and improved designs that significantly reduce the risk of catheter-related bloodstream infections (CRBSIs). This focus on patient safety is a major growth catalyst. Furthermore, an aging European population, coupled with increasing healthcare expenditure and a growing emphasis on minimally invasive procedures, are key economic and demographic drivers. Favorable reimbursement policies in many European countries also support the adoption of sophisticated vascular access devices.
Challenges Impacting Europe Vascular Access Device Market Growth
Several challenges continue to impact the growth of the Europe vascular access device market. The rigorous and time-consuming regulatory approval process imposed by European health authorities can be a significant hurdle for manufacturers seeking to introduce new and innovative products. Supply chain volatility, highlighted by recent global events, poses a continuous risk of material shortages and increased production costs, affecting product availability and affordability. Intense competition among established players and emerging manufacturers leads to price pressures, particularly for more commoditized products. Moreover, the ongoing threat of healthcare-associated infections (HAIs), despite advancements in device technology, necessitates continuous vigilance and adherence to strict infection control protocols, adding to healthcare system complexities and costs.
Key Players Shaping the Europe Vascular Access Device Market Market
- Becton Dickinson and Company
- Nipro Medical Corporation
- ICU Medical Inc.
- Edwards Lifesciences Corporation
- Medtronic
- Fresenius Medical Care AG & Co KGaA
- 3M
- AngioDynamics
- Terumo Medical Corporation
- Teleflex Incorporated
- B Braun SE
- Prodimed
Significant Europe Vascular Access Device Market Industry Milestones
- June 2023: BIOTRONIK launched its Oscar (One Solution: Cross. Adjust. Restore) multifunctional peripheral catheter, initiating promotional activities at the LINC (Leipzig Interventional Course) in Germany. This device offers enhanced access and dilation capabilities for stenoses in femoral, popliteal, and infrapopliteal arteries, significantly impacting peripheral interventional procedures.
- September 2022: Delta Med Group, an Italy-based specialist in vascular access devices for oncology and analgesic therapies, formed a strategic partnership with Pentaferte. This collaboration aims to strengthen their competitive position in the European infusion medical device market, focusing on expanding product offerings and market reach.
Future Outlook for Europe Vascular Access Device Market Market
The future outlook for the Europe vascular access device market is exceptionally promising, driven by sustained demand for advanced and safe healthcare solutions. Key growth catalysts include the ongoing rise in chronic disease prevalence, a continuously aging population, and significant advancements in medical technology leading to more sophisticated and patient-centric devices. Opportunities lie in the development and adoption of smart vascular access devices with integrated monitoring capabilities, and further innovation in antimicrobial technologies to combat healthcare-associated infections. The increasing shift towards home healthcare and outpatient settings will also create new market segments for specialized and user-friendly devices. Strategic collaborations, targeted product development for niche applications like oncology and critical care, and a keen understanding of evolving regulatory landscapes will be crucial for sustained success in this expanding market.
Europe Vascular Access Device Market Segmentation
-
1. Device Type
-
1.1. Central Vascular Access Devices
- 1.1.1. Peripherally Inserted Central Catheters
- 1.1.2. Percutaneous Non-tunneled Catheters
- 1.1.3. Other Central Vascular Access Devices
-
1.2. Peripheral Vascular Access Devices
- 1.2.1. Peripheral Catheter
- 1.2.2. Midline Catheter
- 1.2.3. Other Peripheral Vascular Access Devices
-
1.1. Central Vascular Access Devices
-
2. Application
- 2.1. Administration of Drugs
- 2.2. Administration of Fluid and Nutrition
- 2.3. Diagnostics and Testing
- 2.4. Other Applications
-
3. End-User
- 3.1. Hospital/Clinic
- 3.2. Diagnostic Centers
- 3.3. Other End-Users
Europe Vascular Access Device Market Segmentation By Geography
- 1. Germany
- 2. United Kingdom
- 3. France
- 4. Italy
- 5. Spain
- 6. Rest of Europe

Europe Vascular Access Device Market Regional Market Share

Geographic Coverage of Europe Vascular Access Device Market
Europe Vascular Access Device Market REPORT HIGHLIGHTS
| Aspects | Details |
|---|---|
| Study Period | 2020-2034 |
| Base Year | 2025 |
| Estimated Year | 2026 |
| Forecast Period | 2026-2034 |
| Historical Period | 2020-2025 |
| Growth Rate | CAGR of 6.91% from 2020-2034 |
| Segmentation |
|
Table of Contents
- 1. Introduction
- 1.1. Research Scope
- 1.2. Market Segmentation
- 1.3. Research Objective
- 1.4. Definitions and Assumptions
- 2. Executive Summary
- 2.1. Market Snapshot
- 3. Market Dynamics
- 3.1. Market Drivers
- 3.2. Market Restrains
- 3.3. Market Trends
- 3.4. Market Opportunities
- 4. Market Factor Analysis
- 4.1. Porters Five Forces
- 4.1.1. Bargaining Power of Suppliers
- 4.1.2. Bargaining Power of Buyers
- 4.1.3. Threat of New Entrants
- 4.1.4. Threat of Substitutes
- 4.1.5. Competitive Rivalry
- 4.2. PESTEL analysis
- 4.3. BCG Analysis
- 4.3.1. Stars (High Growth, High Market Share)
- 4.3.2. Cash Cows (Low Growth, High Market Share)
- 4.3.3. Question Mark (High Growth, Low Market Share)
- 4.3.4. Dogs (Low Growth, Low Market Share)
- 4.4. Ansoff Matrix Analysis
- 4.5. Supply Chain Analysis
- 4.6. Regulatory Landscape
- 4.7. Current Market Potential and Opportunity Assessment (TAM–SAM–SOM Framework)
- 4.8. TIR Analyst Note
- 4.1. Porters Five Forces
- 5. Market Analysis, Insights and Forecast 2021-2033
- 5.1. Market Analysis, Insights and Forecast - by Device Type
- 5.1.1. Central Vascular Access Devices
- 5.1.1.1. Peripherally Inserted Central Catheters
- 5.1.1.2. Percutaneous Non-tunneled Catheters
- 5.1.1.3. Other Central Vascular Access Devices
- 5.1.2. Peripheral Vascular Access Devices
- 5.1.2.1. Peripheral Catheter
- 5.1.2.2. Midline Catheter
- 5.1.2.3. Other Peripheral Vascular Access Devices
- 5.1.1. Central Vascular Access Devices
- 5.2. Market Analysis, Insights and Forecast - by Application
- 5.2.1. Administration of Drugs
- 5.2.2. Administration of Fluid and Nutrition
- 5.2.3. Diagnostics and Testing
- 5.2.4. Other Applications
- 5.3. Market Analysis, Insights and Forecast - by End-User
- 5.3.1. Hospital/Clinic
- 5.3.2. Diagnostic Centers
- 5.3.3. Other End-Users
- 5.4. Market Analysis, Insights and Forecast - by Region
- 5.4.1. Germany
- 5.4.2. United Kingdom
- 5.4.3. France
- 5.4.4. Italy
- 5.4.5. Spain
- 5.4.6. Rest of Europe
- 5.1. Market Analysis, Insights and Forecast - by Device Type
- 6. Europe Vascular Access Device Market Analysis, Insights and Forecast, 2021-2033
- 6.1. Market Analysis, Insights and Forecast - by Device Type
- 6.1.1. Central Vascular Access Devices
- 6.1.1.1. Peripherally Inserted Central Catheters
- 6.1.1.2. Percutaneous Non-tunneled Catheters
- 6.1.1.3. Other Central Vascular Access Devices
- 6.1.2. Peripheral Vascular Access Devices
- 6.1.2.1. Peripheral Catheter
- 6.1.2.2. Midline Catheter
- 6.1.2.3. Other Peripheral Vascular Access Devices
- 6.1.1. Central Vascular Access Devices
- 6.2. Market Analysis, Insights and Forecast - by Application
- 6.2.1. Administration of Drugs
- 6.2.2. Administration of Fluid and Nutrition
- 6.2.3. Diagnostics and Testing
- 6.2.4. Other Applications
- 6.3. Market Analysis, Insights and Forecast - by End-User
- 6.3.1. Hospital/Clinic
- 6.3.2. Diagnostic Centers
- 6.3.3. Other End-Users
- 6.1. Market Analysis, Insights and Forecast - by Device Type
- 7. Germany Europe Vascular Access Device Market Analysis, Insights and Forecast, 2020-2032
- 7.1. Market Analysis, Insights and Forecast - by Device Type
- 7.1.1. Central Vascular Access Devices
- 7.1.1.1. Peripherally Inserted Central Catheters
- 7.1.1.2. Percutaneous Non-tunneled Catheters
- 7.1.1.3. Other Central Vascular Access Devices
- 7.1.2. Peripheral Vascular Access Devices
- 7.1.2.1. Peripheral Catheter
- 7.1.2.2. Midline Catheter
- 7.1.2.3. Other Peripheral Vascular Access Devices
- 7.1.1. Central Vascular Access Devices
- 7.2. Market Analysis, Insights and Forecast - by Application
- 7.2.1. Administration of Drugs
- 7.2.2. Administration of Fluid and Nutrition
- 7.2.3. Diagnostics and Testing
- 7.2.4. Other Applications
- 7.3. Market Analysis, Insights and Forecast - by End-User
- 7.3.1. Hospital/Clinic
- 7.3.2. Diagnostic Centers
- 7.3.3. Other End-Users
- 7.1. Market Analysis, Insights and Forecast - by Device Type
- 8. United Kingdom Europe Vascular Access Device Market Analysis, Insights and Forecast, 2020-2032
- 8.1. Market Analysis, Insights and Forecast - by Device Type
- 8.1.1. Central Vascular Access Devices
- 8.1.1.1. Peripherally Inserted Central Catheters
- 8.1.1.2. Percutaneous Non-tunneled Catheters
- 8.1.1.3. Other Central Vascular Access Devices
- 8.1.2. Peripheral Vascular Access Devices
- 8.1.2.1. Peripheral Catheter
- 8.1.2.2. Midline Catheter
- 8.1.2.3. Other Peripheral Vascular Access Devices
- 8.1.1. Central Vascular Access Devices
- 8.2. Market Analysis, Insights and Forecast - by Application
- 8.2.1. Administration of Drugs
- 8.2.2. Administration of Fluid and Nutrition
- 8.2.3. Diagnostics and Testing
- 8.2.4. Other Applications
- 8.3. Market Analysis, Insights and Forecast - by End-User
- 8.3.1. Hospital/Clinic
- 8.3.2. Diagnostic Centers
- 8.3.3. Other End-Users
- 8.1. Market Analysis, Insights and Forecast - by Device Type
- 9. France Europe Vascular Access Device Market Analysis, Insights and Forecast, 2020-2032
- 9.1. Market Analysis, Insights and Forecast - by Device Type
- 9.1.1. Central Vascular Access Devices
- 9.1.1.1. Peripherally Inserted Central Catheters
- 9.1.1.2. Percutaneous Non-tunneled Catheters
- 9.1.1.3. Other Central Vascular Access Devices
- 9.1.2. Peripheral Vascular Access Devices
- 9.1.2.1. Peripheral Catheter
- 9.1.2.2. Midline Catheter
- 9.1.2.3. Other Peripheral Vascular Access Devices
- 9.1.1. Central Vascular Access Devices
- 9.2. Market Analysis, Insights and Forecast - by Application
- 9.2.1. Administration of Drugs
- 9.2.2. Administration of Fluid and Nutrition
- 9.2.3. Diagnostics and Testing
- 9.2.4. Other Applications
- 9.3. Market Analysis, Insights and Forecast - by End-User
- 9.3.1. Hospital/Clinic
- 9.3.2. Diagnostic Centers
- 9.3.3. Other End-Users
- 9.1. Market Analysis, Insights and Forecast - by Device Type
- 10. Italy Europe Vascular Access Device Market Analysis, Insights and Forecast, 2020-2032
- 10.1. Market Analysis, Insights and Forecast - by Device Type
- 10.1.1. Central Vascular Access Devices
- 10.1.1.1. Peripherally Inserted Central Catheters
- 10.1.1.2. Percutaneous Non-tunneled Catheters
- 10.1.1.3. Other Central Vascular Access Devices
- 10.1.2. Peripheral Vascular Access Devices
- 10.1.2.1. Peripheral Catheter
- 10.1.2.2. Midline Catheter
- 10.1.2.3. Other Peripheral Vascular Access Devices
- 10.1.1. Central Vascular Access Devices
- 10.2. Market Analysis, Insights and Forecast - by Application
- 10.2.1. Administration of Drugs
- 10.2.2. Administration of Fluid and Nutrition
- 10.2.3. Diagnostics and Testing
- 10.2.4. Other Applications
- 10.3. Market Analysis, Insights and Forecast - by End-User
- 10.3.1. Hospital/Clinic
- 10.3.2. Diagnostic Centers
- 10.3.3. Other End-Users
- 10.1. Market Analysis, Insights and Forecast - by Device Type
- 11. Spain Europe Vascular Access Device Market Analysis, Insights and Forecast, 2020-2032
- 11.1. Market Analysis, Insights and Forecast - by Device Type
- 11.1.1. Central Vascular Access Devices
- 11.1.1.1. Peripherally Inserted Central Catheters
- 11.1.1.2. Percutaneous Non-tunneled Catheters
- 11.1.1.3. Other Central Vascular Access Devices
- 11.1.2. Peripheral Vascular Access Devices
- 11.1.2.1. Peripheral Catheter
- 11.1.2.2. Midline Catheter
- 11.1.2.3. Other Peripheral Vascular Access Devices
- 11.1.1. Central Vascular Access Devices
- 11.2. Market Analysis, Insights and Forecast - by Application
- 11.2.1. Administration of Drugs
- 11.2.2. Administration of Fluid and Nutrition
- 11.2.3. Diagnostics and Testing
- 11.2.4. Other Applications
- 11.3. Market Analysis, Insights and Forecast - by End-User
- 11.3.1. Hospital/Clinic
- 11.3.2. Diagnostic Centers
- 11.3.3. Other End-Users
- 11.1. Market Analysis, Insights and Forecast - by Device Type
- 12. Rest of Europe Europe Vascular Access Device Market Analysis, Insights and Forecast, 2020-2032
- 12.1. Market Analysis, Insights and Forecast - by Device Type
- 12.1.1. Central Vascular Access Devices
- 12.1.1.1. Peripherally Inserted Central Catheters
- 12.1.1.2. Percutaneous Non-tunneled Catheters
- 12.1.1.3. Other Central Vascular Access Devices
- 12.1.2. Peripheral Vascular Access Devices
- 12.1.2.1. Peripheral Catheter
- 12.1.2.2. Midline Catheter
- 12.1.2.3. Other Peripheral Vascular Access Devices
- 12.1.1. Central Vascular Access Devices
- 12.2. Market Analysis, Insights and Forecast - by Application
- 12.2.1. Administration of Drugs
- 12.2.2. Administration of Fluid and Nutrition
- 12.2.3. Diagnostics and Testing
- 12.2.4. Other Applications
- 12.3. Market Analysis, Insights and Forecast - by End-User
- 12.3.1. Hospital/Clinic
- 12.3.2. Diagnostic Centers
- 12.3.3. Other End-Users
- 12.1. Market Analysis, Insights and Forecast - by Device Type
- 13. Competitive Analysis
- 13.1. Company Profiles
- 13.1.1 Becton Dickinson and Company
- 13.1.1.1. Company Overview
- 13.1.1.2. Products
- 13.1.1.3. Company Financials
- 13.1.1.4. SWOT Analysis
- 13.1.2 Nipro Medical Corporation
- 13.1.2.1. Company Overview
- 13.1.2.2. Products
- 13.1.2.3. Company Financials
- 13.1.2.4. SWOT Analysis
- 13.1.3 ICU Medical Inc
- 13.1.3.1. Company Overview
- 13.1.3.2. Products
- 13.1.3.3. Company Financials
- 13.1.3.4. SWOT Analysis
- 13.1.4 Edwards Lifesciences Corporation
- 13.1.4.1. Company Overview
- 13.1.4.2. Products
- 13.1.4.3. Company Financials
- 13.1.4.4. SWOT Analysis
- 13.1.5 Medtronic
- 13.1.5.1. Company Overview
- 13.1.5.2. Products
- 13.1.5.3. Company Financials
- 13.1.5.4. SWOT Analysis
- 13.1.6 Fresenius Medical Care AG & Co KGaA*List Not Exhaustive
- 13.1.6.1. Company Overview
- 13.1.6.2. Products
- 13.1.6.3. Company Financials
- 13.1.6.4. SWOT Analysis
- 13.1.7 3M
- 13.1.7.1. Company Overview
- 13.1.7.2. Products
- 13.1.7.3. Company Financials
- 13.1.7.4. SWOT Analysis
- 13.1.8 AngioDynamics
- 13.1.8.1. Company Overview
- 13.1.8.2. Products
- 13.1.8.3. Company Financials
- 13.1.8.4. SWOT Analysis
- 13.1.9 Terumo Medical Corporation
- 13.1.9.1. Company Overview
- 13.1.9.2. Products
- 13.1.9.3. Company Financials
- 13.1.9.4. SWOT Analysis
- 13.1.10 Teleflex Incorporated
- 13.1.10.1. Company Overview
- 13.1.10.2. Products
- 13.1.10.3. Company Financials
- 13.1.10.4. SWOT Analysis
- 13.1.11 B Braun SE
- 13.1.11.1. Company Overview
- 13.1.11.2. Products
- 13.1.11.3. Company Financials
- 13.1.11.4. SWOT Analysis
- 13.1.12 Prodimed
- 13.1.12.1. Company Overview
- 13.1.12.2. Products
- 13.1.12.3. Company Financials
- 13.1.12.4. SWOT Analysis
- 13.1.1 Becton Dickinson and Company
- 13.2. Market Entropy
- 13.2.1 Company's Key Areas Served
- 13.2.2 Recent Developments
- 13.3. Company Market Share Analysis 2025
- 13.3.1 Top 5 Companies Market Share Analysis
- 13.3.2 Top 3 Companies Market Share Analysis
- 13.4. List of Potential Customers
- 14. Research Methodology
List of Figures
- Figure 1: Europe Vascular Access Device Market Revenue Breakdown (billion, %) by Product 2025 & 2033
- Figure 2: Europe Vascular Access Device Market Share (%) by Company 2025
List of Tables
- Table 1: Europe Vascular Access Device Market Revenue billion Forecast, by Device Type 2020 & 2033
- Table 2: Europe Vascular Access Device Market Revenue billion Forecast, by Application 2020 & 2033
- Table 3: Europe Vascular Access Device Market Revenue billion Forecast, by End-User 2020 & 2033
- Table 4: Europe Vascular Access Device Market Revenue billion Forecast, by Region 2020 & 2033
- Table 5: Europe Vascular Access Device Market Revenue billion Forecast, by Device Type 2020 & 2033
- Table 6: Europe Vascular Access Device Market Revenue billion Forecast, by Application 2020 & 2033
- Table 7: Europe Vascular Access Device Market Revenue billion Forecast, by End-User 2020 & 2033
- Table 8: Europe Vascular Access Device Market Revenue billion Forecast, by Country 2020 & 2033
- Table 9: Europe Vascular Access Device Market Revenue billion Forecast, by Device Type 2020 & 2033
- Table 10: Europe Vascular Access Device Market Revenue billion Forecast, by Application 2020 & 2033
- Table 11: Europe Vascular Access Device Market Revenue billion Forecast, by End-User 2020 & 2033
- Table 12: Europe Vascular Access Device Market Revenue billion Forecast, by Country 2020 & 2033
- Table 13: Europe Vascular Access Device Market Revenue billion Forecast, by Device Type 2020 & 2033
- Table 14: Europe Vascular Access Device Market Revenue billion Forecast, by Application 2020 & 2033
- Table 15: Europe Vascular Access Device Market Revenue billion Forecast, by End-User 2020 & 2033
- Table 16: Europe Vascular Access Device Market Revenue billion Forecast, by Country 2020 & 2033
- Table 17: Europe Vascular Access Device Market Revenue billion Forecast, by Device Type 2020 & 2033
- Table 18: Europe Vascular Access Device Market Revenue billion Forecast, by Application 2020 & 2033
- Table 19: Europe Vascular Access Device Market Revenue billion Forecast, by End-User 2020 & 2033
- Table 20: Europe Vascular Access Device Market Revenue billion Forecast, by Country 2020 & 2033
- Table 21: Europe Vascular Access Device Market Revenue billion Forecast, by Device Type 2020 & 2033
- Table 22: Europe Vascular Access Device Market Revenue billion Forecast, by Application 2020 & 2033
- Table 23: Europe Vascular Access Device Market Revenue billion Forecast, by End-User 2020 & 2033
- Table 24: Europe Vascular Access Device Market Revenue billion Forecast, by Country 2020 & 2033
- Table 25: Europe Vascular Access Device Market Revenue billion Forecast, by Device Type 2020 & 2033
- Table 26: Europe Vascular Access Device Market Revenue billion Forecast, by Application 2020 & 2033
- Table 27: Europe Vascular Access Device Market Revenue billion Forecast, by End-User 2020 & 2033
- Table 28: Europe Vascular Access Device Market Revenue billion Forecast, by Country 2020 & 2033
Frequently Asked Questions
1. What is the projected Compound Annual Growth Rate (CAGR) of the Europe Vascular Access Device Market?
The projected CAGR is approximately 6.91%.
2. Which companies are prominent players in the Europe Vascular Access Device Market?
Key companies in the market include Becton Dickinson and Company, Nipro Medical Corporation, ICU Medical Inc, Edwards Lifesciences Corporation, Medtronic, Fresenius Medical Care AG & Co KGaA*List Not Exhaustive, 3M, AngioDynamics, Terumo Medical Corporation, Teleflex Incorporated, B Braun SE, Prodimed.
3. What are the main segments of the Europe Vascular Access Device Market?
The market segments include Device Type, Application, End-User.
4. Can you provide details about the market size?
The market size is estimated to be USD 1.64 billion as of 2022.
5. What are some drivers contributing to market growth?
Growing prevalence of Chronic Disease; Increasing Number of Chemotherapy Procedures with High Hospitalization Rates; Rising Use of Vascular Access Devices among Paediatric Patients.
6. What are the notable trends driving market growth?
The Administration of Drugs Segment is Expected to Garner Significant Share of the Market.
7. Are there any restraints impacting market growth?
Risks Associated with Catheter Usage; Stringent Regulations and Product Recalls.
8. Can you provide examples of recent developments in the market?
June 2023: BIOTRONIK launched its Oscar (One Solution: Cross. Adjust. Restore) multifunctional peripheral catheter and started promotional activities at LINC, the Leipzig Interventional Course held in Leipzig, Germany. The device offers access into and to dilate stenoses in femoral, popliteal, and infrapopliteal arteries.
9. What pricing options are available for accessing the report?
Pricing options include single-user, multi-user, and enterprise licenses priced at USD 4750, USD 5250, and USD 8750 respectively.
10. Is the market size provided in terms of value or volume?
The market size is provided in terms of value, measured in billion.
11. Are there any specific market keywords associated with the report?
Yes, the market keyword associated with the report is "Europe Vascular Access Device Market," which aids in identifying and referencing the specific market segment covered.
12. How do I determine which pricing option suits my needs best?
The pricing options vary based on user requirements and access needs. Individual users may opt for single-user licenses, while businesses requiring broader access may choose multi-user or enterprise licenses for cost-effective access to the report.
13. Are there any additional resources or data provided in the Europe Vascular Access Device Market report?
While the report offers comprehensive insights, it's advisable to review the specific contents or supplementary materials provided to ascertain if additional resources or data are available.
14. How can I stay updated on further developments or reports in the Europe Vascular Access Device Market?
To stay informed about further developments, trends, and reports in the Europe Vascular Access Device Market, consider subscribing to industry newsletters, following relevant companies and organizations, or regularly checking reputable industry news sources and publications.
Methodology
Step 1 - Identification of Relevant Samples Size from Population Database



Step 2 - Approaches for Defining Global Market Size (Value, Volume* & Price*)

Note*: In applicable scenarios
Step 3 - Data Sources
Primary Research
- Web Analytics
- Survey Reports
- Research Institute
- Latest Research Reports
- Opinion Leaders
Secondary Research
- Annual Reports
- White Paper
- Latest Press Release
- Industry Association
- Paid Database
- Investor Presentations

Step 4 - Data Triangulation
Involves using different sources of information in order to increase the validity of a study
These sources are likely to be stakeholders in a program - participants, other researchers, program staff, other community members, and so on.
Then we put all data in single framework & apply various statistical tools to find out the dynamic on the market.
During the analysis stage, feedback from the stakeholder groups would be compared to determine areas of agreement as well as areas of divergence


