Key Insights
The circRNA-LNP formulation market is poised for significant expansion, projected to reach $300.72 million in 2025 with a robust Compound Annual Growth Rate (CAGR) of 5.67% through 2033. This dynamic growth is primarily fueled by the burgeoning demand for advanced therapeutic delivery systems, particularly within the pharmaceutical and scientific research sectors. Circular RNAs (circRNAs) are gaining substantial traction due to their inherent stability and regulatory roles in gene expression, making them attractive candidates for novel drug development. Lipid Nanoparticles (LNPs) have emerged as the leading delivery vehicle for these RNA therapeutics, offering efficient cellular uptake and protection from degradation. The increasing investment in RNA-based therapies for a wide range of diseases, including oncology, infectious diseases, and genetic disorders, directly translates into a heightened need for sophisticated circRNA-LNP formulation technologies. Moreover, ongoing advancements in LNP engineering, focusing on improved targeting, reduced immunogenicity, and enhanced manufacturing scalability, are further bolstering market confidence and driving innovation.
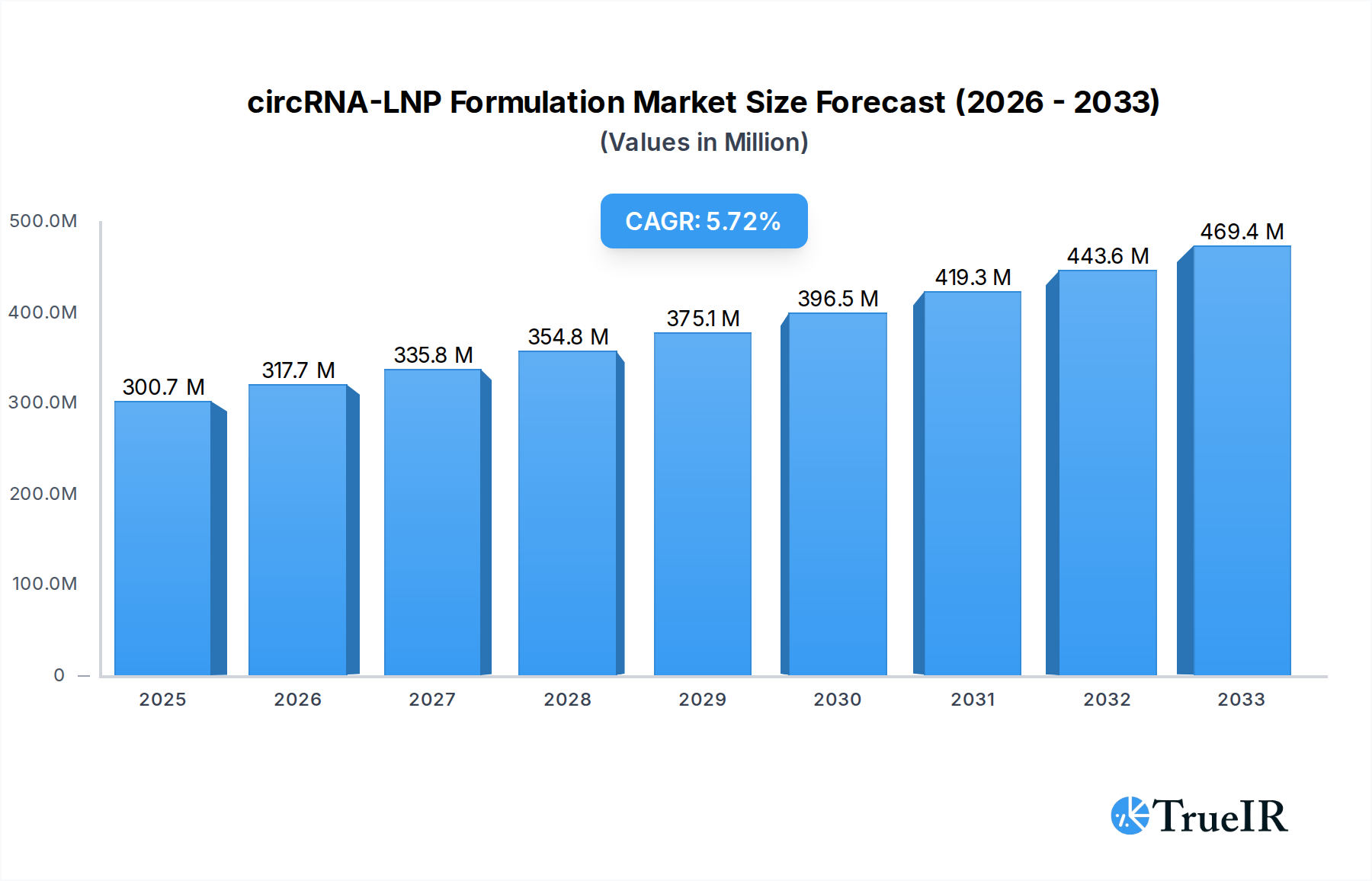
circRNA-LNP Formulation Market Size (In Million)

The market's trajectory is further shaped by a growing emphasis on personalized medicine and the development of customized circRNA-LNP formulations tailored to specific patient needs and therapeutic targets. Companies are increasingly focusing on developing both "premade" solutions for rapid research and development, as well as highly specialized "customized" formulations for advanced clinical applications. Key market drivers include the expanding pipeline of RNA-based drugs, the increasing prevalence of chronic and rare diseases requiring novel treatment modalities, and supportive regulatory frameworks encouraging the development of innovative therapies. While the market enjoys strong growth, challenges such as manufacturing scalability and the high cost of specialized formulations need continuous attention. Nonetheless, the significant therapeutic potential of circRNA-LNP formulations, coupled with a supportive innovation ecosystem, positions this market for sustained and impactful growth in the coming years.
circRNA-LNP Formulation Company Market Share

This in-depth report provides an authoritative analysis of the circRNA-LNP Formulation market, a critical component for the advancement of next-generation therapeutics. Leveraging high-volume keywords such as "circRNA delivery," "lipid nanoparticle formulation," "RNA therapeutics," "gene editing," and "drug delivery systems," this SEO-optimized report is designed to engage researchers, pharmaceutical companies, and investors within the biotechnology and life sciences sectors. We delve into market dynamics, technological innovations, competitive landscapes, and future projections, offering actionable insights for stakeholders.
circRNA-LNP Formulation Market Structure & Competitive Landscape
The circRNA-LNP Formulation market exhibits a dynamic and evolving competitive landscape, characterized by moderate to high market concentration within key specialized areas of LNP development and circRNA synthesis. Innovation remains a primary driver, fueled by substantial investments in research and development aimed at optimizing LNP efficiency, targeting capabilities, and biosafety for circRNA delivery. Regulatory frameworks surrounding RNA-based therapeutics are progressively maturing, influencing market entry strategies and demanding rigorous clinical validation. While direct product substitutes for circRNA-LNP formulations are nascent, advancements in other nucleic acid delivery platforms, such as viral vectors and other non-viral systems, represent indirect competitive forces. End-user segmentation reveals a strong demand from the Pharmaceuticals sector for therapeutic applications, alongside significant utilization within Scientific Research for preclinical studies and platform development. Mergers and acquisitions (M&A) are anticipated to play a crucial role in consolidating expertise and market share, with an estimated volume of approximately 10-15 M&A activities projected over the forecast period. Key players are actively seeking strategic partnerships to leverage complementary technologies and expand their manufacturing capabilities. The market is currently characterized by a few leading innovators in LNP technology and circRNA synthesis, alongside a growing number of emerging players specializing in specific formulation aspects. Concentration ratios for core LNP technologies are estimated to be in the range of 0.50-0.70, indicating a degree of dominance by a select group of companies.
circRNA-LNP Formulation Market Trends & Opportunities
The global circRNA-LNP Formulation market is poised for substantial growth, driven by the burgeoning field of RNA therapeutics and the unique advantages offered by circular RNAs. Market size is projected to expand from an estimated USD 2,500 million in the base year 2025 to potentially exceed USD 10,000 million by 2033, reflecting a Compound Annual Growth Rate (CAGR) of approximately 15-20%. Technological shifts are central to this expansion, with continuous advancements in LNP chemistry, lipid composition optimization, and manufacturing scalability enabling more efficient and precise delivery of circRNAs. The development of novel lipid excipients and formulation strategies is critical for overcoming biological barriers and enhancing intracellular uptake. Consumer preferences, particularly within the pharmaceutical industry, are increasingly favoring non-immunogenic and highly specific delivery systems, making LNPs an attractive modality. The growing understanding of circRNAs' therapeutic potential, including their inherent stability and role in gene regulation, is creating unprecedented opportunities for developing treatments for a wide range of diseases, from oncology to rare genetic disorders. Competitive dynamics are intensifying as more companies enter the space, fostering innovation and driving down costs. Early-stage market penetration rates are still relatively low, indicating significant headroom for growth as clinical trials progress and regulatory approvals are secured for circRNA-based therapies. Opportunities lie in developing specialized LNP formulations tailored to specific circRNA payloads, improving manufacturing processes to achieve large-scale production of over 1,000 million doses annually, and establishing robust analytical methods for quality control. The integration of artificial intelligence and machine learning in LNP design and optimization is another emerging trend that promises to accelerate development timelines and improve formulation success rates. Furthermore, the expanding pipeline of circRNA therapeutics in preclinical and early-stage clinical development acts as a significant tailwind for the circRNA-LNP formulation market, creating a sustained demand for advanced delivery solutions. The increasing recognition of circRNAs as potential therapeutic agents, distinct from linear mRNA, is a key factor driving investment and innovation in this specific LNP formulation niche, contributing to an estimated market value of over USD 2,500 million in the base year.
Dominant Markets & Segments in circRNA-LNP Formulation
The circRNA-LNP Formulation market is currently dominated by regions and segments that exhibit strong foundational capabilities in biotechnology, advanced pharmaceutical research, and supportive regulatory environments.
Leading Region:
- North America, particularly the United States, is anticipated to remain the dominant market due to its robust biopharmaceutical ecosystem, substantial government funding for scientific research, and a high concentration of leading pharmaceutical and biotechnology companies. The presence of established LNP technology developers and a strong pipeline of RNA-based drug candidates significantly contribute to its leadership. Infrastructure supporting advanced manufacturing and a well-defined regulatory pathway for novel therapeutics further solidify its position.
Leading Country within North America:
- United States: Continued investment in R&D, a large pool of skilled scientific talent, and a favorable venture capital landscape enable rapid innovation and commercialization of circRNA-LNP technologies. The country is a hub for both LNP formulation expertise and the discovery of novel circRNA applications.
Dominant Application Segment:
- Pharmaceuticals: This segment represents the largest and fastest-growing application for circRNA-LNP formulations. The potential of circRNAs as therapeutic agents for a vast array of diseases, including cancer, infectious diseases, and genetic disorders, drives significant demand for advanced delivery systems. Pharmaceutical companies are investing heavily in developing circRNA-based drugs, necessitating scalable and efficient LNP formulation capabilities. The market value within this segment is estimated to be over USD 2,000 million in the base year.
- Key Growth Drivers:
- Unmet medical needs in various therapeutic areas.
- Advancements in understanding circRNA biology and therapeutic mechanisms.
- Increased pharmaceutical investment in RNA therapeutics.
- Growing pipeline of circRNA drugs in clinical development.
- Key Growth Drivers:
Dominant Type Segment:
- Customized: While premade formulations offer initial utility, the Customized segment is experiencing exponential growth. The diverse nature of circRNAs, their varied therapeutic targets, and the requirement for highly specific delivery profiles necessitate tailor-made LNP formulations. Companies are increasingly seeking custom solutions to optimize payload encapsulation, targeting efficiency, and immunogenicity for their unique circRNA candidates. The ability to fine-tune lipid composition, particle size, and surface modifications allows for enhanced therapeutic outcomes.
- Key Growth Drivers:
- Need for optimized delivery for specific circRNA sequences and targets.
- Demand for personalized medicine approaches.
- Advancements in formulation technologies enabling precise customization.
- Overcoming unique delivery challenges for different cell types and tissues.
- Key Growth Drivers:
The interplay between these dominant segments—North America's robust R&D environment, the pharmaceutical industry's therapeutic ambitions, and the growing demand for bespoke formulation solutions—underscores the pivotal role of circRNA-LNP formulations in shaping the future of medicine.
circRNA-LNP Formulation Product Analysis
The circRNA-LNP Formulation market is characterized by continuous product innovation focused on enhancing delivery efficiency, payload protection, and therapeutic specificity. Leading products differentiate themselves through proprietary lipid compositions, optimized particle sizes ranging from 50-200 nanometers, and advanced surface modifications for targeted delivery to specific cell types or tissues. Technological advancements in self-assembling lipid nanoparticles enable superior encapsulation of circRNA molecules, ensuring their stability and preventing premature degradation. Competitive advantages stem from superior transfection efficiency, reduced immunogenicity, and the ability to scale up manufacturing to meet the demands of clinical trials and commercialization, with potential to produce over 500 million doses annually.
Key Drivers, Barriers & Challenges in circRNA-LNP Formulation
Key Drivers:
The circRNA-LNP Formulation market is propelled by several key drivers. Technological innovation in LNP design, lipid chemistry, and manufacturing processes is paramount, enabling more efficient and safer delivery of circRNA payloads. Increasing understanding of circRNA biology and its therapeutic potential across various disease areas, including oncology and rare genetic disorders, fuels R&D investment. Growing demand for RNA-based therapeutics, exemplified by the success of mRNA vaccines, creates a favorable market environment. Supportive regulatory pathways in key markets are also facilitating the development and approval of novel circRNA-based therapies.
Barriers & Challenges:
Despite its potential, the market faces significant challenges. Complex regulatory hurdles for novel RNA therapeutics and their delivery systems can prolong development timelines and increase costs, with an estimated 3-5 years for regulatory approval. Manufacturing scalability and cost-effectiveness remain critical concerns, particularly for large-scale therapeutic applications, with current costs per dose potentially ranging from hundreds to thousands of USD. Supply chain complexities for specialized lipids and reagents can impact production consistency and availability, potentially affecting the supply of over 100 million units annually. Intellectual property landscapes are intricate, with numerous patents related to LNP technologies and circRNA applications, necessitating careful navigation for new entrants. Furthermore, potential immunogenicity and off-target effects of LNP formulations require rigorous preclinical and clinical evaluation.
Growth Drivers in the circRNA-LNP Formulation Market
Key growth drivers in the circRNA-LNP Formulation market include rapid advancements in lipid nanoparticle technology, leading to enhanced encapsulation efficiency and targeted delivery capabilities. The burgeoning field of RNA therapeutics, with circRNAs emerging as a promising class due to their inherent stability and gene regulatory functions, is a significant catalyst. Economic factors such as increasing investment in biotechnology R&D and venture capital funding for RNA-focused startups are providing substantial financial impetus. Regulatory tailwinds, with evolving frameworks for nucleic acid-based therapies, are also fostering market growth.
Challenges Impacting circRNA-LNP Formulation Growth
Challenges impacting circRNA-LNP formulation growth encompass the complexities of regulatory approval pathways for novel RNA-based therapies, which can be time-consuming and costly. Manufacturing scalability and cost-efficiency remain significant barriers, particularly for achieving widespread therapeutic accessibility, with production costs potentially exceeding USD 500 per million doses. Supply chain vulnerabilities for specialized lipids and reagents can disrupt consistent production. Potential immunogenicity and off-target effects of LNP delivery systems necessitate extensive safety testing, adding to development timelines and costs.
Key Players Shaping the circRNA-LNP Formulation Market
- Orna Therapeutics
- GenScript
- Creative Biogene
- Catalent
- Creative Biolabs
- VectorBuilder
- Guangzhou Geneseed Biotech. Co.,Ltd.
- Porton Pharma Solutions,Ltd
- Recipharm AB
- Precigenome LLC
Significant circRNA-LNP Formulation Industry Milestones
- 2019: Initial research publications highlighting the potential of circRNAs for therapeutic applications begin to gain traction.
- 2020: Increased investment in RNA delivery technologies, including lipid nanoparticles, driven by the success of mRNA vaccines.
- 2021: Early-stage development of proprietary LNP formulations specifically designed for circRNA encapsulation by several biotech firms.
- 2022: First preclinical studies demonstrating the efficacy of circRNA delivered via LNPs for various disease models.
- 2023: Establishment of dedicated circRNA discovery and development platforms by major pharmaceutical companies.
- 2024: Advancements in manufacturing technologies enabling the production of larger quantities of circRNA and corresponding LNP formulations, with potential to produce over 10 million units.
- 2025 (Estimated): Anticipated commencement of early-phase clinical trials for circRNA-LNP therapeutics.
Future Outlook for circRNA-LNP Formulation Market
The future outlook for the circRNA-LNP Formulation market is exceptionally bright, fueled by ongoing innovation in LNP design and a deeper understanding of circRNA's therapeutic capabilities. Strategic opportunities lie in developing next-generation LNPs with enhanced targeting specificity and reduced immunogenicity, alongside advancements in scalable and cost-effective manufacturing processes capable of producing over 1,000 million doses annually. The expanding pipeline of circRNA therapeutics across diverse disease indications promises sustained demand for advanced formulation solutions, positioning the market for significant growth and transformative impact on human health.
circRNA-LNP Formulation Segmentation
-
1. Application
- 1.1. Scientific Research
- 1.2. Pharmaceuticals
-
2. Types
- 2.1. Premade
- 2.2. Customized
circRNA-LNP Formulation Segmentation By Geography
-
1. North America
- 1.1. United States
- 1.2. Canada
- 1.3. Mexico
-
2. South America
- 2.1. Brazil
- 2.2. Argentina
- 2.3. Rest of South America
-
3. Europe
- 3.1. United Kingdom
- 3.2. Germany
- 3.3. France
- 3.4. Italy
- 3.5. Spain
- 3.6. Russia
- 3.7. Benelux
- 3.8. Nordics
- 3.9. Rest of Europe
-
4. Middle East & Africa
- 4.1. Turkey
- 4.2. Israel
- 4.3. GCC
- 4.4. North Africa
- 4.5. South Africa
- 4.6. Rest of Middle East & Africa
-
5. Asia Pacific
- 5.1. China
- 5.2. India
- 5.3. Japan
- 5.4. South Korea
- 5.5. ASEAN
- 5.6. Oceania
- 5.7. Rest of Asia Pacific
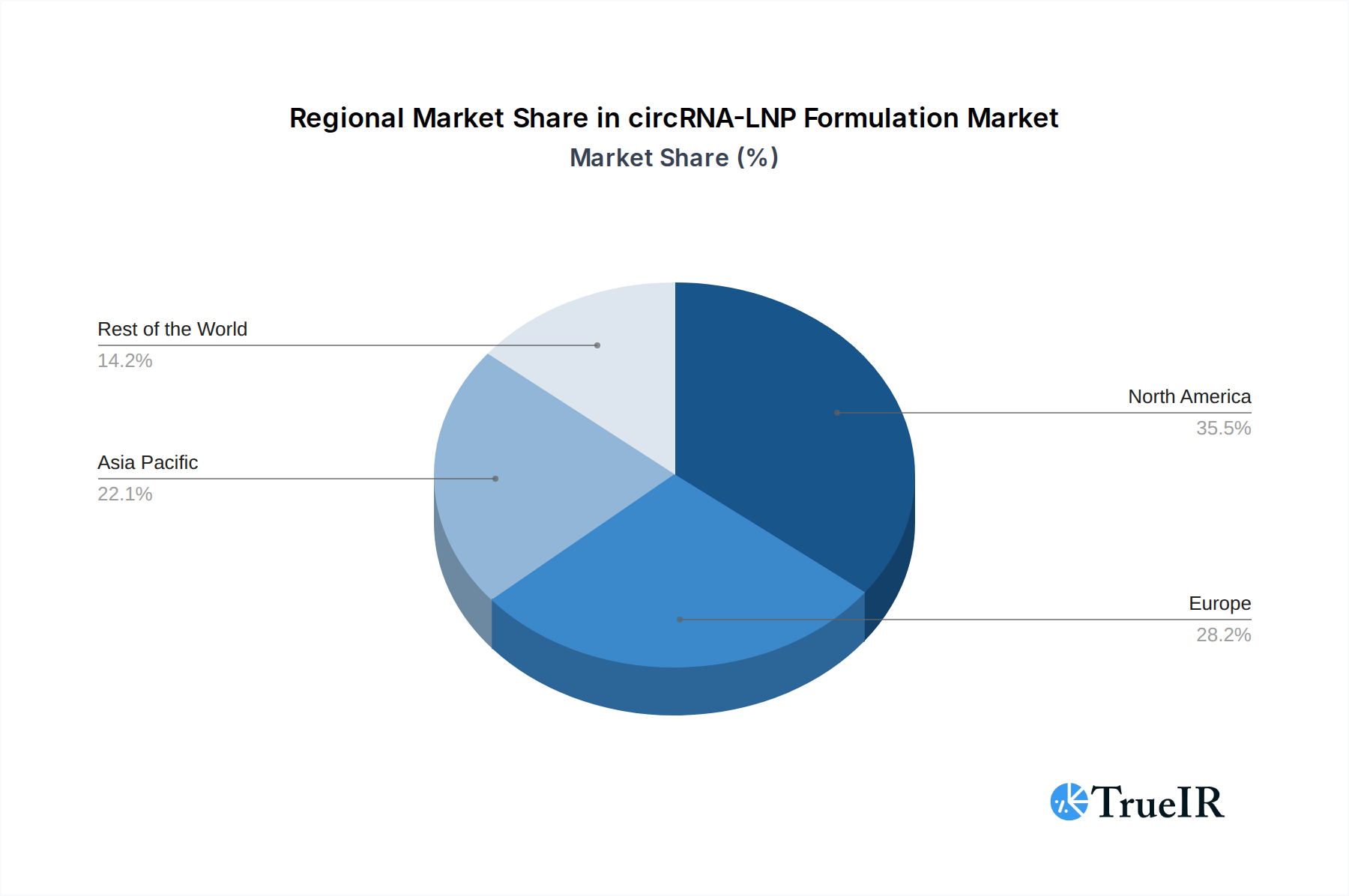
circRNA-LNP Formulation Regional Market Share

Geographic Coverage of circRNA-LNP Formulation
circRNA-LNP Formulation REPORT HIGHLIGHTS
| Aspects | Details |
|---|---|
| Study Period | 2020-2034 |
| Base Year | 2025 |
| Estimated Year | 2026 |
| Forecast Period | 2026-2034 |
| Historical Period | 2020-2025 |
| Growth Rate | CAGR of 5.67% from 2020-2034 |
| Segmentation |
|
Table of Contents
- 1. Introduction
- 1.1. Research Scope
- 1.2. Market Segmentation
- 1.3. Research Methodology
- 1.4. Definitions and Assumptions
- 2. Executive Summary
- 2.1. Introduction
- 3. Market Dynamics
- 3.1. Introduction
- 3.2. Market Drivers
- 3.3. Market Restrains
- 3.4. Market Trends
- 4. Market Factor Analysis
- 4.1. Porters Five Forces
- 4.2. Supply/Value Chain
- 4.3. PESTEL analysis
- 4.4. Market Entropy
- 4.5. Patent/Trademark Analysis
- 5. Global circRNA-LNP Formulation Analysis, Insights and Forecast, 2020-2032
- 5.1. Market Analysis, Insights and Forecast - by Application
- 5.1.1. Scientific Research
- 5.1.2. Pharmaceuticals
- 5.2. Market Analysis, Insights and Forecast - by Types
- 5.2.1. Premade
- 5.2.2. Customized
- 5.3. Market Analysis, Insights and Forecast - by Region
- 5.3.1. North America
- 5.3.2. South America
- 5.3.3. Europe
- 5.3.4. Middle East & Africa
- 5.3.5. Asia Pacific
- 5.1. Market Analysis, Insights and Forecast - by Application
- 6. North America circRNA-LNP Formulation Analysis, Insights and Forecast, 2020-2032
- 6.1. Market Analysis, Insights and Forecast - by Application
- 6.1.1. Scientific Research
- 6.1.2. Pharmaceuticals
- 6.2. Market Analysis, Insights and Forecast - by Types
- 6.2.1. Premade
- 6.2.2. Customized
- 6.1. Market Analysis, Insights and Forecast - by Application
- 7. South America circRNA-LNP Formulation Analysis, Insights and Forecast, 2020-2032
- 7.1. Market Analysis, Insights and Forecast - by Application
- 7.1.1. Scientific Research
- 7.1.2. Pharmaceuticals
- 7.2. Market Analysis, Insights and Forecast - by Types
- 7.2.1. Premade
- 7.2.2. Customized
- 7.1. Market Analysis, Insights and Forecast - by Application
- 8. Europe circRNA-LNP Formulation Analysis, Insights and Forecast, 2020-2032
- 8.1. Market Analysis, Insights and Forecast - by Application
- 8.1.1. Scientific Research
- 8.1.2. Pharmaceuticals
- 8.2. Market Analysis, Insights and Forecast - by Types
- 8.2.1. Premade
- 8.2.2. Customized
- 8.1. Market Analysis, Insights and Forecast - by Application
- 9. Middle East & Africa circRNA-LNP Formulation Analysis, Insights and Forecast, 2020-2032
- 9.1. Market Analysis, Insights and Forecast - by Application
- 9.1.1. Scientific Research
- 9.1.2. Pharmaceuticals
- 9.2. Market Analysis, Insights and Forecast - by Types
- 9.2.1. Premade
- 9.2.2. Customized
- 9.1. Market Analysis, Insights and Forecast - by Application
- 10. Asia Pacific circRNA-LNP Formulation Analysis, Insights and Forecast, 2020-2032
- 10.1. Market Analysis, Insights and Forecast - by Application
- 10.1.1. Scientific Research
- 10.1.2. Pharmaceuticals
- 10.2. Market Analysis, Insights and Forecast - by Types
- 10.2.1. Premade
- 10.2.2. Customized
- 10.1. Market Analysis, Insights and Forecast - by Application
- 11. Competitive Analysis
- 11.1. Global Market Share Analysis 2025
- 11.2. Company Profiles
- 11.2.1 Orna Therapeutics
- 11.2.1.1. Overview
- 11.2.1.2. Products
- 11.2.1.3. SWOT Analysis
- 11.2.1.4. Recent Developments
- 11.2.1.5. Financials (Based on Availability)
- 11.2.2 GenScript
- 11.2.2.1. Overview
- 11.2.2.2. Products
- 11.2.2.3. SWOT Analysis
- 11.2.2.4. Recent Developments
- 11.2.2.5. Financials (Based on Availability)
- 11.2.3 Creative Biogene
- 11.2.3.1. Overview
- 11.2.3.2. Products
- 11.2.3.3. SWOT Analysis
- 11.2.3.4. Recent Developments
- 11.2.3.5. Financials (Based on Availability)
- 11.2.4 Catalent
- 11.2.4.1. Overview
- 11.2.4.2. Products
- 11.2.4.3. SWOT Analysis
- 11.2.4.4. Recent Developments
- 11.2.4.5. Financials (Based on Availability)
- 11.2.5 Creative Biolabs
- 11.2.5.1. Overview
- 11.2.5.2. Products
- 11.2.5.3. SWOT Analysis
- 11.2.5.4. Recent Developments
- 11.2.5.5. Financials (Based on Availability)
- 11.2.6 VectorBuilder
- 11.2.6.1. Overview
- 11.2.6.2. Products
- 11.2.6.3. SWOT Analysis
- 11.2.6.4. Recent Developments
- 11.2.6.5. Financials (Based on Availability)
- 11.2.7 Guangzhou Geneseed Biotech. Co.
- 11.2.7.1. Overview
- 11.2.7.2. Products
- 11.2.7.3. SWOT Analysis
- 11.2.7.4. Recent Developments
- 11.2.7.5. Financials (Based on Availability)
- 11.2.8 Ltd.
- 11.2.8.1. Overview
- 11.2.8.2. Products
- 11.2.8.3. SWOT Analysis
- 11.2.8.4. Recent Developments
- 11.2.8.5. Financials (Based on Availability)
- 11.2.9 Porton Pharma Solutions
- 11.2.9.1. Overview
- 11.2.9.2. Products
- 11.2.9.3. SWOT Analysis
- 11.2.9.4. Recent Developments
- 11.2.9.5. Financials (Based on Availability)
- 11.2.10 Ltd
- 11.2.10.1. Overview
- 11.2.10.2. Products
- 11.2.10.3. SWOT Analysis
- 11.2.10.4. Recent Developments
- 11.2.10.5. Financials (Based on Availability)
- 11.2.11 Recipharm AB
- 11.2.11.1. Overview
- 11.2.11.2. Products
- 11.2.11.3. SWOT Analysis
- 11.2.11.4. Recent Developments
- 11.2.11.5. Financials (Based on Availability)
- 11.2.12 Precigenome LLC
- 11.2.12.1. Overview
- 11.2.12.2. Products
- 11.2.12.3. SWOT Analysis
- 11.2.12.4. Recent Developments
- 11.2.12.5. Financials (Based on Availability)
- 11.2.1 Orna Therapeutics
List of Figures
- Figure 1: Global circRNA-LNP Formulation Revenue Breakdown (million, %) by Region 2025 & 2033
- Figure 2: Global circRNA-LNP Formulation Volume Breakdown (K, %) by Region 2025 & 2033
- Figure 3: North America circRNA-LNP Formulation Revenue (million), by Application 2025 & 2033
- Figure 4: North America circRNA-LNP Formulation Volume (K), by Application 2025 & 2033
- Figure 5: North America circRNA-LNP Formulation Revenue Share (%), by Application 2025 & 2033
- Figure 6: North America circRNA-LNP Formulation Volume Share (%), by Application 2025 & 2033
- Figure 7: North America circRNA-LNP Formulation Revenue (million), by Types 2025 & 2033
- Figure 8: North America circRNA-LNP Formulation Volume (K), by Types 2025 & 2033
- Figure 9: North America circRNA-LNP Formulation Revenue Share (%), by Types 2025 & 2033
- Figure 10: North America circRNA-LNP Formulation Volume Share (%), by Types 2025 & 2033
- Figure 11: North America circRNA-LNP Formulation Revenue (million), by Country 2025 & 2033
- Figure 12: North America circRNA-LNP Formulation Volume (K), by Country 2025 & 2033
- Figure 13: North America circRNA-LNP Formulation Revenue Share (%), by Country 2025 & 2033
- Figure 14: North America circRNA-LNP Formulation Volume Share (%), by Country 2025 & 2033
- Figure 15: South America circRNA-LNP Formulation Revenue (million), by Application 2025 & 2033
- Figure 16: South America circRNA-LNP Formulation Volume (K), by Application 2025 & 2033
- Figure 17: South America circRNA-LNP Formulation Revenue Share (%), by Application 2025 & 2033
- Figure 18: South America circRNA-LNP Formulation Volume Share (%), by Application 2025 & 2033
- Figure 19: South America circRNA-LNP Formulation Revenue (million), by Types 2025 & 2033
- Figure 20: South America circRNA-LNP Formulation Volume (K), by Types 2025 & 2033
- Figure 21: South America circRNA-LNP Formulation Revenue Share (%), by Types 2025 & 2033
- Figure 22: South America circRNA-LNP Formulation Volume Share (%), by Types 2025 & 2033
- Figure 23: South America circRNA-LNP Formulation Revenue (million), by Country 2025 & 2033
- Figure 24: South America circRNA-LNP Formulation Volume (K), by Country 2025 & 2033
- Figure 25: South America circRNA-LNP Formulation Revenue Share (%), by Country 2025 & 2033
- Figure 26: South America circRNA-LNP Formulation Volume Share (%), by Country 2025 & 2033
- Figure 27: Europe circRNA-LNP Formulation Revenue (million), by Application 2025 & 2033
- Figure 28: Europe circRNA-LNP Formulation Volume (K), by Application 2025 & 2033
- Figure 29: Europe circRNA-LNP Formulation Revenue Share (%), by Application 2025 & 2033
- Figure 30: Europe circRNA-LNP Formulation Volume Share (%), by Application 2025 & 2033
- Figure 31: Europe circRNA-LNP Formulation Revenue (million), by Types 2025 & 2033
- Figure 32: Europe circRNA-LNP Formulation Volume (K), by Types 2025 & 2033
- Figure 33: Europe circRNA-LNP Formulation Revenue Share (%), by Types 2025 & 2033
- Figure 34: Europe circRNA-LNP Formulation Volume Share (%), by Types 2025 & 2033
- Figure 35: Europe circRNA-LNP Formulation Revenue (million), by Country 2025 & 2033
- Figure 36: Europe circRNA-LNP Formulation Volume (K), by Country 2025 & 2033
- Figure 37: Europe circRNA-LNP Formulation Revenue Share (%), by Country 2025 & 2033
- Figure 38: Europe circRNA-LNP Formulation Volume Share (%), by Country 2025 & 2033
- Figure 39: Middle East & Africa circRNA-LNP Formulation Revenue (million), by Application 2025 & 2033
- Figure 40: Middle East & Africa circRNA-LNP Formulation Volume (K), by Application 2025 & 2033
- Figure 41: Middle East & Africa circRNA-LNP Formulation Revenue Share (%), by Application 2025 & 2033
- Figure 42: Middle East & Africa circRNA-LNP Formulation Volume Share (%), by Application 2025 & 2033
- Figure 43: Middle East & Africa circRNA-LNP Formulation Revenue (million), by Types 2025 & 2033
- Figure 44: Middle East & Africa circRNA-LNP Formulation Volume (K), by Types 2025 & 2033
- Figure 45: Middle East & Africa circRNA-LNP Formulation Revenue Share (%), by Types 2025 & 2033
- Figure 46: Middle East & Africa circRNA-LNP Formulation Volume Share (%), by Types 2025 & 2033
- Figure 47: Middle East & Africa circRNA-LNP Formulation Revenue (million), by Country 2025 & 2033
- Figure 48: Middle East & Africa circRNA-LNP Formulation Volume (K), by Country 2025 & 2033
- Figure 49: Middle East & Africa circRNA-LNP Formulation Revenue Share (%), by Country 2025 & 2033
- Figure 50: Middle East & Africa circRNA-LNP Formulation Volume Share (%), by Country 2025 & 2033
- Figure 51: Asia Pacific circRNA-LNP Formulation Revenue (million), by Application 2025 & 2033
- Figure 52: Asia Pacific circRNA-LNP Formulation Volume (K), by Application 2025 & 2033
- Figure 53: Asia Pacific circRNA-LNP Formulation Revenue Share (%), by Application 2025 & 2033
- Figure 54: Asia Pacific circRNA-LNP Formulation Volume Share (%), by Application 2025 & 2033
- Figure 55: Asia Pacific circRNA-LNP Formulation Revenue (million), by Types 2025 & 2033
- Figure 56: Asia Pacific circRNA-LNP Formulation Volume (K), by Types 2025 & 2033
- Figure 57: Asia Pacific circRNA-LNP Formulation Revenue Share (%), by Types 2025 & 2033
- Figure 58: Asia Pacific circRNA-LNP Formulation Volume Share (%), by Types 2025 & 2033
- Figure 59: Asia Pacific circRNA-LNP Formulation Revenue (million), by Country 2025 & 2033
- Figure 60: Asia Pacific circRNA-LNP Formulation Volume (K), by Country 2025 & 2033
- Figure 61: Asia Pacific circRNA-LNP Formulation Revenue Share (%), by Country 2025 & 2033
- Figure 62: Asia Pacific circRNA-LNP Formulation Volume Share (%), by Country 2025 & 2033
List of Tables
- Table 1: Global circRNA-LNP Formulation Revenue million Forecast, by Application 2020 & 2033
- Table 2: Global circRNA-LNP Formulation Volume K Forecast, by Application 2020 & 2033
- Table 3: Global circRNA-LNP Formulation Revenue million Forecast, by Types 2020 & 2033
- Table 4: Global circRNA-LNP Formulation Volume K Forecast, by Types 2020 & 2033
- Table 5: Global circRNA-LNP Formulation Revenue million Forecast, by Region 2020 & 2033
- Table 6: Global circRNA-LNP Formulation Volume K Forecast, by Region 2020 & 2033
- Table 7: Global circRNA-LNP Formulation Revenue million Forecast, by Application 2020 & 2033
- Table 8: Global circRNA-LNP Formulation Volume K Forecast, by Application 2020 & 2033
- Table 9: Global circRNA-LNP Formulation Revenue million Forecast, by Types 2020 & 2033
- Table 10: Global circRNA-LNP Formulation Volume K Forecast, by Types 2020 & 2033
- Table 11: Global circRNA-LNP Formulation Revenue million Forecast, by Country 2020 & 2033
- Table 12: Global circRNA-LNP Formulation Volume K Forecast, by Country 2020 & 2033
- Table 13: United States circRNA-LNP Formulation Revenue (million) Forecast, by Application 2020 & 2033
- Table 14: United States circRNA-LNP Formulation Volume (K) Forecast, by Application 2020 & 2033
- Table 15: Canada circRNA-LNP Formulation Revenue (million) Forecast, by Application 2020 & 2033
- Table 16: Canada circRNA-LNP Formulation Volume (K) Forecast, by Application 2020 & 2033
- Table 17: Mexico circRNA-LNP Formulation Revenue (million) Forecast, by Application 2020 & 2033
- Table 18: Mexico circRNA-LNP Formulation Volume (K) Forecast, by Application 2020 & 2033
- Table 19: Global circRNA-LNP Formulation Revenue million Forecast, by Application 2020 & 2033
- Table 20: Global circRNA-LNP Formulation Volume K Forecast, by Application 2020 & 2033
- Table 21: Global circRNA-LNP Formulation Revenue million Forecast, by Types 2020 & 2033
- Table 22: Global circRNA-LNP Formulation Volume K Forecast, by Types 2020 & 2033
- Table 23: Global circRNA-LNP Formulation Revenue million Forecast, by Country 2020 & 2033
- Table 24: Global circRNA-LNP Formulation Volume K Forecast, by Country 2020 & 2033
- Table 25: Brazil circRNA-LNP Formulation Revenue (million) Forecast, by Application 2020 & 2033
- Table 26: Brazil circRNA-LNP Formulation Volume (K) Forecast, by Application 2020 & 2033
- Table 27: Argentina circRNA-LNP Formulation Revenue (million) Forecast, by Application 2020 & 2033
- Table 28: Argentina circRNA-LNP Formulation Volume (K) Forecast, by Application 2020 & 2033
- Table 29: Rest of South America circRNA-LNP Formulation Revenue (million) Forecast, by Application 2020 & 2033
- Table 30: Rest of South America circRNA-LNP Formulation Volume (K) Forecast, by Application 2020 & 2033
- Table 31: Global circRNA-LNP Formulation Revenue million Forecast, by Application 2020 & 2033
- Table 32: Global circRNA-LNP Formulation Volume K Forecast, by Application 2020 & 2033
- Table 33: Global circRNA-LNP Formulation Revenue million Forecast, by Types 2020 & 2033
- Table 34: Global circRNA-LNP Formulation Volume K Forecast, by Types 2020 & 2033
- Table 35: Global circRNA-LNP Formulation Revenue million Forecast, by Country 2020 & 2033
- Table 36: Global circRNA-LNP Formulation Volume K Forecast, by Country 2020 & 2033
- Table 37: United Kingdom circRNA-LNP Formulation Revenue (million) Forecast, by Application 2020 & 2033
- Table 38: United Kingdom circRNA-LNP Formulation Volume (K) Forecast, by Application 2020 & 2033
- Table 39: Germany circRNA-LNP Formulation Revenue (million) Forecast, by Application 2020 & 2033
- Table 40: Germany circRNA-LNP Formulation Volume (K) Forecast, by Application 2020 & 2033
- Table 41: France circRNA-LNP Formulation Revenue (million) Forecast, by Application 2020 & 2033
- Table 42: France circRNA-LNP Formulation Volume (K) Forecast, by Application 2020 & 2033
- Table 43: Italy circRNA-LNP Formulation Revenue (million) Forecast, by Application 2020 & 2033
- Table 44: Italy circRNA-LNP Formulation Volume (K) Forecast, by Application 2020 & 2033
- Table 45: Spain circRNA-LNP Formulation Revenue (million) Forecast, by Application 2020 & 2033
- Table 46: Spain circRNA-LNP Formulation Volume (K) Forecast, by Application 2020 & 2033
- Table 47: Russia circRNA-LNP Formulation Revenue (million) Forecast, by Application 2020 & 2033
- Table 48: Russia circRNA-LNP Formulation Volume (K) Forecast, by Application 2020 & 2033
- Table 49: Benelux circRNA-LNP Formulation Revenue (million) Forecast, by Application 2020 & 2033
- Table 50: Benelux circRNA-LNP Formulation Volume (K) Forecast, by Application 2020 & 2033
- Table 51: Nordics circRNA-LNP Formulation Revenue (million) Forecast, by Application 2020 & 2033
- Table 52: Nordics circRNA-LNP Formulation Volume (K) Forecast, by Application 2020 & 2033
- Table 53: Rest of Europe circRNA-LNP Formulation Revenue (million) Forecast, by Application 2020 & 2033
- Table 54: Rest of Europe circRNA-LNP Formulation Volume (K) Forecast, by Application 2020 & 2033
- Table 55: Global circRNA-LNP Formulation Revenue million Forecast, by Application 2020 & 2033
- Table 56: Global circRNA-LNP Formulation Volume K Forecast, by Application 2020 & 2033
- Table 57: Global circRNA-LNP Formulation Revenue million Forecast, by Types 2020 & 2033
- Table 58: Global circRNA-LNP Formulation Volume K Forecast, by Types 2020 & 2033
- Table 59: Global circRNA-LNP Formulation Revenue million Forecast, by Country 2020 & 2033
- Table 60: Global circRNA-LNP Formulation Volume K Forecast, by Country 2020 & 2033
- Table 61: Turkey circRNA-LNP Formulation Revenue (million) Forecast, by Application 2020 & 2033
- Table 62: Turkey circRNA-LNP Formulation Volume (K) Forecast, by Application 2020 & 2033
- Table 63: Israel circRNA-LNP Formulation Revenue (million) Forecast, by Application 2020 & 2033
- Table 64: Israel circRNA-LNP Formulation Volume (K) Forecast, by Application 2020 & 2033
- Table 65: GCC circRNA-LNP Formulation Revenue (million) Forecast, by Application 2020 & 2033
- Table 66: GCC circRNA-LNP Formulation Volume (K) Forecast, by Application 2020 & 2033
- Table 67: North Africa circRNA-LNP Formulation Revenue (million) Forecast, by Application 2020 & 2033
- Table 68: North Africa circRNA-LNP Formulation Volume (K) Forecast, by Application 2020 & 2033
- Table 69: South Africa circRNA-LNP Formulation Revenue (million) Forecast, by Application 2020 & 2033
- Table 70: South Africa circRNA-LNP Formulation Volume (K) Forecast, by Application 2020 & 2033
- Table 71: Rest of Middle East & Africa circRNA-LNP Formulation Revenue (million) Forecast, by Application 2020 & 2033
- Table 72: Rest of Middle East & Africa circRNA-LNP Formulation Volume (K) Forecast, by Application 2020 & 2033
- Table 73: Global circRNA-LNP Formulation Revenue million Forecast, by Application 2020 & 2033
- Table 74: Global circRNA-LNP Formulation Volume K Forecast, by Application 2020 & 2033
- Table 75: Global circRNA-LNP Formulation Revenue million Forecast, by Types 2020 & 2033
- Table 76: Global circRNA-LNP Formulation Volume K Forecast, by Types 2020 & 2033
- Table 77: Global circRNA-LNP Formulation Revenue million Forecast, by Country 2020 & 2033
- Table 78: Global circRNA-LNP Formulation Volume K Forecast, by Country 2020 & 2033
- Table 79: China circRNA-LNP Formulation Revenue (million) Forecast, by Application 2020 & 2033
- Table 80: China circRNA-LNP Formulation Volume (K) Forecast, by Application 2020 & 2033
- Table 81: India circRNA-LNP Formulation Revenue (million) Forecast, by Application 2020 & 2033
- Table 82: India circRNA-LNP Formulation Volume (K) Forecast, by Application 2020 & 2033
- Table 83: Japan circRNA-LNP Formulation Revenue (million) Forecast, by Application 2020 & 2033
- Table 84: Japan circRNA-LNP Formulation Volume (K) Forecast, by Application 2020 & 2033
- Table 85: South Korea circRNA-LNP Formulation Revenue (million) Forecast, by Application 2020 & 2033
- Table 86: South Korea circRNA-LNP Formulation Volume (K) Forecast, by Application 2020 & 2033
- Table 87: ASEAN circRNA-LNP Formulation Revenue (million) Forecast, by Application 2020 & 2033
- Table 88: ASEAN circRNA-LNP Formulation Volume (K) Forecast, by Application 2020 & 2033
- Table 89: Oceania circRNA-LNP Formulation Revenue (million) Forecast, by Application 2020 & 2033
- Table 90: Oceania circRNA-LNP Formulation Volume (K) Forecast, by Application 2020 & 2033
- Table 91: Rest of Asia Pacific circRNA-LNP Formulation Revenue (million) Forecast, by Application 2020 & 2033
- Table 92: Rest of Asia Pacific circRNA-LNP Formulation Volume (K) Forecast, by Application 2020 & 2033
Frequently Asked Questions
1. What is the projected Compound Annual Growth Rate (CAGR) of the circRNA-LNP Formulation?
The projected CAGR is approximately 5.67%.
2. Which companies are prominent players in the circRNA-LNP Formulation?
Key companies in the market include Orna Therapeutics, GenScript, Creative Biogene, Catalent, Creative Biolabs, VectorBuilder, Guangzhou Geneseed Biotech. Co., Ltd., Porton Pharma Solutions, Ltd, Recipharm AB, Precigenome LLC.
3. What are the main segments of the circRNA-LNP Formulation?
The market segments include Application, Types.
4. Can you provide details about the market size?
The market size is estimated to be USD 300.72 million as of 2022.
5. What are some drivers contributing to market growth?
N/A
6. What are the notable trends driving market growth?
N/A
7. Are there any restraints impacting market growth?
N/A
8. Can you provide examples of recent developments in the market?
N/A
9. What pricing options are available for accessing the report?
Pricing options include single-user, multi-user, and enterprise licenses priced at USD 3950.00, USD 5925.00, and USD 7900.00 respectively.
10. Is the market size provided in terms of value or volume?
The market size is provided in terms of value, measured in million and volume, measured in K.
11. Are there any specific market keywords associated with the report?
Yes, the market keyword associated with the report is "circRNA-LNP Formulation," which aids in identifying and referencing the specific market segment covered.
12. How do I determine which pricing option suits my needs best?
The pricing options vary based on user requirements and access needs. Individual users may opt for single-user licenses, while businesses requiring broader access may choose multi-user or enterprise licenses for cost-effective access to the report.
13. Are there any additional resources or data provided in the circRNA-LNP Formulation report?
While the report offers comprehensive insights, it's advisable to review the specific contents or supplementary materials provided to ascertain if additional resources or data are available.
14. How can I stay updated on further developments or reports in the circRNA-LNP Formulation?
To stay informed about further developments, trends, and reports in the circRNA-LNP Formulation, consider subscribing to industry newsletters, following relevant companies and organizations, or regularly checking reputable industry news sources and publications.
Methodology
Step 1 - Identification of Relevant Samples Size from Population Database



Step 2 - Approaches for Defining Global Market Size (Value, Volume* & Price*)

Note*: In applicable scenarios
Step 3 - Data Sources
Primary Research
- Web Analytics
- Survey Reports
- Research Institute
- Latest Research Reports
- Opinion Leaders
Secondary Research
- Annual Reports
- White Paper
- Latest Press Release
- Industry Association
- Paid Database
- Investor Presentations

Step 4 - Data Triangulation
Involves using different sources of information in order to increase the validity of a study
These sources are likely to be stakeholders in a program - participants, other researchers, program staff, other community members, and so on.
Then we put all data in single framework & apply various statistical tools to find out the dynamic on the market.
During the analysis stage, feedback from the stakeholder groups would be compared to determine areas of agreement as well as areas of divergence


