Key Insights
The In-Vitro Toxicology Testing Industry is experiencing robust growth, driven by an increasing emphasis on ethical and efficient drug development and chemical safety assessment. With a projected market size of $14.95 billion in 2025, the industry is poised for significant expansion, fueled by a compound annual growth rate (CAGR) of 10.1% over the forecast period. This upward trajectory is largely attributed to the growing adoption of advanced technologies such as cell culture, high-throughput screening, molecular imaging, and OMICS, which offer more predictive and human-relevant results compared to traditional animal testing. Furthermore, regulatory pressures worldwide are increasingly favoring non-animal testing methods, creating a substantial tailwind for the in-vitro toxicology market. The demand for cellular assays and biochemical assays, alongside emerging in-silico and ex-vivo methodologies, highlights a dynamic shift towards precision and efficiency in toxicological evaluations across various applications, including systemic, dermal, endocrine disruption, and ocular toxicity.
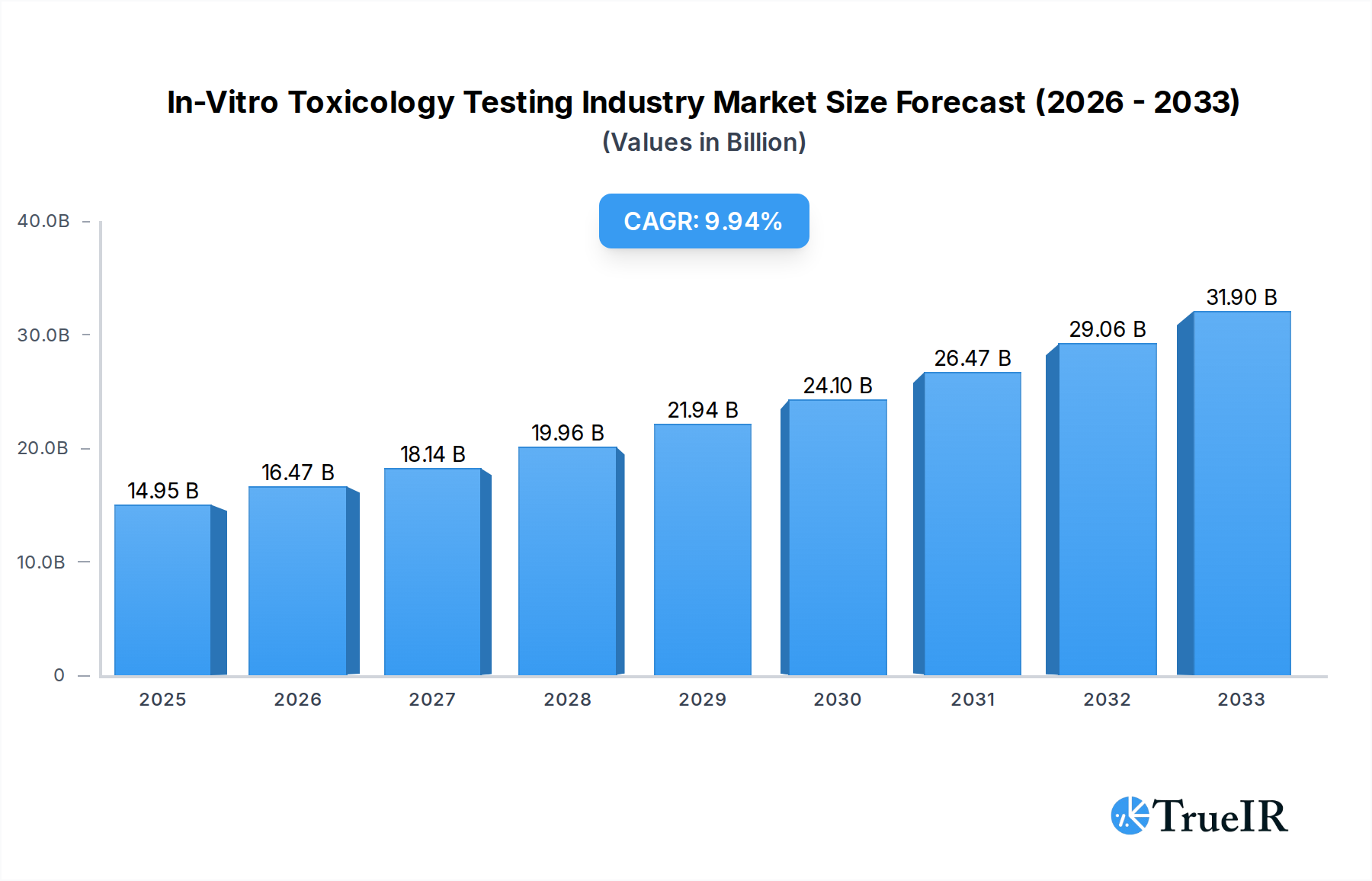
In-Vitro Toxicology Testing Industry Market Size (In Billion)

The market's expansion is further propelled by the pharmaceutical and biotechnology sectors' relentless pursuit of novel therapeutics and the diagnostics industry's need for accurate safety profiling. While the market is dominated by established players like Thermo Fisher Scientific, Merck KGaA, and Eurofins Scientific, the competitive landscape is also characterized by innovation and strategic collaborations. Restraints, such as the initial high cost of advanced technology implementation and the need for standardization across different testing platforms, are being addressed through ongoing research and development. Geographically, North America and Europe currently lead the market due to stringent regulatory frameworks and substantial R&D investments, with the Asia Pacific region emerging as a rapidly growing segment driven by increasing awareness and investment in advanced testing capabilities. This sustained growth underscores the indispensable role of in-vitro toxicology in ensuring product safety and accelerating innovation in life sciences.
In-Vitro Toxicology Testing Industry Company Market Share

This comprehensive report delves into the dynamic In-Vitro Toxicology Testing Market, a critical sector projected to reach billions in value. Leveraging advanced technologies like Cell Culture, High Throughput, Molecular Imaging, OMICS, and employing methods such as Cellular Assay, Biochemical Assay, In Silica, and Ex-vivo, the market is essential for assessing Systemic Toxicology, Dermal Toxicity, Endocrine Disruption, Ocular Toxicity, and other vital applications. The primary end-users are Pharmaceutical and Biotechnology companies, alongside Diagnostics providers. The study period spans 2019–2033, with a base year of 2025, an estimated year of 2025, and a forecast period from 2025–2033, building upon the historical data from 2019–2024.
In-Vitro Toxicology Testing Industry Market Structure & Competitive Landscape
The in-vitro toxicology testing industry exhibits a moderately concentrated market structure, driven by significant investments in research and development and stringent regulatory requirements that favor established players. Innovation is a paramount driver, with companies continuously developing novel assay technologies and automation solutions to enhance accuracy, speed, and reduce animal testing. Regulatory landscapes, particularly from bodies like the FDA and EMA, significantly influence market dynamics by mandating specific testing protocols and promoting the adoption of validated in-vitro methods. Product substitutes, such as traditional animal testing, are gradually being phased out due to ethical concerns and cost-effectiveness of in-vitro alternatives. End-user segmentation highlights the dominance of the pharmaceutical and biotechnology sector, which accounts for the largest share of demand, followed by the diagnostics sector. Merger and acquisition (M&A) activities are prominent, with an estimated xx billion in M&A volumes over the historical period, reflecting a consolidation trend as larger companies acquire smaller, innovative firms to expand their service portfolios and market reach. Key companies actively shaping this landscape include Covance, Promega Corporation, Merck KGaA, Eurofins Scientific, GE Healthcare, Abbott Laboratories, Quest Diagnostics, Thermo Fisher Scientific, Bio-Rad Laboratories, and Agilent Technologies, among others.
In-Vitro Toxicology Testing Industry Market Trends & Opportunities
The in-vitro toxicology testing market is experiencing robust growth, with an estimated market size projected to exceed billions by 2033, driven by a compound annual growth rate (CAGR) of approximately xx%. This expansion is fueled by an increasing demand for safer and more effective pharmaceuticals and chemicals, coupled with a global regulatory push towards reducing animal testing. Technological advancements are at the forefront of this evolution. The integration of OMICS technologies, such as genomics, proteomics, and metabolomics, is revolutionizing the ability to understand complex toxicological mechanisms at a molecular level. Similarly, High Throughput Screening (HTS) platforms, powered by advanced automation and robotics, are enabling faster and more cost-effective screening of vast numbers of compounds. The rise of Molecular Imaging in in-vitro settings allows for real-time visualization of cellular and tissue responses to toxic substances, providing deeper insights.
Consumer preferences are increasingly aligning with ethical product development, further accelerating the adoption of non-animal testing methods. Regulatory bodies worldwide are actively promoting and validating in-vitro alternatives, creating a favorable environment for market expansion. The growing emphasis on personalized medicine also necessitates sophisticated toxicological assessments of individual drug responses, a field where in-vitro models excel. The competitive landscape is dynamic, characterized by strategic collaborations, mergers, and acquisitions as companies seek to enhance their capabilities and market share. The increasing penetration of these advanced testing methods across various industries, from cosmetics to agrochemicals, signifies a broadening market base. Opportunities abound for companies that can offer integrated solutions, encompassing a wide range of in-vitro technologies and expertise, thereby addressing the multifaceted toxicological needs of their clients. The development of predictive toxicology models, leveraging artificial intelligence and machine learning, presents a significant future growth avenue, promising even greater accuracy and efficiency in hazard assessment.
Dominant Markets & Segments in In-Vitro Toxicology Testing Industry
The in-vitro toxicology testing industry is witnessing significant dominance from specific regions and segments. Geographically, North America and Europe are leading markets, driven by well-established pharmaceutical and biotechnology industries, stringent regulatory frameworks that mandate advanced safety testing, and substantial R&D investments. The Asia-Pacific region is emerging as a rapidly growing market, fueled by increasing investments in life sciences, a growing generics market, and the expansion of contract research organizations (CROs).
Within the Technology segment, Cell Culture remains the foundational technology, underpinning many in-vitro assays. However, High Throughput screening technologies are experiencing exponential growth due to their efficiency in testing large compound libraries. OMICS technologies are gaining significant traction for their ability to provide deep mechanistic insights into toxicity.
In terms of Method, Cellular Assays are the most widely adopted, offering a direct assessment of cellular responses. Biochemical Assays are crucial for evaluating enzyme activity and receptor binding. In Silica (computational toxicology) methods are increasingly integrated to predict potential toxicity early in the drug discovery pipeline, complementing experimental approaches. Ex-vivo methods, utilizing tissue explants, provide a more complex biological context for testing.
The Application segment is dominated by Systemic Toxicology, as it is essential for understanding the effects of substances on the entire body. Dermal Toxicity testing is also a major segment, particularly for consumer products and pharmaceuticals applied to the skin. Endocrine Disruption testing is gaining prominence due to increasing regulatory scrutiny and public concern.
The End User segment is overwhelmingly driven by the Pharmaceutical and Biotechnology industry, which constitutes the largest share of the market. This is due to the extensive preclinical and clinical testing requirements for drug development. The Diagnostics sector is also a significant user, employing in-vitro toxicology for various diagnostic applications and safety assessments.
Key growth drivers within these segments include:
- Infrastructure: Development of state-of-the-art laboratories equipped with advanced HTS platforms and OMICS capabilities.
- Policies: Favorable government regulations promoting the adoption of in-vitro methods and phasing out animal testing, along with increased funding for toxicology research.
- Technological Advancements: Continuous innovation in assay development, automation, and data analysis tools.
- Industry Demand: Growing demand for novel therapeutics, safer chemicals, and reliable diagnostic tools.
In-Vitro Toxicology Testing Industry Product Analysis
In-vitro toxicology testing products are characterized by rapid technological advancements and a focus on enhanced predictive accuracy and efficiency. Innovations include sophisticated 3D cell culture models and organ-on-a-chip technologies that better mimic human physiology compared to traditional 2D cultures. High-throughput screening platforms, coupled with advanced robotics and AI-driven data analysis, are enabling faster and more cost-effective evaluation of drug candidates and chemicals. The integration of omics technologies provides deeper mechanistic insights into toxicological pathways. These products offer significant competitive advantages by reducing reliance on animal testing, accelerating development timelines, and improving the safety assessment of new products.
Key Drivers, Barriers & Challenges in In-Vitro Toxicology Testing Industry
Key Drivers: The primary forces propelling the in-vitro toxicology testing market include stringent regulatory mandates pushing for the reduction and replacement of animal testing, significant advancements in assay technologies offering greater accuracy and efficiency, and the increasing demand from the pharmaceutical and biotechnology sectors for faster drug development cycles. Economic drivers include the cost-effectiveness of in-vitro methods compared to traditional animal studies, and policy-driven support for ethical research practices.
Barriers & Challenges: Key challenges impacting growth include the need for further validation of certain complex in-vitro models by regulatory bodies, the high initial investment required for advanced instrumentation and expertise, and the complexity of interpreting in-vitro data in the context of human response for certain toxicological endpoints. Supply chain issues for specialized reagents and consumables, and the ongoing competitive pressure to constantly innovate and demonstrate superiority over existing methods, also pose significant hurdles.
Growth Drivers in the In-Vitro Toxicology Testing Industry Market
Growth in the in-vitro toxicology testing industry is significantly propelled by several factors. Technological innovation is a primary driver, with the development of novel assay platforms, organ-on-a-chip technologies, and advanced OMICS integration offering enhanced predictive capabilities. Economic factors, such as the long-term cost-effectiveness of in-vitro methods compared to animal testing and the increasing R&D budgets of pharmaceutical and biotechnology companies, also fuel growth. Regulatory imperatives, particularly the global push to reduce and replace animal testing, are creating a strong demand for validated in-vitro alternatives.
Challenges Impacting In-Vitro Toxicology Testing Industry Growth
Several challenges impede the full growth potential of the in-vitro toxicology testing industry. Regulatory complexities remain a significant hurdle, as the validation and acceptance of new in-vitro methods can be a lengthy and intricate process for certain toxicological endpoints. Supply chain disruptions for specialized reagents and cell culture materials can impact the continuity of research and testing. Intense competitive pressures necessitate continuous innovation and demonstration of superior performance, while the high initial investment for cutting-edge technologies can be a barrier for smaller organizations.
Key Players Shaping the In-Vitro Toxicology Testing Industry Market
- Covance
- Promega Corporation
- Merck KGaA
- Eurofins Scientific
- GE Healthcare
- Abbott Laboratories
- Quest Diagnostics
- Thermo Fisher Scientific
- Bio-Rad Laboratories
- Agilent Technologies
Significant In-Vitro Toxicology Testing Industry Industry Milestones
- 2019: Increased regulatory acceptance of specific cellular assay methods for carcinogenicity testing.
- 2020: Launch of advanced organ-on-a-chip models with enhanced complexity for drug metabolism studies.
- 2021: Significant investments in AI and machine learning for predictive toxicology model development.
- 2022: Expansion of high-throughput screening capabilities in emerging markets.
- 2023: Mergers and acquisitions focused on integrating OMICS technologies into existing toxicology platforms.
- 2024: Enhanced focus on developing in-vitro models for complex neurological and immunological toxicities.
Future Outlook for In-Vitro Toxicology Testing Industry Market
The future outlook for the in-vitro toxicology testing market is exceptionally promising, driven by an accelerating trend towards non-animal testing and the increasing sophistication of scientific methodologies. Strategic opportunities lie in the continued development and regulatory acceptance of advanced 3D cell cultures, organoids, and organ-on-a-chip systems that offer more physiologically relevant data. The integration of artificial intelligence and machine learning will further enhance predictive toxicology, enabling earlier identification of potential safety concerns. The market potential will be amplified by expanding applications beyond pharmaceuticals, including consumer products, environmental safety, and advanced materials testing, all contributing to a projected market value in the billions.
In-Vitro Toxicology Testing Industry Segmentation
-
1. Technology
- 1.1. Cell Culture
- 1.2. High Throughput
- 1.3. Molecular Imaging
- 1.4. OMICS
-
2. Method
- 2.1. Cellular Assay
- 2.2. Biochemical Assay
- 2.3. In Silica
- 2.4. Ex-vivo
-
3. Application
- 3.1. Systemic Toxicology
- 3.2. Dermal Toxicity
- 3.3. Endorine Disruption
- 3.4. Occular Toxicity
- 3.5. Other Applications
-
4. End User
- 4.1. Pharmaceutical and Biotechnology
- 4.2. Diagnostics
- 4.3. Other End User
In-Vitro Toxicology Testing Industry Segmentation By Geography
-
1. North America
- 1.1. United States
- 1.2. Canada
- 1.3. Mexico
-
2. Europe
- 2.1. Germany
- 2.2. United Kingdom
- 2.3. France
- 2.4. Italy
- 2.5. Spain
- 2.6. Rest of Europe
-
3. Asia Pacific
- 3.1. China
- 3.2. Japan
- 3.3. India
- 3.4. Australia
- 3.5. South Korea
- 3.6. Rest of Asia Pacific
- 4. Middle East
-
5. GCC
- 5.1. South Africa
- 5.2. Rest of Middle East
-
6. South America
- 6.1. Brazil
- 6.2. Argentina
- 6.3. Rest of South America
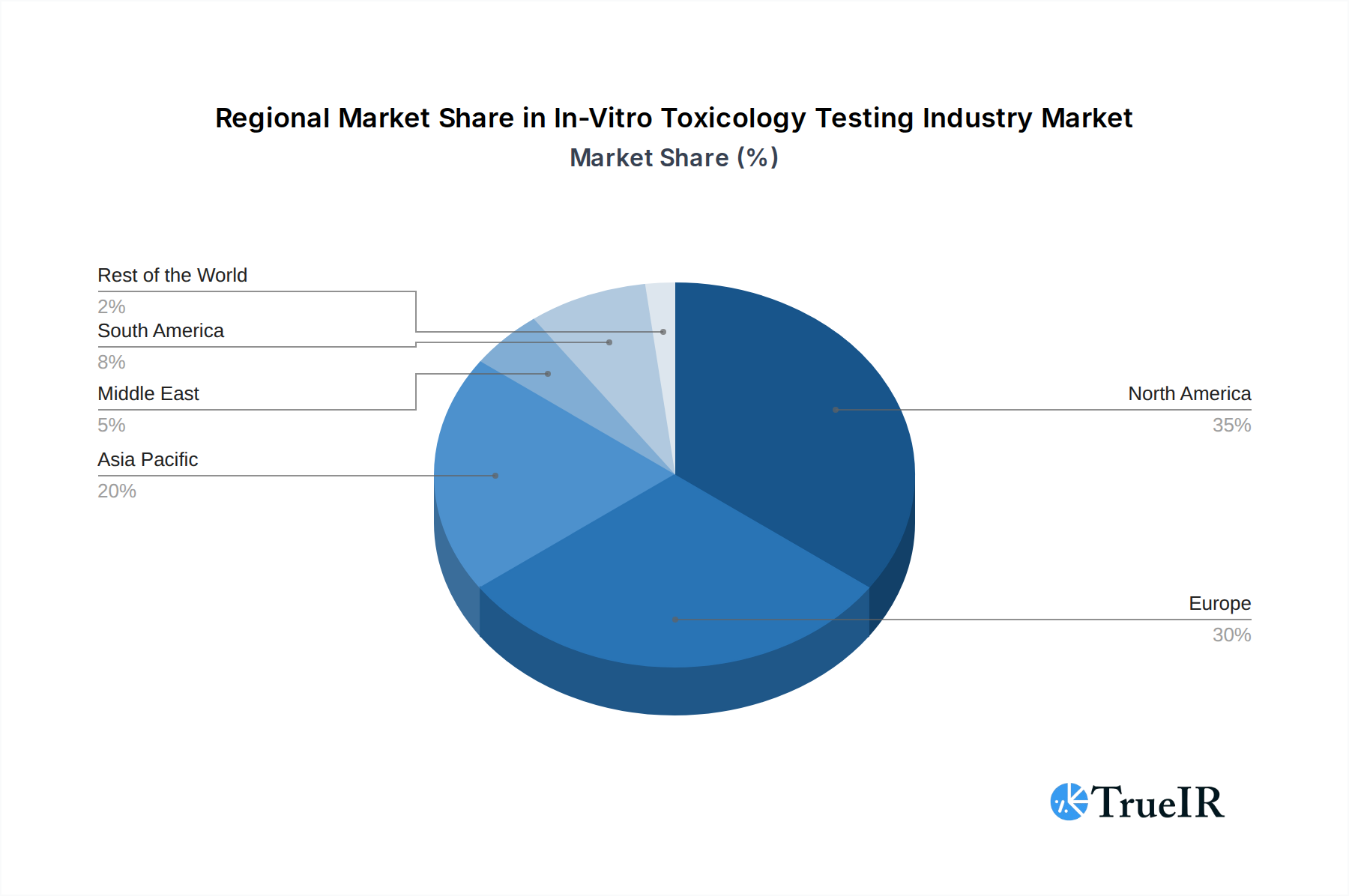
In-Vitro Toxicology Testing Industry Regional Market Share

Geographic Coverage of In-Vitro Toxicology Testing Industry
In-Vitro Toxicology Testing Industry REPORT HIGHLIGHTS
| Aspects | Details |
|---|---|
| Study Period | 2020-2034 |
| Base Year | 2025 |
| Estimated Year | 2026 |
| Forecast Period | 2026-2034 |
| Historical Period | 2020-2025 |
| Growth Rate | CAGR of 10.1% from 2020-2034 |
| Segmentation |
|
Table of Contents
- 1. Introduction
- 1.1. Research Scope
- 1.2. Market Segmentation
- 1.3. Research Objective
- 1.4. Definitions and Assumptions
- 2. Executive Summary
- 2.1. Market Snapshot
- 3. Market Dynamics
- 3.1. Market Drivers
- 3.2. Market Restrains
- 3.3. Market Trends
- 3.4. Market Opportunities
- 4. Market Factor Analysis
- 4.1. Porters Five Forces
- 4.1.1. Bargaining Power of Suppliers
- 4.1.2. Bargaining Power of Buyers
- 4.1.3. Threat of New Entrants
- 4.1.4. Threat of Substitutes
- 4.1.5. Competitive Rivalry
- 4.2. PESTEL analysis
- 4.3. BCG Analysis
- 4.3.1. Stars (High Growth, High Market Share)
- 4.3.2. Cash Cows (Low Growth, High Market Share)
- 4.3.3. Question Mark (High Growth, Low Market Share)
- 4.3.4. Dogs (Low Growth, Low Market Share)
- 4.4. Ansoff Matrix Analysis
- 4.5. Supply Chain Analysis
- 4.6. Regulatory Landscape
- 4.7. Current Market Potential and Opportunity Assessment (TAM–SAM–SOM Framework)
- 4.8. TIR Analyst Note
- 4.1. Porters Five Forces
- 5. Market Analysis, Insights and Forecast 2021-2033
- 5.1. Market Analysis, Insights and Forecast - by Technology
- 5.1.1. Cell Culture
- 5.1.2. High Throughput
- 5.1.3. Molecular Imaging
- 5.1.4. OMICS
- 5.2. Market Analysis, Insights and Forecast - by Method
- 5.2.1. Cellular Assay
- 5.2.2. Biochemical Assay
- 5.2.3. In Silica
- 5.2.4. Ex-vivo
- 5.3. Market Analysis, Insights and Forecast - by Application
- 5.3.1. Systemic Toxicology
- 5.3.2. Dermal Toxicity
- 5.3.3. Endorine Disruption
- 5.3.4. Occular Toxicity
- 5.3.5. Other Applications
- 5.4. Market Analysis, Insights and Forecast - by End User
- 5.4.1. Pharmaceutical and Biotechnology
- 5.4.2. Diagnostics
- 5.4.3. Other End User
- 5.5. Market Analysis, Insights and Forecast - by Region
- 5.5.1. North America
- 5.5.2. Europe
- 5.5.3. Asia Pacific
- 5.5.4. Middle East
- 5.5.5. GCC
- 5.5.6. South America
- 5.1. Market Analysis, Insights and Forecast - by Technology
- 6. Global In-Vitro Toxicology Testing Industry Analysis, Insights and Forecast, 2021-2033
- 6.1. Market Analysis, Insights and Forecast - by Technology
- 6.1.1. Cell Culture
- 6.1.2. High Throughput
- 6.1.3. Molecular Imaging
- 6.1.4. OMICS
- 6.2. Market Analysis, Insights and Forecast - by Method
- 6.2.1. Cellular Assay
- 6.2.2. Biochemical Assay
- 6.2.3. In Silica
- 6.2.4. Ex-vivo
- 6.3. Market Analysis, Insights and Forecast - by Application
- 6.3.1. Systemic Toxicology
- 6.3.2. Dermal Toxicity
- 6.3.3. Endorine Disruption
- 6.3.4. Occular Toxicity
- 6.3.5. Other Applications
- 6.4. Market Analysis, Insights and Forecast - by End User
- 6.4.1. Pharmaceutical and Biotechnology
- 6.4.2. Diagnostics
- 6.4.3. Other End User
- 6.1. Market Analysis, Insights and Forecast - by Technology
- 7. North America In-Vitro Toxicology Testing Industry Analysis, Insights and Forecast, 2020-2032
- 7.1. Market Analysis, Insights and Forecast - by Technology
- 7.1.1. Cell Culture
- 7.1.2. High Throughput
- 7.1.3. Molecular Imaging
- 7.1.4. OMICS
- 7.2. Market Analysis, Insights and Forecast - by Method
- 7.2.1. Cellular Assay
- 7.2.2. Biochemical Assay
- 7.2.3. In Silica
- 7.2.4. Ex-vivo
- 7.3. Market Analysis, Insights and Forecast - by Application
- 7.3.1. Systemic Toxicology
- 7.3.2. Dermal Toxicity
- 7.3.3. Endorine Disruption
- 7.3.4. Occular Toxicity
- 7.3.5. Other Applications
- 7.4. Market Analysis, Insights and Forecast - by End User
- 7.4.1. Pharmaceutical and Biotechnology
- 7.4.2. Diagnostics
- 7.4.3. Other End User
- 7.1. Market Analysis, Insights and Forecast - by Technology
- 8. Europe In-Vitro Toxicology Testing Industry Analysis, Insights and Forecast, 2020-2032
- 8.1. Market Analysis, Insights and Forecast - by Technology
- 8.1.1. Cell Culture
- 8.1.2. High Throughput
- 8.1.3. Molecular Imaging
- 8.1.4. OMICS
- 8.2. Market Analysis, Insights and Forecast - by Method
- 8.2.1. Cellular Assay
- 8.2.2. Biochemical Assay
- 8.2.3. In Silica
- 8.2.4. Ex-vivo
- 8.3. Market Analysis, Insights and Forecast - by Application
- 8.3.1. Systemic Toxicology
- 8.3.2. Dermal Toxicity
- 8.3.3. Endorine Disruption
- 8.3.4. Occular Toxicity
- 8.3.5. Other Applications
- 8.4. Market Analysis, Insights and Forecast - by End User
- 8.4.1. Pharmaceutical and Biotechnology
- 8.4.2. Diagnostics
- 8.4.3. Other End User
- 8.1. Market Analysis, Insights and Forecast - by Technology
- 9. Asia Pacific In-Vitro Toxicology Testing Industry Analysis, Insights and Forecast, 2020-2032
- 9.1. Market Analysis, Insights and Forecast - by Technology
- 9.1.1. Cell Culture
- 9.1.2. High Throughput
- 9.1.3. Molecular Imaging
- 9.1.4. OMICS
- 9.2. Market Analysis, Insights and Forecast - by Method
- 9.2.1. Cellular Assay
- 9.2.2. Biochemical Assay
- 9.2.3. In Silica
- 9.2.4. Ex-vivo
- 9.3. Market Analysis, Insights and Forecast - by Application
- 9.3.1. Systemic Toxicology
- 9.3.2. Dermal Toxicity
- 9.3.3. Endorine Disruption
- 9.3.4. Occular Toxicity
- 9.3.5. Other Applications
- 9.4. Market Analysis, Insights and Forecast - by End User
- 9.4.1. Pharmaceutical and Biotechnology
- 9.4.2. Diagnostics
- 9.4.3. Other End User
- 9.1. Market Analysis, Insights and Forecast - by Technology
- 10. Middle East In-Vitro Toxicology Testing Industry Analysis, Insights and Forecast, 2020-2032
- 10.1. Market Analysis, Insights and Forecast - by Technology
- 10.1.1. Cell Culture
- 10.1.2. High Throughput
- 10.1.3. Molecular Imaging
- 10.1.4. OMICS
- 10.2. Market Analysis, Insights and Forecast - by Method
- 10.2.1. Cellular Assay
- 10.2.2. Biochemical Assay
- 10.2.3. In Silica
- 10.2.4. Ex-vivo
- 10.3. Market Analysis, Insights and Forecast - by Application
- 10.3.1. Systemic Toxicology
- 10.3.2. Dermal Toxicity
- 10.3.3. Endorine Disruption
- 10.3.4. Occular Toxicity
- 10.3.5. Other Applications
- 10.4. Market Analysis, Insights and Forecast - by End User
- 10.4.1. Pharmaceutical and Biotechnology
- 10.4.2. Diagnostics
- 10.4.3. Other End User
- 10.1. Market Analysis, Insights and Forecast - by Technology
- 11. GCC In-Vitro Toxicology Testing Industry Analysis, Insights and Forecast, 2020-2032
- 11.1. Market Analysis, Insights and Forecast - by Technology
- 11.1.1. Cell Culture
- 11.1.2. High Throughput
- 11.1.3. Molecular Imaging
- 11.1.4. OMICS
- 11.2. Market Analysis, Insights and Forecast - by Method
- 11.2.1. Cellular Assay
- 11.2.2. Biochemical Assay
- 11.2.3. In Silica
- 11.2.4. Ex-vivo
- 11.3. Market Analysis, Insights and Forecast - by Application
- 11.3.1. Systemic Toxicology
- 11.3.2. Dermal Toxicity
- 11.3.3. Endorine Disruption
- 11.3.4. Occular Toxicity
- 11.3.5. Other Applications
- 11.4. Market Analysis, Insights and Forecast - by End User
- 11.4.1. Pharmaceutical and Biotechnology
- 11.4.2. Diagnostics
- 11.4.3. Other End User
- 11.1. Market Analysis, Insights and Forecast - by Technology
- 12. South America In-Vitro Toxicology Testing Industry Analysis, Insights and Forecast, 2020-2032
- 12.1. Market Analysis, Insights and Forecast - by Technology
- 12.1.1. Cell Culture
- 12.1.2. High Throughput
- 12.1.3. Molecular Imaging
- 12.1.4. OMICS
- 12.2. Market Analysis, Insights and Forecast - by Method
- 12.2.1. Cellular Assay
- 12.2.2. Biochemical Assay
- 12.2.3. In Silica
- 12.2.4. Ex-vivo
- 12.3. Market Analysis, Insights and Forecast - by Application
- 12.3.1. Systemic Toxicology
- 12.3.2. Dermal Toxicity
- 12.3.3. Endorine Disruption
- 12.3.4. Occular Toxicity
- 12.3.5. Other Applications
- 12.4. Market Analysis, Insights and Forecast - by End User
- 12.4.1. Pharmaceutical and Biotechnology
- 12.4.2. Diagnostics
- 12.4.3. Other End User
- 12.1. Market Analysis, Insights and Forecast - by Technology
- 13. Competitive Analysis
- 13.1. Company Profiles
- 13.1.1 Covance
- 13.1.1.1. Company Overview
- 13.1.1.2. Products
- 13.1.1.3. Company Financials
- 13.1.1.4. SWOT Analysis
- 13.1.2 Promega Corporation
- 13.1.2.1. Company Overview
- 13.1.2.2. Products
- 13.1.2.3. Company Financials
- 13.1.2.4. SWOT Analysis
- 13.1.3 Merck KGaA
- 13.1.3.1. Company Overview
- 13.1.3.2. Products
- 13.1.3.3. Company Financials
- 13.1.3.4. SWOT Analysis
- 13.1.4 Eurofins Scientific
- 13.1.4.1. Company Overview
- 13.1.4.2. Products
- 13.1.4.3. Company Financials
- 13.1.4.4. SWOT Analysis
- 13.1.5 GE Healthcare
- 13.1.5.1. Company Overview
- 13.1.5.2. Products
- 13.1.5.3. Company Financials
- 13.1.5.4. SWOT Analysis
- 13.1.6 Abbott Laboratories
- 13.1.6.1. Company Overview
- 13.1.6.2. Products
- 13.1.6.3. Company Financials
- 13.1.6.4. SWOT Analysis
- 13.1.7 Quest Diagnostics
- 13.1.7.1. Company Overview
- 13.1.7.2. Products
- 13.1.7.3. Company Financials
- 13.1.7.4. SWOT Analysis
- 13.1.8 Thermo Fisher Scientific*List Not Exhaustive
- 13.1.8.1. Company Overview
- 13.1.8.2. Products
- 13.1.8.3. Company Financials
- 13.1.8.4. SWOT Analysis
- 13.1.9 Bio-Rad Laboratories
- 13.1.9.1. Company Overview
- 13.1.9.2. Products
- 13.1.9.3. Company Financials
- 13.1.9.4. SWOT Analysis
- 13.1.10 Agilent Technologies
- 13.1.10.1. Company Overview
- 13.1.10.2. Products
- 13.1.10.3. Company Financials
- 13.1.10.4. SWOT Analysis
- 13.1.1 Covance
- 13.2. Market Entropy
- 13.2.1 Company's Key Areas Served
- 13.2.2 Recent Developments
- 13.3. Company Market Share Analysis 2025
- 13.3.1 Top 5 Companies Market Share Analysis
- 13.3.2 Top 3 Companies Market Share Analysis
- 13.4. List of Potential Customers
- 14. Research Methodology
List of Figures
- Figure 1: Global In-Vitro Toxicology Testing Industry Revenue Breakdown (undefined, %) by Region 2025 & 2033
- Figure 2: North America In-Vitro Toxicology Testing Industry Revenue (undefined), by Technology 2025 & 2033
- Figure 3: North America In-Vitro Toxicology Testing Industry Revenue Share (%), by Technology 2025 & 2033
- Figure 4: North America In-Vitro Toxicology Testing Industry Revenue (undefined), by Method 2025 & 2033
- Figure 5: North America In-Vitro Toxicology Testing Industry Revenue Share (%), by Method 2025 & 2033
- Figure 6: North America In-Vitro Toxicology Testing Industry Revenue (undefined), by Application 2025 & 2033
- Figure 7: North America In-Vitro Toxicology Testing Industry Revenue Share (%), by Application 2025 & 2033
- Figure 8: North America In-Vitro Toxicology Testing Industry Revenue (undefined), by End User 2025 & 2033
- Figure 9: North America In-Vitro Toxicology Testing Industry Revenue Share (%), by End User 2025 & 2033
- Figure 10: North America In-Vitro Toxicology Testing Industry Revenue (undefined), by Country 2025 & 2033
- Figure 11: North America In-Vitro Toxicology Testing Industry Revenue Share (%), by Country 2025 & 2033
- Figure 12: Europe In-Vitro Toxicology Testing Industry Revenue (undefined), by Technology 2025 & 2033
- Figure 13: Europe In-Vitro Toxicology Testing Industry Revenue Share (%), by Technology 2025 & 2033
- Figure 14: Europe In-Vitro Toxicology Testing Industry Revenue (undefined), by Method 2025 & 2033
- Figure 15: Europe In-Vitro Toxicology Testing Industry Revenue Share (%), by Method 2025 & 2033
- Figure 16: Europe In-Vitro Toxicology Testing Industry Revenue (undefined), by Application 2025 & 2033
- Figure 17: Europe In-Vitro Toxicology Testing Industry Revenue Share (%), by Application 2025 & 2033
- Figure 18: Europe In-Vitro Toxicology Testing Industry Revenue (undefined), by End User 2025 & 2033
- Figure 19: Europe In-Vitro Toxicology Testing Industry Revenue Share (%), by End User 2025 & 2033
- Figure 20: Europe In-Vitro Toxicology Testing Industry Revenue (undefined), by Country 2025 & 2033
- Figure 21: Europe In-Vitro Toxicology Testing Industry Revenue Share (%), by Country 2025 & 2033
- Figure 22: Asia Pacific In-Vitro Toxicology Testing Industry Revenue (undefined), by Technology 2025 & 2033
- Figure 23: Asia Pacific In-Vitro Toxicology Testing Industry Revenue Share (%), by Technology 2025 & 2033
- Figure 24: Asia Pacific In-Vitro Toxicology Testing Industry Revenue (undefined), by Method 2025 & 2033
- Figure 25: Asia Pacific In-Vitro Toxicology Testing Industry Revenue Share (%), by Method 2025 & 2033
- Figure 26: Asia Pacific In-Vitro Toxicology Testing Industry Revenue (undefined), by Application 2025 & 2033
- Figure 27: Asia Pacific In-Vitro Toxicology Testing Industry Revenue Share (%), by Application 2025 & 2033
- Figure 28: Asia Pacific In-Vitro Toxicology Testing Industry Revenue (undefined), by End User 2025 & 2033
- Figure 29: Asia Pacific In-Vitro Toxicology Testing Industry Revenue Share (%), by End User 2025 & 2033
- Figure 30: Asia Pacific In-Vitro Toxicology Testing Industry Revenue (undefined), by Country 2025 & 2033
- Figure 31: Asia Pacific In-Vitro Toxicology Testing Industry Revenue Share (%), by Country 2025 & 2033
- Figure 32: Middle East In-Vitro Toxicology Testing Industry Revenue (undefined), by Technology 2025 & 2033
- Figure 33: Middle East In-Vitro Toxicology Testing Industry Revenue Share (%), by Technology 2025 & 2033
- Figure 34: Middle East In-Vitro Toxicology Testing Industry Revenue (undefined), by Method 2025 & 2033
- Figure 35: Middle East In-Vitro Toxicology Testing Industry Revenue Share (%), by Method 2025 & 2033
- Figure 36: Middle East In-Vitro Toxicology Testing Industry Revenue (undefined), by Application 2025 & 2033
- Figure 37: Middle East In-Vitro Toxicology Testing Industry Revenue Share (%), by Application 2025 & 2033
- Figure 38: Middle East In-Vitro Toxicology Testing Industry Revenue (undefined), by End User 2025 & 2033
- Figure 39: Middle East In-Vitro Toxicology Testing Industry Revenue Share (%), by End User 2025 & 2033
- Figure 40: Middle East In-Vitro Toxicology Testing Industry Revenue (undefined), by Country 2025 & 2033
- Figure 41: Middle East In-Vitro Toxicology Testing Industry Revenue Share (%), by Country 2025 & 2033
- Figure 42: GCC In-Vitro Toxicology Testing Industry Revenue (undefined), by Technology 2025 & 2033
- Figure 43: GCC In-Vitro Toxicology Testing Industry Revenue Share (%), by Technology 2025 & 2033
- Figure 44: GCC In-Vitro Toxicology Testing Industry Revenue (undefined), by Method 2025 & 2033
- Figure 45: GCC In-Vitro Toxicology Testing Industry Revenue Share (%), by Method 2025 & 2033
- Figure 46: GCC In-Vitro Toxicology Testing Industry Revenue (undefined), by Application 2025 & 2033
- Figure 47: GCC In-Vitro Toxicology Testing Industry Revenue Share (%), by Application 2025 & 2033
- Figure 48: GCC In-Vitro Toxicology Testing Industry Revenue (undefined), by End User 2025 & 2033
- Figure 49: GCC In-Vitro Toxicology Testing Industry Revenue Share (%), by End User 2025 & 2033
- Figure 50: GCC In-Vitro Toxicology Testing Industry Revenue (undefined), by Country 2025 & 2033
- Figure 51: GCC In-Vitro Toxicology Testing Industry Revenue Share (%), by Country 2025 & 2033
- Figure 52: South America In-Vitro Toxicology Testing Industry Revenue (undefined), by Technology 2025 & 2033
- Figure 53: South America In-Vitro Toxicology Testing Industry Revenue Share (%), by Technology 2025 & 2033
- Figure 54: South America In-Vitro Toxicology Testing Industry Revenue (undefined), by Method 2025 & 2033
- Figure 55: South America In-Vitro Toxicology Testing Industry Revenue Share (%), by Method 2025 & 2033
- Figure 56: South America In-Vitro Toxicology Testing Industry Revenue (undefined), by Application 2025 & 2033
- Figure 57: South America In-Vitro Toxicology Testing Industry Revenue Share (%), by Application 2025 & 2033
- Figure 58: South America In-Vitro Toxicology Testing Industry Revenue (undefined), by End User 2025 & 2033
- Figure 59: South America In-Vitro Toxicology Testing Industry Revenue Share (%), by End User 2025 & 2033
- Figure 60: South America In-Vitro Toxicology Testing Industry Revenue (undefined), by Country 2025 & 2033
- Figure 61: South America In-Vitro Toxicology Testing Industry Revenue Share (%), by Country 2025 & 2033
List of Tables
- Table 1: Global In-Vitro Toxicology Testing Industry Revenue undefined Forecast, by Technology 2020 & 2033
- Table 2: Global In-Vitro Toxicology Testing Industry Revenue undefined Forecast, by Method 2020 & 2033
- Table 3: Global In-Vitro Toxicology Testing Industry Revenue undefined Forecast, by Application 2020 & 2033
- Table 4: Global In-Vitro Toxicology Testing Industry Revenue undefined Forecast, by End User 2020 & 2033
- Table 5: Global In-Vitro Toxicology Testing Industry Revenue undefined Forecast, by Region 2020 & 2033
- Table 6: Global In-Vitro Toxicology Testing Industry Revenue undefined Forecast, by Technology 2020 & 2033
- Table 7: Global In-Vitro Toxicology Testing Industry Revenue undefined Forecast, by Method 2020 & 2033
- Table 8: Global In-Vitro Toxicology Testing Industry Revenue undefined Forecast, by Application 2020 & 2033
- Table 9: Global In-Vitro Toxicology Testing Industry Revenue undefined Forecast, by End User 2020 & 2033
- Table 10: Global In-Vitro Toxicology Testing Industry Revenue undefined Forecast, by Country 2020 & 2033
- Table 11: United States In-Vitro Toxicology Testing Industry Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 12: Canada In-Vitro Toxicology Testing Industry Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 13: Mexico In-Vitro Toxicology Testing Industry Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 14: Global In-Vitro Toxicology Testing Industry Revenue undefined Forecast, by Technology 2020 & 2033
- Table 15: Global In-Vitro Toxicology Testing Industry Revenue undefined Forecast, by Method 2020 & 2033
- Table 16: Global In-Vitro Toxicology Testing Industry Revenue undefined Forecast, by Application 2020 & 2033
- Table 17: Global In-Vitro Toxicology Testing Industry Revenue undefined Forecast, by End User 2020 & 2033
- Table 18: Global In-Vitro Toxicology Testing Industry Revenue undefined Forecast, by Country 2020 & 2033
- Table 19: Germany In-Vitro Toxicology Testing Industry Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 20: United Kingdom In-Vitro Toxicology Testing Industry Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 21: France In-Vitro Toxicology Testing Industry Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 22: Italy In-Vitro Toxicology Testing Industry Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 23: Spain In-Vitro Toxicology Testing Industry Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 24: Rest of Europe In-Vitro Toxicology Testing Industry Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 25: Global In-Vitro Toxicology Testing Industry Revenue undefined Forecast, by Technology 2020 & 2033
- Table 26: Global In-Vitro Toxicology Testing Industry Revenue undefined Forecast, by Method 2020 & 2033
- Table 27: Global In-Vitro Toxicology Testing Industry Revenue undefined Forecast, by Application 2020 & 2033
- Table 28: Global In-Vitro Toxicology Testing Industry Revenue undefined Forecast, by End User 2020 & 2033
- Table 29: Global In-Vitro Toxicology Testing Industry Revenue undefined Forecast, by Country 2020 & 2033
- Table 30: China In-Vitro Toxicology Testing Industry Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 31: Japan In-Vitro Toxicology Testing Industry Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 32: India In-Vitro Toxicology Testing Industry Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 33: Australia In-Vitro Toxicology Testing Industry Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 34: South Korea In-Vitro Toxicology Testing Industry Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 35: Rest of Asia Pacific In-Vitro Toxicology Testing Industry Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 36: Global In-Vitro Toxicology Testing Industry Revenue undefined Forecast, by Technology 2020 & 2033
- Table 37: Global In-Vitro Toxicology Testing Industry Revenue undefined Forecast, by Method 2020 & 2033
- Table 38: Global In-Vitro Toxicology Testing Industry Revenue undefined Forecast, by Application 2020 & 2033
- Table 39: Global In-Vitro Toxicology Testing Industry Revenue undefined Forecast, by End User 2020 & 2033
- Table 40: Global In-Vitro Toxicology Testing Industry Revenue undefined Forecast, by Country 2020 & 2033
- Table 41: Global In-Vitro Toxicology Testing Industry Revenue undefined Forecast, by Technology 2020 & 2033
- Table 42: Global In-Vitro Toxicology Testing Industry Revenue undefined Forecast, by Method 2020 & 2033
- Table 43: Global In-Vitro Toxicology Testing Industry Revenue undefined Forecast, by Application 2020 & 2033
- Table 44: Global In-Vitro Toxicology Testing Industry Revenue undefined Forecast, by End User 2020 & 2033
- Table 45: Global In-Vitro Toxicology Testing Industry Revenue undefined Forecast, by Country 2020 & 2033
- Table 46: South Africa In-Vitro Toxicology Testing Industry Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 47: Rest of Middle East In-Vitro Toxicology Testing Industry Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 48: Global In-Vitro Toxicology Testing Industry Revenue undefined Forecast, by Technology 2020 & 2033
- Table 49: Global In-Vitro Toxicology Testing Industry Revenue undefined Forecast, by Method 2020 & 2033
- Table 50: Global In-Vitro Toxicology Testing Industry Revenue undefined Forecast, by Application 2020 & 2033
- Table 51: Global In-Vitro Toxicology Testing Industry Revenue undefined Forecast, by End User 2020 & 2033
- Table 52: Global In-Vitro Toxicology Testing Industry Revenue undefined Forecast, by Country 2020 & 2033
- Table 53: Brazil In-Vitro Toxicology Testing Industry Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 54: Argentina In-Vitro Toxicology Testing Industry Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 55: Rest of South America In-Vitro Toxicology Testing Industry Revenue (undefined) Forecast, by Application 2020 & 2033
Frequently Asked Questions
1. What is the projected Compound Annual Growth Rate (CAGR) of the In-Vitro Toxicology Testing Industry?
The projected CAGR is approximately 10.1%.
2. Which companies are prominent players in the In-Vitro Toxicology Testing Industry?
Key companies in the market include Covance, Promega Corporation, Merck KGaA, Eurofins Scientific, GE Healthcare, Abbott Laboratories, Quest Diagnostics, Thermo Fisher Scientific*List Not Exhaustive, Bio-Rad Laboratories, Agilent Technologies.
3. What are the main segments of the In-Vitro Toxicology Testing Industry?
The market segments include Technology, Method, Application, End User.
4. Can you provide details about the market size?
The market size is estimated to be USD XXX N/A as of 2022.
5. What are some drivers contributing to market growth?
; Opposition to the Usage of Animals in Pre-clinical Research; Significant Advancements In-vitro Toxicology Assays; Increasing Awareness Regarding Drug Product Safety.
6. What are the notable trends driving market growth?
Cell Culture is Expected to Hold Significant Market Share in the Technology Type.
7. Are there any restraints impacting market growth?
; Incapability of In-vitro Models to Determine Autoimmunity and Immunostimulation; Stringent Regulatory Framework for the In-vitro Tests.
8. Can you provide examples of recent developments in the market?
N/A
9. What pricing options are available for accessing the report?
Pricing options include single-user, multi-user, and enterprise licenses priced at USD 4750, USD 5250, and USD 8750 respectively.
10. Is the market size provided in terms of value or volume?
The market size is provided in terms of value, measured in N/A.
11. Are there any specific market keywords associated with the report?
Yes, the market keyword associated with the report is "In-Vitro Toxicology Testing Industry," which aids in identifying and referencing the specific market segment covered.
12. How do I determine which pricing option suits my needs best?
The pricing options vary based on user requirements and access needs. Individual users may opt for single-user licenses, while businesses requiring broader access may choose multi-user or enterprise licenses for cost-effective access to the report.
13. Are there any additional resources or data provided in the In-Vitro Toxicology Testing Industry report?
While the report offers comprehensive insights, it's advisable to review the specific contents or supplementary materials provided to ascertain if additional resources or data are available.
14. How can I stay updated on further developments or reports in the In-Vitro Toxicology Testing Industry?
To stay informed about further developments, trends, and reports in the In-Vitro Toxicology Testing Industry, consider subscribing to industry newsletters, following relevant companies and organizations, or regularly checking reputable industry news sources and publications.
Methodology
Step 1 - Identification of Relevant Samples Size from Population Database



Step 2 - Approaches for Defining Global Market Size (Value, Volume* & Price*)

Note*: In applicable scenarios
Step 3 - Data Sources
Primary Research
- Web Analytics
- Survey Reports
- Research Institute
- Latest Research Reports
- Opinion Leaders
Secondary Research
- Annual Reports
- White Paper
- Latest Press Release
- Industry Association
- Paid Database
- Investor Presentations

Step 4 - Data Triangulation
Involves using different sources of information in order to increase the validity of a study
These sources are likely to be stakeholders in a program - participants, other researchers, program staff, other community members, and so on.
Then we put all data in single framework & apply various statistical tools to find out the dynamic on the market.
During the analysis stage, feedback from the stakeholder groups would be compared to determine areas of agreement as well as areas of divergence


