Key Insights
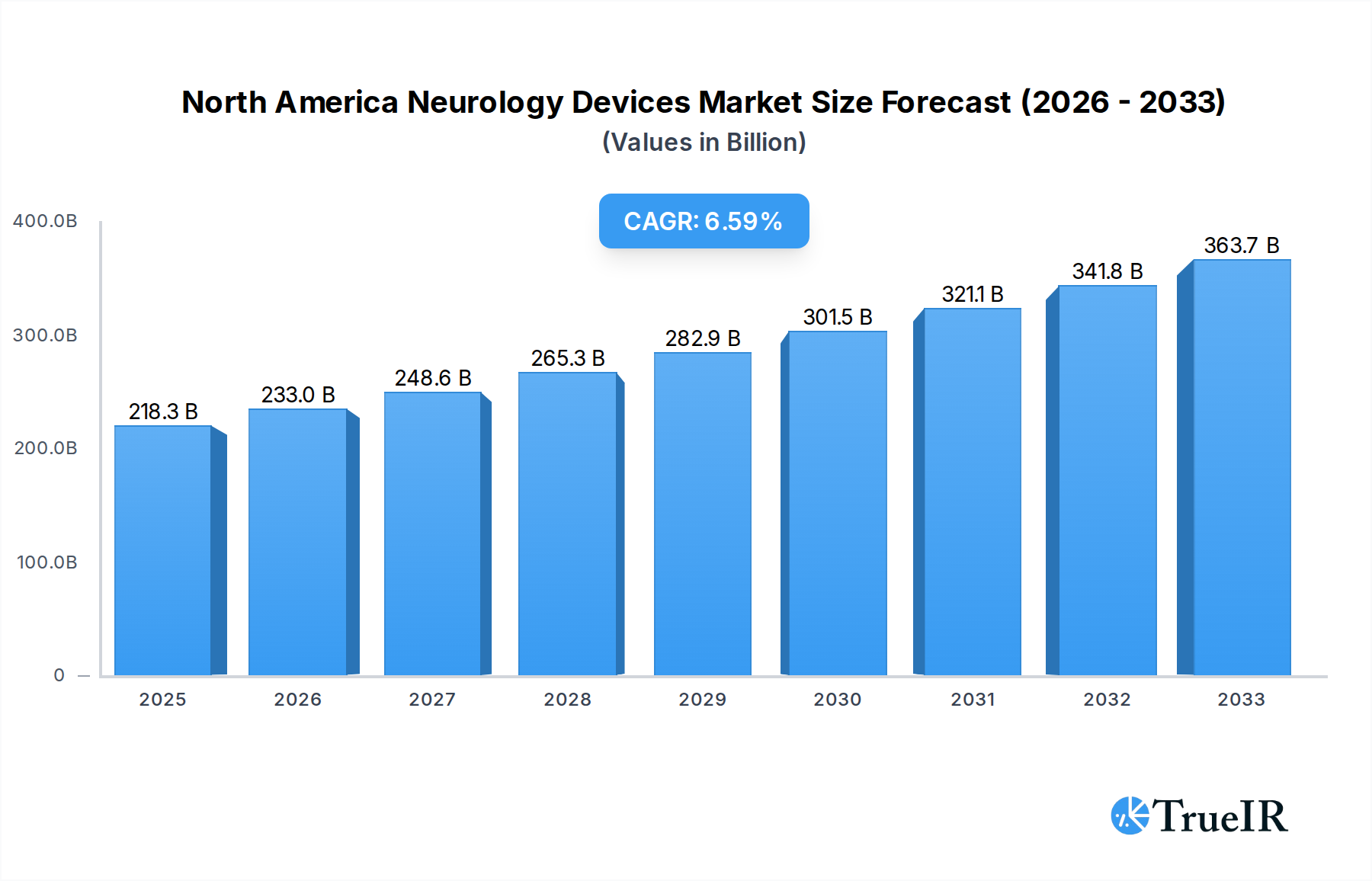
The North America Neurology Devices Market is poised for significant expansion, projected to reach USD 218.26 billion in 2025, with a robust CAGR of 6.7% anticipated throughout the forecast period extending to 2033. This impressive growth is primarily fueled by the increasing prevalence of neurological disorders such as Alzheimer's, Parkinson's, stroke, and epilepsy, coupled with an aging global population that is more susceptible to these conditions. Advancements in minimally invasive surgical techniques and the development of sophisticated diagnostic and therapeutic devices are also key drivers, offering improved patient outcomes and reduced recovery times. Furthermore, the growing demand for neurostimulation devices for pain management and the treatment of mood disorders, alongside the continuous innovation in interventional neurology devices for stroke management and aneurysm treatment, are contributing to the market's upward trajectory. The market's dynamism is further amplified by substantial investments in research and development by leading companies, aimed at creating next-generation neurological solutions.
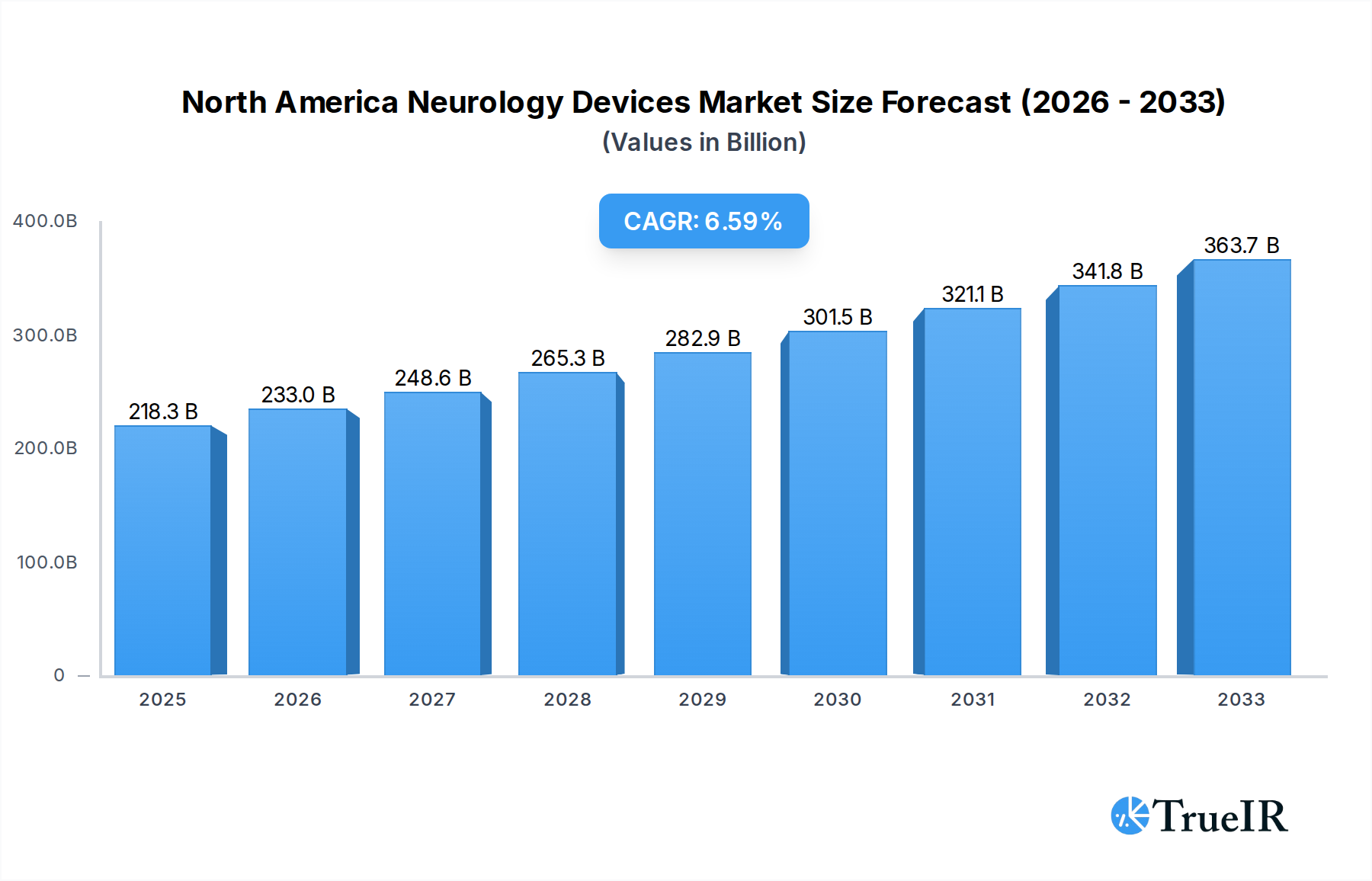
North America Neurology Devices Market Market Size (In Billion)

The market's segmentation reveals a diverse landscape of opportunities across various device categories and geographical regions. Cerebrospinal Fluid Management Devices, Interventional Neurology Devices (including neurothrombectomy, carotid artery stents, and embolic coils), Neurosurgery Devices (such as neuroendoscopes and stereotactic systems), and Neurostimulation Devices (covering spinal cord and deep brain stimulation) are all expected to witness substantial growth. Geographically, the United States, Canada, and Mexico collectively form a dynamic North American market, with the United States likely to hold the largest share due to its advanced healthcare infrastructure, high adoption rates of new technologies, and significant healthcare expenditure. The competitive landscape is characterized by the presence of major global players like Medtronic PLC, Abbott Laboratories, Johnson and Johnson, and Stryker Corporation, who are actively engaged in product innovation, strategic collaborations, and market expansion to capitalize on the burgeoning demand for advanced neurological solutions.
North America Neurology Devices Market Company Market Share

This in-depth report provides a definitive analysis of the North America Neurology Devices Market, meticulously detailing its current state, projected growth, and the intricate factors shaping its future. Covering the historical period from 2019 to 2024 and extending to a comprehensive forecast up to 2033, with a base and estimated year of 2025, this study is an indispensable resource for stakeholders seeking to understand the dynamic landscape of neurological treatment technologies across the United States, Canada, and Mexico. The market is projected to reach $XX billion by 2033, exhibiting a robust CAGR of XX% during the forecast period.
North America Neurology Devices Market Market Structure & Competitive Landscape
The North America Neurology Devices Market is characterized by a moderate to highly concentrated competitive landscape, with a significant presence of established global players and emerging innovators. Key companies like Smith & Nephew, Abbott Laboratories, Medtronic PLC, Johnson and Johnson, and Stryker Corporation dominate the market share, driven by continuous product development and strategic acquisitions. Innovation is a critical driver, fueled by advancements in minimally invasive techniques, improved diagnostic tools, and the integration of artificial intelligence and robotics in neurosurgery. Regulatory frameworks, particularly those from the FDA in the United States, play a pivotal role, influencing product approvals, market entry, and post-market surveillance. While direct product substitutes are limited, the development of alternative treatment modalities and preventative neurological care strategies present an indirect competitive pressure. End-user segmentation includes hospitals, neurosurgery clinics, diagnostic centers, and academic research institutions, each with distinct purchasing patterns and needs. Mergers and acquisitions (M&A) are prevalent, with an estimated XX significant M&A activities recorded in the historical period, aimed at consolidating market positions, expanding product portfolios, and gaining access to new technologies and geographical markets. The market concentration ratio for the top five players is estimated to be around XX%.
North America Neurology Devices Market Market Trends & Opportunities
The North America Neurology Devices Market is experiencing a period of significant expansion, propelled by a confluence of technological advancements, evolving patient demographics, and an increasing burden of neurological disorders. The market size, valued at $XX billion in the base year 2025, is projected to witness substantial growth in the coming decade. Key trends include the burgeoning demand for minimally invasive neurosurgical procedures, which offer reduced recovery times and lower complication rates, thereby enhancing patient outcomes. This trend is directly fueling the growth of interventional neurology devices, such as neurothrombectomy devices and embolic coils.
Technological shifts are at the forefront of market evolution. The integration of artificial intelligence (AI) and machine learning (ML) is revolutionizing diagnostics and treatment planning, enabling more personalized and effective interventions. Robotic-assisted surgery platforms are gaining traction, offering enhanced precision and control for complex neurological procedures. Furthermore, the rise of digital health and connectivity is transforming patient monitoring and management. Upgraded digital health apps, like Abbott's NeuroSphere myPath, exemplify this trend, allowing for remote patient monitoring and optimized treatment adjustments, particularly for chronic pain management through neurostimulation devices.
Consumer preferences are increasingly leaning towards less invasive and more patient-centric treatments. This is driving innovation in areas such as neurostimulation devices for chronic pain, epilepsy, and movement disorders, with a growing emphasis on implantable devices with advanced functionality and long-term efficacy. The increasing prevalence of age-related neurological conditions, such as Alzheimer's disease and Parkinson's disease, coupled with a rise in stroke incidence, further amplifies the demand for sophisticated diagnostic and therapeutic neurology devices.
Competitive dynamics are intensifying, with companies investing heavily in research and development to gain a competitive edge. Strategic collaborations and partnerships are becoming crucial for accelerating innovation and expanding market reach. Opportunities abound in developing cost-effective solutions for emerging markets within North America, enhancing the accessibility of advanced neurological treatments. The market penetration rate for advanced neurostimulation devices is currently estimated at XX% and is expected to rise significantly. The overall market is forecast to grow at a Compound Annual Growth Rate (CAGR) of XX% from 2025 to 2033.
Dominant Markets & Segments in North America Neurology Devices Market
The United States stands as the dominant market within North America for neurology devices, accounting for approximately XX% of the regional market share. This leadership is attributed to several key factors, including a highly developed healthcare infrastructure, substantial investment in medical research and development, a favorable reimbursement landscape for advanced neurological treatments, and a high prevalence of neurological disorders coupled with an aging population. The robust presence of leading medical device manufacturers and a strong ecosystem of specialized neurological hospitals and research institutions further solidify the U.S.'s position.
Within the device segments, Neurostimulation Devices currently represent the largest and fastest-growing segment, projected to reach $XX billion by 2033, driven by increasing diagnoses of chronic pain, epilepsy, and movement disorders such as Parkinson's disease. The demand for spinal cord stimulation (SCS) devices and deep brain stimulation (DBS) devices is particularly strong, fueled by their proven efficacy in managing debilitating conditions and advancements in device technology, including rechargeable systems and closed-loop functionalities.
Interventional Neurology Devices also hold a significant market share and are expected to witness robust growth, driven by the increasing incidence of stroke and the rising adoption of minimally invasive endovascular procedures. Key sub-segments like neurothrombectomy devices and embolic coils are witnessing heightened demand due to their critical role in acute stroke treatment and the management of aneurysms. The development of improved stent retrievers and advanced embolic agents is further propelling this segment.
The United States leads in all major device segments, followed by Canada and then Mexico. However, Mexico presents a significant growth opportunity due to its expanding healthcare sector, increasing medical tourism, and growing awareness of neurological conditions.
Key growth drivers across these dominant markets and segments include:
- Infrastructure: Extensive network of specialized neurological centers and hospitals in the U.S. and Canada, equipped with advanced diagnostic and surgical capabilities.
- Policies: Favorable regulatory pathways for novel device approvals and robust reimbursement policies for neurological treatments, particularly in the United States.
- Technological Advancements: Continuous innovation in minimally invasive techniques, robotic surgery, AI-powered diagnostics, and advanced implantable devices.
- Disease Prevalence: High and increasing incidence of neurological disorders, including stroke, Alzheimer's, Parkinson's, epilepsy, and chronic pain, creating sustained demand for effective treatment solutions.
- Aging Population: The demographic shift towards an older population in North America directly correlates with a higher susceptibility to various neurological conditions.
The Neurosurgery Devices segment, including neuroendoscopes and stereotactic systems, also contributes significantly, driven by the need for precision in complex brain surgeries. Cerebrospinal Fluid Management Devices are crucial for treating conditions like hydrocephalus, while Other Devices encompass a range of diagnostic and therapeutic tools essential for comprehensive neurological care. The synergistic growth of these segments, underpinned by innovation and demand, will continue to shape the North America Neurology Devices Market.
North America Neurology Devices Market Product Analysis
The North America Neurology Devices Market is defined by a continuous stream of product innovations aimed at enhancing patient outcomes, improving procedural efficiency, and expanding treatment accessibility. Recent advancements include the development of smarter, more miniaturized neurostimulation devices with enhanced programmability for personalized therapy. In interventional neurology, the focus is on developing thrombectomy devices with improved navigation and clot retrieval capabilities, alongside advanced embolic coils designed for aneurysm treatment with greater precision and reduced recurrence rates. Neurosurgery is witnessing the integration of robotics and AI for enhanced surgical precision and real-time imaging feedback. Cerebrospinal fluid management devices are evolving to offer more intelligent monitoring and regulation. These innovations offer significant competitive advantages by addressing unmet clinical needs and providing superior therapeutic efficacy.
Key Drivers, Barriers & Challenges in North America Neurology Devices Market
The North America Neurology Devices Market is propelled by several key drivers, including the escalating prevalence of neurological disorders, rapid technological advancements leading to innovative treatments, and increasing healthcare expenditure across the region. The aging demographic, a growing awareness of neurological conditions, and favorable reimbursement policies in key markets like the United States further fuel market growth.
However, the market faces significant challenges. High research and development costs and lengthy regulatory approval processes pose substantial barriers. Intense competition from established players and emerging companies can also impact market dynamics. Supply chain disruptions, particularly in the wake of global events, and cybersecurity concerns related to connected medical devices represent ongoing restraints. Moreover, the need for specialized training for healthcare professionals to utilize advanced neurology devices can also limit their widespread adoption in certain areas.
Growth Drivers in the North America Neurology Devices Market Market
The growth of the North America Neurology Devices Market is primarily driven by the increasing burden of neurological diseases, such as stroke, Alzheimer's, Parkinson's, and epilepsy, necessitating advanced treatment solutions. Rapid technological advancements in areas like neurostimulation, interventional neurology, and neurosurgery are creating novel and more effective therapeutic options. An aging population demographic across the United States, Canada, and Mexico further escalates the demand for neurological care. Supportive government initiatives and expanding healthcare infrastructure in certain regions also contribute to market expansion.
Challenges Impacting North America Neurology Devices Market Growth
Despite robust growth prospects, the North America Neurology Devices Market is constrained by several factors. The high cost associated with research, development, and manufacturing of sophisticated neurology devices leads to premium pricing, potentially limiting affordability and access. Stringent regulatory approval processes, while ensuring safety and efficacy, can significantly prolong time-to-market for new innovations. Intense market competition necessitates continuous innovation and significant investment, creating pressure on profit margins. Moreover, challenges related to the supply chain, including sourcing of specialized components and logistics, can disrupt production and distribution.
Key Players Shaping the North America Neurology Devices Market Market
- Smith & Nephew
- Abbott Laboratories
- Medtronic PLC
- Johnson and Johnson
- Stryker Corporation
- MicroPort Scientific Corporation
- Boston Scientific Corporation
- Nihon Kohden Corporation
- B Braun SE
- Penumbra Inc
Significant North America Neurology Devices Market Industry Milestones
- October 2022: Medtronic launched the Neurovascular Co-Lab Platform for accelerating innovation in stroke care and treatment to drive collaboration and connections to help bring neurovascular innovations to life.
- April 2022: Abbott launched an upgraded version of its digital health app, NeuroSphere myPath. The app's functionality has been enhanced so that it may better assist physicians in monitoring their patients as they utilize Abbott neurostimulation devices to treat their chronic pain.
Future Outlook for North America Neurology Devices Market Market
The future outlook for the North America Neurology Devices Market is exceptionally promising, characterized by sustained growth driven by unmet clinical needs and relentless technological innovation. The increasing adoption of minimally invasive procedures, coupled with advancements in AI-powered diagnostics and robotic-assisted surgery, will redefine neurological treatment paradigms. Expansion opportunities lie in developing more affordable and accessible solutions, particularly for chronic pain management and stroke rehabilitation. Strategic collaborations and a focus on personalized medicine will be crucial for market leaders to capitalize on emerging trends and maintain a competitive edge, ensuring improved patient outcomes and driving market expansion through 2033.
North America Neurology Devices Market Segmentation
-
1. Device
- 1.1. Cerebrospinal Fluid Management Devices
-
1.2. Interventional Neurology Devices
- 1.2.1. Interventional/Surgical Simulators
- 1.2.2. Neurothrombectomy Devices
- 1.2.3. Carotid Artery Stents
- 1.2.4. Embolic Coils
- 1.2.5. Support Devices
-
1.3. Neurosurgery Devices
- 1.3.1. Neuroendoscopes
- 1.3.2. Stereotactic Systems
- 1.3.3. Aneurysm Clips
- 1.3.4. Other Neurosurgery Devices
-
1.4. Neurostimulation Devices
- 1.4.1. Spinal Cord Stimulation Devices
- 1.4.2. Deep Brain Stimulation Devices
- 1.4.3. Sacral Nerve Stimulation Devices
- 1.4.4. Other Neurostimulation Devices
- 1.5. Other Devices
-
2. Geography
- 2.1. United States
- 2.2. Canada
- 2.3. Mexico
North America Neurology Devices Market Segmentation By Geography
- 1. United States
- 2. Canada
- 3. Mexico
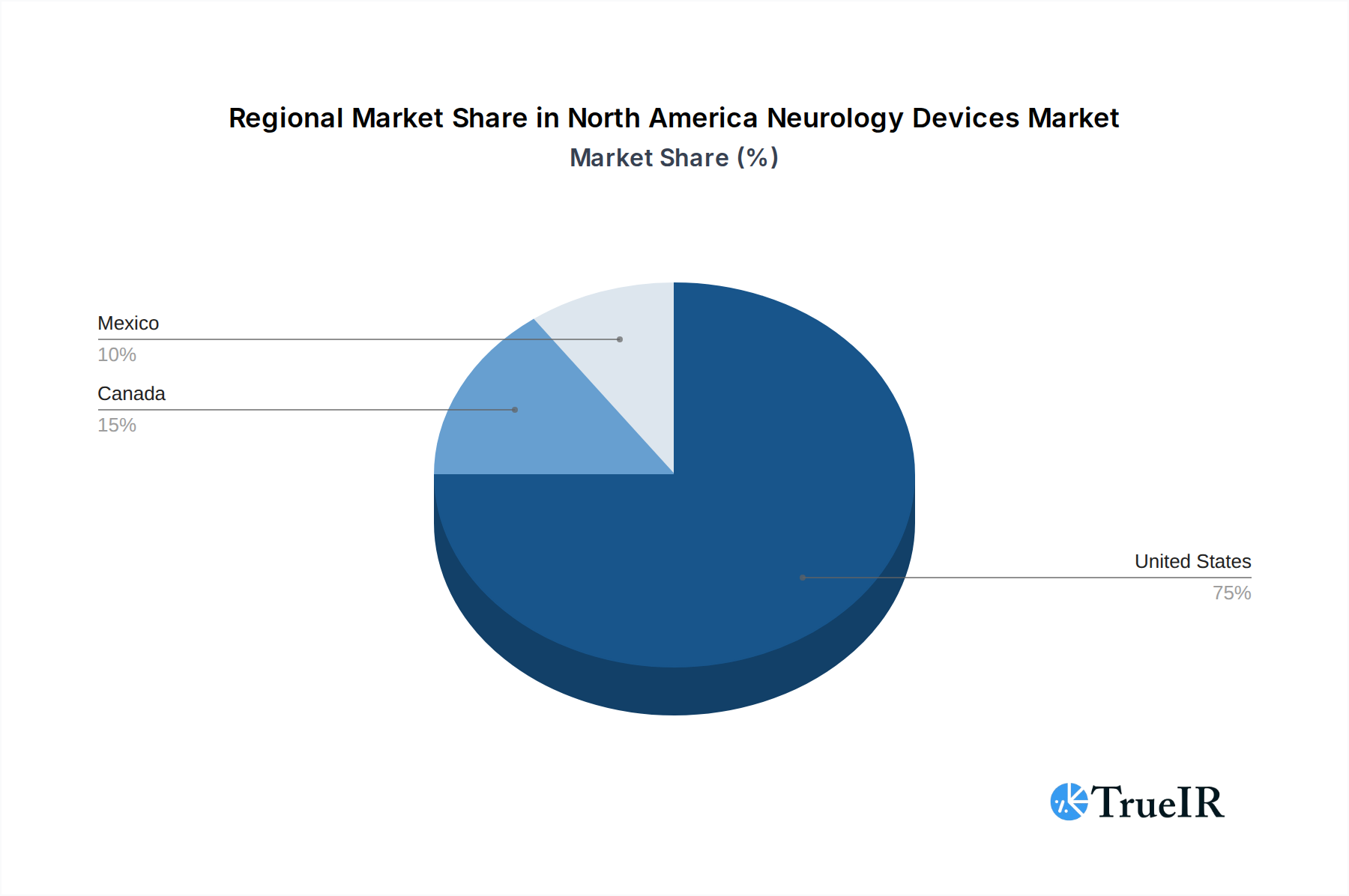
North America Neurology Devices Market Regional Market Share

Geographic Coverage of North America Neurology Devices Market
North America Neurology Devices Market REPORT HIGHLIGHTS
| Aspects | Details |
|---|---|
| Study Period | 2020-2034 |
| Base Year | 2025 |
| Estimated Year | 2026 |
| Forecast Period | 2026-2034 |
| Historical Period | 2020-2025 |
| Growth Rate | CAGR of 6.7% from 2020-2034 |
| Segmentation |
|
Table of Contents
- 1. Introduction
- 1.1. Research Scope
- 1.2. Market Segmentation
- 1.3. Research Objective
- 1.4. Definitions and Assumptions
- 2. Executive Summary
- 2.1. Market Snapshot
- 3. Market Dynamics
- 3.1. Market Drivers
- 3.2. Market Restrains
- 3.3. Market Trends
- 3.4. Market Opportunities
- 4. Market Factor Analysis
- 4.1. Porters Five Forces
- 4.1.1. Bargaining Power of Suppliers
- 4.1.2. Bargaining Power of Buyers
- 4.1.3. Threat of New Entrants
- 4.1.4. Threat of Substitutes
- 4.1.5. Competitive Rivalry
- 4.2. PESTEL analysis
- 4.3. BCG Analysis
- 4.3.1. Stars (High Growth, High Market Share)
- 4.3.2. Cash Cows (Low Growth, High Market Share)
- 4.3.3. Question Mark (High Growth, Low Market Share)
- 4.3.4. Dogs (Low Growth, Low Market Share)
- 4.4. Ansoff Matrix Analysis
- 4.5. Supply Chain Analysis
- 4.6. Regulatory Landscape
- 4.7. Current Market Potential and Opportunity Assessment (TAM–SAM–SOM Framework)
- 4.8. TIR Analyst Note
- 4.1. Porters Five Forces
- 5. Market Analysis, Insights and Forecast 2021-2033
- 5.1. Market Analysis, Insights and Forecast - by Device
- 5.1.1. Cerebrospinal Fluid Management Devices
- 5.1.2. Interventional Neurology Devices
- 5.1.2.1. Interventional/Surgical Simulators
- 5.1.2.2. Neurothrombectomy Devices
- 5.1.2.3. Carotid Artery Stents
- 5.1.2.4. Embolic Coils
- 5.1.2.5. Support Devices
- 5.1.3. Neurosurgery Devices
- 5.1.3.1. Neuroendoscopes
- 5.1.3.2. Stereotactic Systems
- 5.1.3.3. Aneurysm Clips
- 5.1.3.4. Other Neurosurgery Devices
- 5.1.4. Neurostimulation Devices
- 5.1.4.1. Spinal Cord Stimulation Devices
- 5.1.4.2. Deep Brain Stimulation Devices
- 5.1.4.3. Sacral Nerve Stimulation Devices
- 5.1.4.4. Other Neurostimulation Devices
- 5.1.5. Other Devices
- 5.2. Market Analysis, Insights and Forecast - by Geography
- 5.2.1. United States
- 5.2.2. Canada
- 5.2.3. Mexico
- 5.3. Market Analysis, Insights and Forecast - by Region
- 5.3.1. United States
- 5.3.2. Canada
- 5.3.3. Mexico
- 5.1. Market Analysis, Insights and Forecast - by Device
- 6. North America Neurology Devices Market Analysis, Insights and Forecast, 2021-2033
- 6.1. Market Analysis, Insights and Forecast - by Device
- 6.1.1. Cerebrospinal Fluid Management Devices
- 6.1.2. Interventional Neurology Devices
- 6.1.2.1. Interventional/Surgical Simulators
- 6.1.2.2. Neurothrombectomy Devices
- 6.1.2.3. Carotid Artery Stents
- 6.1.2.4. Embolic Coils
- 6.1.2.5. Support Devices
- 6.1.3. Neurosurgery Devices
- 6.1.3.1. Neuroendoscopes
- 6.1.3.2. Stereotactic Systems
- 6.1.3.3. Aneurysm Clips
- 6.1.3.4. Other Neurosurgery Devices
- 6.1.4. Neurostimulation Devices
- 6.1.4.1. Spinal Cord Stimulation Devices
- 6.1.4.2. Deep Brain Stimulation Devices
- 6.1.4.3. Sacral Nerve Stimulation Devices
- 6.1.4.4. Other Neurostimulation Devices
- 6.1.5. Other Devices
- 6.2. Market Analysis, Insights and Forecast - by Geography
- 6.2.1. United States
- 6.2.2. Canada
- 6.2.3. Mexico
- 6.1. Market Analysis, Insights and Forecast - by Device
- 7. United States North America Neurology Devices Market Analysis, Insights and Forecast, 2020-2032
- 7.1. Market Analysis, Insights and Forecast - by Device
- 7.1.1. Cerebrospinal Fluid Management Devices
- 7.1.2. Interventional Neurology Devices
- 7.1.2.1. Interventional/Surgical Simulators
- 7.1.2.2. Neurothrombectomy Devices
- 7.1.2.3. Carotid Artery Stents
- 7.1.2.4. Embolic Coils
- 7.1.2.5. Support Devices
- 7.1.3. Neurosurgery Devices
- 7.1.3.1. Neuroendoscopes
- 7.1.3.2. Stereotactic Systems
- 7.1.3.3. Aneurysm Clips
- 7.1.3.4. Other Neurosurgery Devices
- 7.1.4. Neurostimulation Devices
- 7.1.4.1. Spinal Cord Stimulation Devices
- 7.1.4.2. Deep Brain Stimulation Devices
- 7.1.4.3. Sacral Nerve Stimulation Devices
- 7.1.4.4. Other Neurostimulation Devices
- 7.1.5. Other Devices
- 7.2. Market Analysis, Insights and Forecast - by Geography
- 7.2.1. United States
- 7.2.2. Canada
- 7.2.3. Mexico
- 7.1. Market Analysis, Insights and Forecast - by Device
- 8. Canada North America Neurology Devices Market Analysis, Insights and Forecast, 2020-2032
- 8.1. Market Analysis, Insights and Forecast - by Device
- 8.1.1. Cerebrospinal Fluid Management Devices
- 8.1.2. Interventional Neurology Devices
- 8.1.2.1. Interventional/Surgical Simulators
- 8.1.2.2. Neurothrombectomy Devices
- 8.1.2.3. Carotid Artery Stents
- 8.1.2.4. Embolic Coils
- 8.1.2.5. Support Devices
- 8.1.3. Neurosurgery Devices
- 8.1.3.1. Neuroendoscopes
- 8.1.3.2. Stereotactic Systems
- 8.1.3.3. Aneurysm Clips
- 8.1.3.4. Other Neurosurgery Devices
- 8.1.4. Neurostimulation Devices
- 8.1.4.1. Spinal Cord Stimulation Devices
- 8.1.4.2. Deep Brain Stimulation Devices
- 8.1.4.3. Sacral Nerve Stimulation Devices
- 8.1.4.4. Other Neurostimulation Devices
- 8.1.5. Other Devices
- 8.2. Market Analysis, Insights and Forecast - by Geography
- 8.2.1. United States
- 8.2.2. Canada
- 8.2.3. Mexico
- 8.1. Market Analysis, Insights and Forecast - by Device
- 9. Mexico North America Neurology Devices Market Analysis, Insights and Forecast, 2020-2032
- 9.1. Market Analysis, Insights and Forecast - by Device
- 9.1.1. Cerebrospinal Fluid Management Devices
- 9.1.2. Interventional Neurology Devices
- 9.1.2.1. Interventional/Surgical Simulators
- 9.1.2.2. Neurothrombectomy Devices
- 9.1.2.3. Carotid Artery Stents
- 9.1.2.4. Embolic Coils
- 9.1.2.5. Support Devices
- 9.1.3. Neurosurgery Devices
- 9.1.3.1. Neuroendoscopes
- 9.1.3.2. Stereotactic Systems
- 9.1.3.3. Aneurysm Clips
- 9.1.3.4. Other Neurosurgery Devices
- 9.1.4. Neurostimulation Devices
- 9.1.4.1. Spinal Cord Stimulation Devices
- 9.1.4.2. Deep Brain Stimulation Devices
- 9.1.4.3. Sacral Nerve Stimulation Devices
- 9.1.4.4. Other Neurostimulation Devices
- 9.1.5. Other Devices
- 9.2. Market Analysis, Insights and Forecast - by Geography
- 9.2.1. United States
- 9.2.2. Canada
- 9.2.3. Mexico
- 9.1. Market Analysis, Insights and Forecast - by Device
- 10. Competitive Analysis
- 10.1. Company Profiles
- 10.1.1 Smith & Nephew
- 10.1.1.1. Company Overview
- 10.1.1.2. Products
- 10.1.1.3. Company Financials
- 10.1.1.4. SWOT Analysis
- 10.1.2 Abbott Laboratories
- 10.1.2.1. Company Overview
- 10.1.2.2. Products
- 10.1.2.3. Company Financials
- 10.1.2.4. SWOT Analysis
- 10.1.3 Medtronic PLC
- 10.1.3.1. Company Overview
- 10.1.3.2. Products
- 10.1.3.3. Company Financials
- 10.1.3.4. SWOT Analysis
- 10.1.4 Johnson and Johnson
- 10.1.4.1. Company Overview
- 10.1.4.2. Products
- 10.1.4.3. Company Financials
- 10.1.4.4. SWOT Analysis
- 10.1.5 Stryker Corporation*List Not Exhaustive
- 10.1.5.1. Company Overview
- 10.1.5.2. Products
- 10.1.5.3. Company Financials
- 10.1.5.4. SWOT Analysis
- 10.1.6 MicroPort Scientific Corporation
- 10.1.6.1. Company Overview
- 10.1.6.2. Products
- 10.1.6.3. Company Financials
- 10.1.6.4. SWOT Analysis
- 10.1.7 Boston Scientific Corporation
- 10.1.7.1. Company Overview
- 10.1.7.2. Products
- 10.1.7.3. Company Financials
- 10.1.7.4. SWOT Analysis
- 10.1.8 Nihon Kohden Corporation
- 10.1.8.1. Company Overview
- 10.1.8.2. Products
- 10.1.8.3. Company Financials
- 10.1.8.4. SWOT Analysis
- 10.1.9 B Braun SE
- 10.1.9.1. Company Overview
- 10.1.9.2. Products
- 10.1.9.3. Company Financials
- 10.1.9.4. SWOT Analysis
- 10.1.10 Penumbra Inc
- 10.1.10.1. Company Overview
- 10.1.10.2. Products
- 10.1.10.3. Company Financials
- 10.1.10.4. SWOT Analysis
- 10.1.1 Smith & Nephew
- 10.2. Market Entropy
- 10.2.1 Company's Key Areas Served
- 10.2.2 Recent Developments
- 10.3. Company Market Share Analysis 2025
- 10.3.1 Top 5 Companies Market Share Analysis
- 10.3.2 Top 3 Companies Market Share Analysis
- 10.4. List of Potential Customers
- 11. Research Methodology
List of Figures
- Figure 1: North America Neurology Devices Market Revenue Breakdown (undefined, %) by Product 2025 & 2033
- Figure 2: North America Neurology Devices Market Share (%) by Company 2025
List of Tables
- Table 1: North America Neurology Devices Market Revenue undefined Forecast, by Device 2020 & 2033
- Table 2: North America Neurology Devices Market Volume K Units Forecast, by Device 2020 & 2033
- Table 3: North America Neurology Devices Market Revenue undefined Forecast, by Geography 2020 & 2033
- Table 4: North America Neurology Devices Market Volume K Units Forecast, by Geography 2020 & 2033
- Table 5: North America Neurology Devices Market Revenue undefined Forecast, by Region 2020 & 2033
- Table 6: North America Neurology Devices Market Volume K Units Forecast, by Region 2020 & 2033
- Table 7: North America Neurology Devices Market Revenue undefined Forecast, by Device 2020 & 2033
- Table 8: North America Neurology Devices Market Volume K Units Forecast, by Device 2020 & 2033
- Table 9: North America Neurology Devices Market Revenue undefined Forecast, by Geography 2020 & 2033
- Table 10: North America Neurology Devices Market Volume K Units Forecast, by Geography 2020 & 2033
- Table 11: North America Neurology Devices Market Revenue undefined Forecast, by Country 2020 & 2033
- Table 12: North America Neurology Devices Market Volume K Units Forecast, by Country 2020 & 2033
- Table 13: North America Neurology Devices Market Revenue undefined Forecast, by Device 2020 & 2033
- Table 14: North America Neurology Devices Market Volume K Units Forecast, by Device 2020 & 2033
- Table 15: North America Neurology Devices Market Revenue undefined Forecast, by Geography 2020 & 2033
- Table 16: North America Neurology Devices Market Volume K Units Forecast, by Geography 2020 & 2033
- Table 17: North America Neurology Devices Market Revenue undefined Forecast, by Country 2020 & 2033
- Table 18: North America Neurology Devices Market Volume K Units Forecast, by Country 2020 & 2033
- Table 19: North America Neurology Devices Market Revenue undefined Forecast, by Device 2020 & 2033
- Table 20: North America Neurology Devices Market Volume K Units Forecast, by Device 2020 & 2033
- Table 21: North America Neurology Devices Market Revenue undefined Forecast, by Geography 2020 & 2033
- Table 22: North America Neurology Devices Market Volume K Units Forecast, by Geography 2020 & 2033
- Table 23: North America Neurology Devices Market Revenue undefined Forecast, by Country 2020 & 2033
- Table 24: North America Neurology Devices Market Volume K Units Forecast, by Country 2020 & 2033
Frequently Asked Questions
1. What is the projected Compound Annual Growth Rate (CAGR) of the North America Neurology Devices Market?
The projected CAGR is approximately 6.7%.
2. Which companies are prominent players in the North America Neurology Devices Market?
Key companies in the market include Smith & Nephew, Abbott Laboratories, Medtronic PLC, Johnson and Johnson, Stryker Corporation*List Not Exhaustive, MicroPort Scientific Corporation, Boston Scientific Corporation, Nihon Kohden Corporation, B Braun SE, Penumbra Inc.
3. What are the main segments of the North America Neurology Devices Market?
The market segments include Device, Geography.
4. Can you provide details about the market size?
The market size is estimated to be USD XXX N/A as of 2022.
5. What are some drivers contributing to market growth?
Rising Aging Population and Increase in Incidence of Neurological Disorders; Huge Investments by Private Players in Neurology Devices; Increase in R&D in the Field of Neurotherapies.
6. What are the notable trends driving market growth?
Cerebrospinal Fluid Management Devices is Expected to Witness High Growth over the Forecast Period.
7. Are there any restraints impacting market growth?
High Cost of Equipment; Stringent FDA Validation and Guidelines for New Devices.
8. Can you provide examples of recent developments in the market?
October 2022: Medtronic launched the Neurovascular Co-Lab Platform for accelerating innovation in stroke care and treatment to drive collaboration and connections to help bring neurovascular innovations to life.
9. What pricing options are available for accessing the report?
Pricing options include single-user, multi-user, and enterprise licenses priced at USD 4750, USD 5250, and USD 8750 respectively.
10. Is the market size provided in terms of value or volume?
The market size is provided in terms of value, measured in N/A and volume, measured in K Units.
11. Are there any specific market keywords associated with the report?
Yes, the market keyword associated with the report is "North America Neurology Devices Market," which aids in identifying and referencing the specific market segment covered.
12. How do I determine which pricing option suits my needs best?
The pricing options vary based on user requirements and access needs. Individual users may opt for single-user licenses, while businesses requiring broader access may choose multi-user or enterprise licenses for cost-effective access to the report.
13. Are there any additional resources or data provided in the North America Neurology Devices Market report?
While the report offers comprehensive insights, it's advisable to review the specific contents or supplementary materials provided to ascertain if additional resources or data are available.
14. How can I stay updated on further developments or reports in the North America Neurology Devices Market?
To stay informed about further developments, trends, and reports in the North America Neurology Devices Market, consider subscribing to industry newsletters, following relevant companies and organizations, or regularly checking reputable industry news sources and publications.
Methodology
Step 1 - Identification of Relevant Samples Size from Population Database



Step 2 - Approaches for Defining Global Market Size (Value, Volume* & Price*)

Note*: In applicable scenarios
Step 3 - Data Sources
Primary Research
- Web Analytics
- Survey Reports
- Research Institute
- Latest Research Reports
- Opinion Leaders
Secondary Research
- Annual Reports
- White Paper
- Latest Press Release
- Industry Association
- Paid Database
- Investor Presentations

Step 4 - Data Triangulation
Involves using different sources of information in order to increase the validity of a study
These sources are likely to be stakeholders in a program - participants, other researchers, program staff, other community members, and so on.
Then we put all data in single framework & apply various statistical tools to find out the dynamic on the market.
During the analysis stage, feedback from the stakeholder groups would be compared to determine areas of agreement as well as areas of divergence


