Key Insights
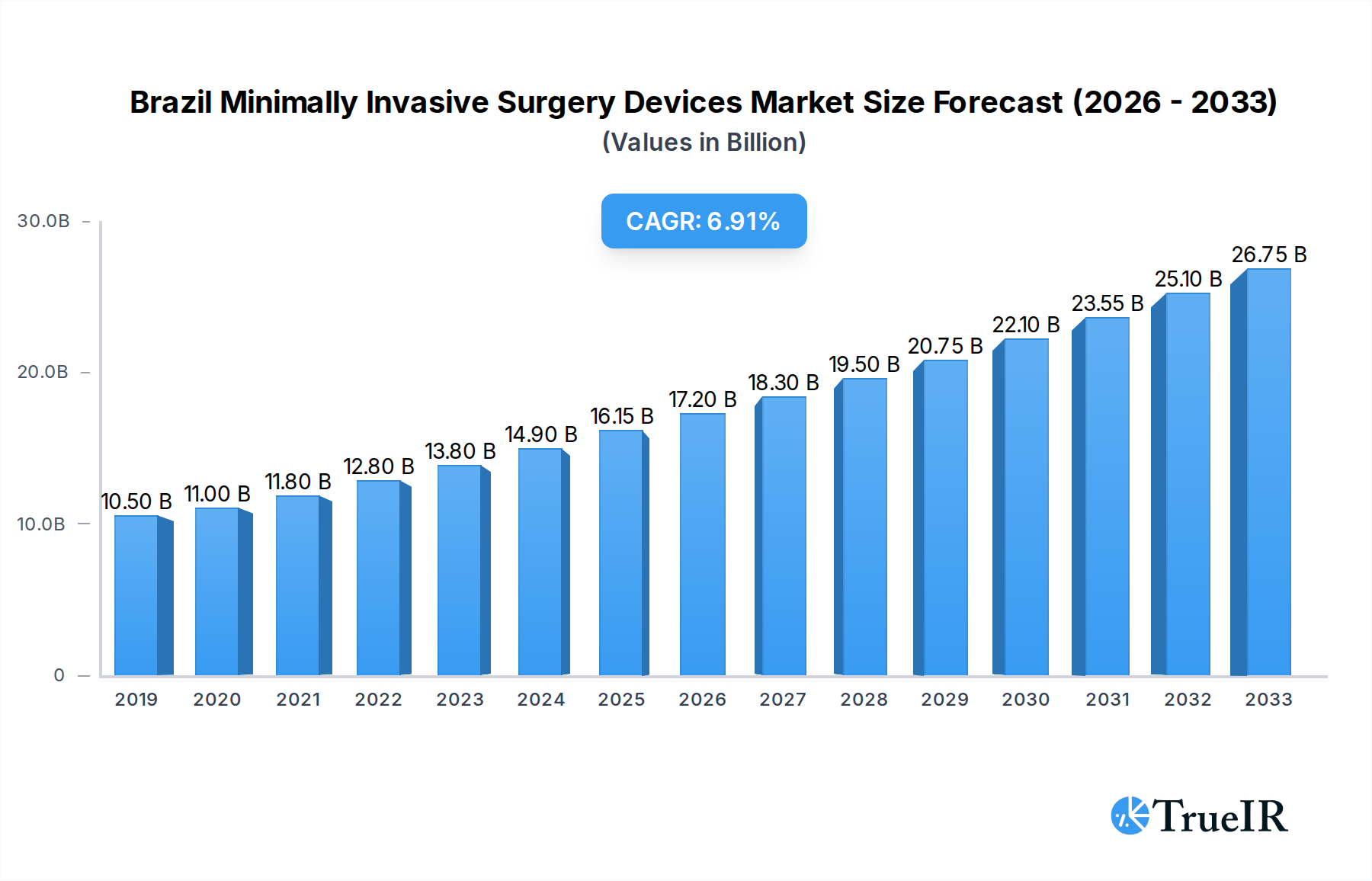
The Brazilian Minimally Invasive Surgery (MIS) Devices Market is poised for significant expansion, projected to reach approximately $16.15 billion in 2025, with an anticipated Compound Annual Growth Rate (CAGR) of 6.4% through 2033. This robust growth trajectory is driven by increasing adoption of advanced surgical techniques, a rising prevalence of chronic diseases requiring surgical intervention, and a growing demand for less invasive procedures due to faster recovery times and reduced patient discomfort. Key product segments, including handheld instruments, guiding devices, and electrosurgical devices, are expected to witness sustained demand. Furthermore, the application spectrum is broad, with aesthetic, cardiovascular, gastrointestinal, and orthopedic procedures leading the charge, reflecting a diversification in the use of MIS technologies across various medical specialties.
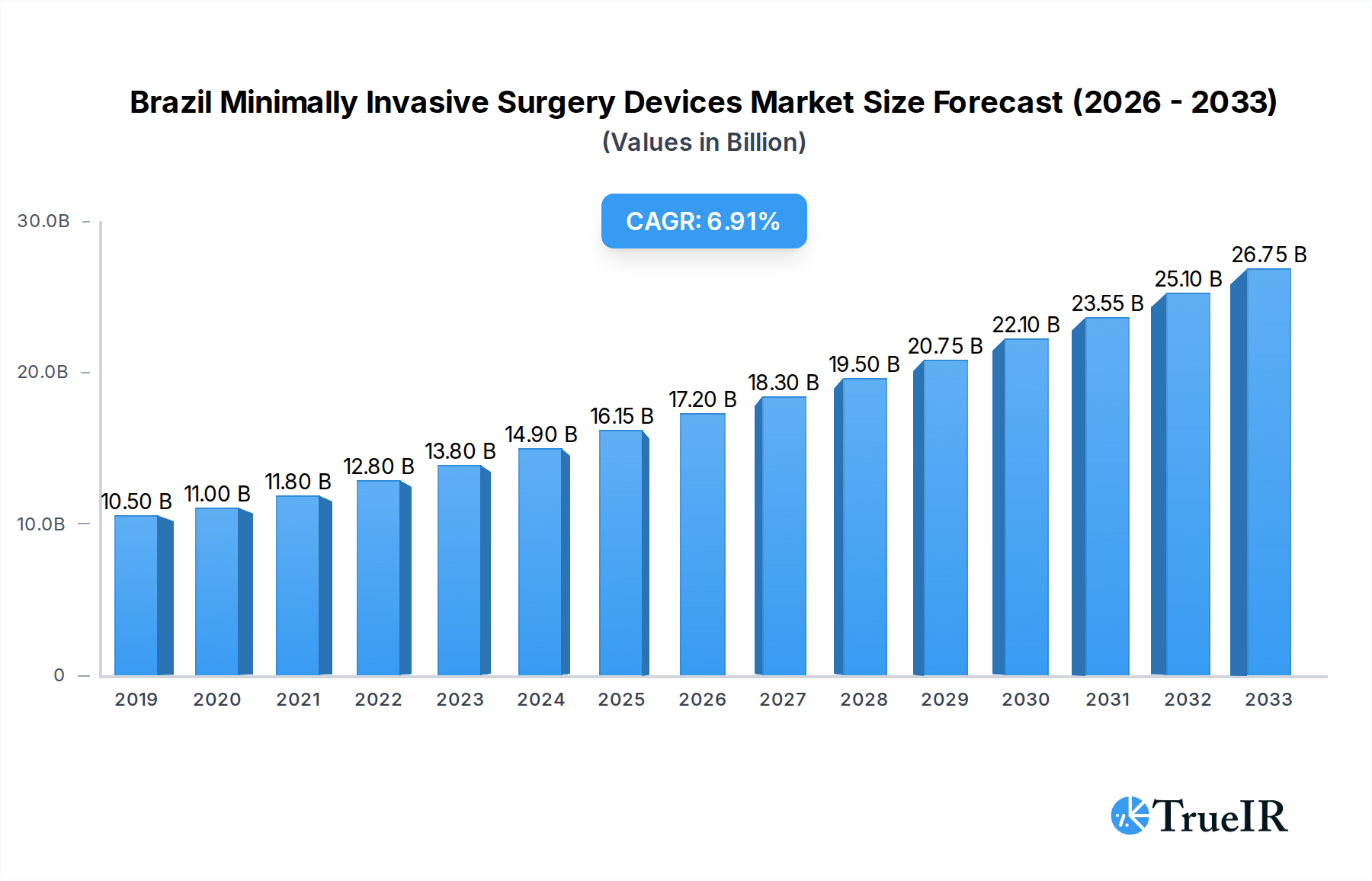
Brazil Minimally Invasive Surgery Devices Market Market Size (In Billion)

Several overarching trends are shaping the Brazilian MIS Devices Market. Technological advancements, such as the integration of artificial intelligence and robotics in surgical systems, are enhancing precision and improving patient outcomes. The increasing focus on patient safety and cost-effectiveness in healthcare is also a significant catalyst, as MIS procedures often lead to shorter hospital stays and lower overall healthcare expenditures. Key players like Medtronic Plc, Stryker Corporation, and Intuitive Surgical Inc. are actively investing in research and development to introduce innovative products and expand their market presence in Brazil. While the market exhibits strong growth potential, potential challenges such as stringent regulatory approvals and the high initial cost of advanced MIS equipment could present some restraints. However, the overall outlook remains highly positive, driven by a confluence of technological innovation, increasing healthcare expenditure, and a growing patient preference for minimally invasive surgical options.
Brazil Minimally Invasive Surgery Devices Market Company Market Share

Brazil Minimally Invasive Surgery Devices Market: Market Size, Trends, and Forecast 2025-2033
This comprehensive report provides an in-depth analysis of the Brazil Minimally Invasive Surgery Devices Market, a rapidly expanding sector driven by advancements in medical technology and an increasing demand for less invasive procedures. The study covers the period from 2019 to 2033, with a base year of 2025, offering detailed insights into market structure, key trends, dominant segments, product innovations, growth drivers, challenges, and a robust competitive landscape. We project the market to reach an estimated value of XX billion in 2025 and grow at a Compound Annual Growth Rate (CAGR) of XX% during the forecast period of 2025–2033. This report is essential for stakeholders seeking to understand the dynamics and future trajectory of the Brazilian minimally invasive surgery devices market.
Brazil Minimally Invasive Surgery Devices Market Market Structure & Competitive Landscape
The Brazil Minimally Invasive Surgery Devices Market is characterized by a moderately concentrated structure, with a significant presence of both global conglomerates and specialized local players. Innovation remains a primary driver, fueled by continuous research and development in areas such as robotics, advanced imaging, and novel instrument design. Regulatory impacts, particularly from Agência Nacional de Vigilância Sanitária (ANVISA), play a crucial role in market access and product approval processes, ensuring patient safety and efficacy. The threat of product substitutes, while present, is mitigated by the inherent benefits of MIS, such as reduced recovery times and improved patient outcomes. End-user segmentation across various medical specialties is diverse, influencing product development and marketing strategies. Mergers and acquisitions (M&A) are also observed, as larger companies seek to expand their portfolios and market reach. For instance, the market has witnessed XX M&A deals in the historical period (2019-2024) valued at approximately XX billion, indicating a consolidation trend. Key players are actively investing in R&D, contributing to an estimated XX% of market revenue stemming from new product launches within the past three years.
Brazil Minimally Invasive Surgery Devices Market Market Trends & Opportunities
The Brazil Minimally Invasive Surgery Devices Market is poised for substantial growth, driven by a confluence of technological advancements, shifting patient preferences, and increasing healthcare infrastructure development. The market size is projected to expand from an estimated XX billion in 2025 to XX billion by 2033, exhibiting a robust CAGR of XX%. Technological shifts are at the forefront, with the integration of artificial intelligence (AI) in surgical planning and execution, the growing adoption of robotic-assisted surgery platforms, and the development of smaller, more sophisticated endoscopic and laparoscopic instruments. Consumer preferences are increasingly leaning towards MIS due to reduced pain, shorter hospital stays, and faster return to daily activities, directly impacting the demand for these advanced devices. Competitive dynamics are intensifying, with companies focusing on product differentiation, strategic partnerships, and market penetration strategies. The market penetration rate for minimally invasive procedures is estimated to be around XX% in 2025, with significant room for growth as awareness and accessibility increase. Opportunities abound in expanding the application of MIS across emerging medical specialties and developing cost-effective solutions for wider accessibility, especially in remote regions of Brazil. The digital transformation in healthcare, including telemedicine and remote surgical training, also presents a significant avenue for market expansion and enhanced service delivery. The increasing prevalence of chronic diseases and an aging population further bolster the demand for efficient and less disruptive surgical interventions.
Dominant Markets & Segments in Brazil Minimally Invasive Surgery Devices Market
The Brazil Minimally Invasive Surgery Devices Market exhibits dominance across several key segments and applications, reflecting the widespread adoption and evolving utility of these technologies. In terms of product categories, Endoscopic and Laparoscopic Devices are anticipated to maintain a leading position, accounting for an estimated XX% of the total market revenue in 2025. This dominance is attributed to their broad applicability across numerous surgical specialties and their continuous evolution with enhanced imaging and instrument capabilities. Monitoring and Visualization Devices also represent a significant and growing segment, crucial for providing surgeons with real-time data and visual feedback during procedures, with an estimated market share of XX%.
Across applications, the Cardiovascular and Gastrointestinal segments are expected to lead market growth. The cardiovascular segment, driven by the increasing incidence of heart disease and the preference for minimally invasive cardiac surgeries (MICS), is projected to contribute XX% to the market by 2033. Similarly, the gastrointestinal segment, fueled by the rising prevalence of conditions like obesity, GERD, and IBD, is a key growth driver, estimated at XX% of the market. The Orthopedic segment is also experiencing substantial expansion due to advancements in arthroscopic surgery for joint repair and replacement.
Key growth drivers for these dominant segments include:
- Infrastructure Development: Expansion of specialized surgical centers and hospitals equipped with advanced MIS technology.
- Policy Support: Government initiatives promoting adoption of advanced medical technologies and patient care improvements.
- Technological Advancements: Continuous innovation in robotics, imaging, and instrumentation specifically for these applications.
- Surgeon Training and Education: Increased availability of training programs for MIS techniques, leading to wider adoption.
- Cost-Effectiveness: While initial investment can be high, the long-term benefits of reduced hospital stays and faster recovery contribute to overall cost-effectiveness in many MIS procedures.
The Urological and Gynecological segments are also showing robust growth, driven by the increasing demand for less invasive treatment options for conditions like prostate cancer, kidney stones, and gynecological disorders. The Aesthetic application, while a smaller segment currently, is expected to witness considerable growth driven by the rising popularity of minimally invasive cosmetic procedures.
Brazil Minimally Invasive Surgery Devices Market Product Analysis
Product innovation in the Brazil Minimally Invasive Surgery Devices Market is rapidly advancing, with a focus on enhancing precision, miniaturization, and user-friendliness. Handheld Instruments are evolving with ergonomic designs and advanced materials for improved dexterity. Guiding Devices are incorporating sophisticated navigation systems for more accurate targeting. Electrosurgical Devices are seeing innovations in energy delivery for enhanced tissue cutting and coagulation with minimal collateral damage. Endoscopic and Laparoscopic Devices are at the forefront of technological integration, featuring high-definition imaging, articulation capabilities, and modular designs. Monitoring and Visualization Devices are benefiting from AI-powered analytics and integrated imaging solutions. Ablation and Laser Based Devices are being refined for targeted lesion removal and tissue treatment. These advancements collectively aim to improve surgical outcomes, reduce patient trauma, and expand the scope of minimally invasive interventions.
Key Drivers, Barriers & Challenges in Brazil Minimally Invasive Surgery Devices Market
Key drivers propelling the Brazil Minimally Invasive Surgery Devices Market include significant technological advancements, such as the increasing integration of robotics and AI in surgical procedures, leading to enhanced precision and reduced invasiveness. Economic factors, including a growing middle class with higher disposable incomes and increased healthcare expenditure, also contribute to market growth. Supportive government policies aimed at improving healthcare access and promoting the adoption of advanced medical technologies are crucial. Furthermore, the rising prevalence of chronic diseases and an aging population necessitate more efficient and less traumatic treatment options, driving demand for MIS devices.
Key challenges impacting market growth include the high initial cost of advanced MIS equipment, which can be a barrier for smaller healthcare facilities and public hospitals. Regulatory hurdles and the lengthy approval processes by ANVISA, while ensuring safety, can slow down market entry for new products. Supply chain complexities, particularly for imported components, can lead to delays and increased costs. Intense competition among global and local players also presents a challenge, driving down profit margins. Moreover, the need for specialized training for surgeons and medical staff to effectively utilize these complex devices requires substantial investment in education and development.
Growth Drivers in the Brazil Minimally Invasive Surgery Devices Market Market
Several key factors are fueling the growth of the Brazil Minimally Invasive Surgery Devices Market. Technological innovation is paramount, with advancements in robotic surgery, high-definition imaging, and miniaturized instruments enabling more complex procedures. The economic landscape, marked by increasing healthcare spending and a growing private insurance sector, is making MIS more accessible. Favorable government policies, such as those encouraging the adoption of advanced medical technologies and improving healthcare infrastructure, are also significant drivers. The demographic shift towards an aging population and the rising incidence of lifestyle-related diseases are creating a sustained demand for less invasive and faster-recovery surgical options.
Challenges Impacting Brazil Minimally Invasive Surgery Devices Market Growth
The growth of the Brazil Minimally Invasive Surgery Devices Market faces several significant challenges. The substantial capital investment required for sophisticated MIS equipment, including robotic systems, can be prohibitive for many healthcare providers, particularly in underserved regions. Navigating the regulatory framework of ANVISA, while essential for patient safety, can lead to prolonged product approval timelines and increased compliance costs. Supply chain disruptions, influenced by global economic conditions and logistics, can impact the availability and cost of essential components and finished devices. The competitive landscape is also intensifying, with established players and emerging entrants vying for market share, potentially leading to price pressures. Furthermore, the continuous need for specialized training and upskilling of healthcare professionals to proficiently operate and maintain these advanced technologies presents an ongoing challenge.
Key Players Shaping the Brazil Minimally Invasive Surgery Devices Market Market
- Smith & Nephew
- Stryker Corporation
- Koninklijke Philips NV
- Intuitive Surgical Inc
- Medtronic Plc
- Siemens Healthineers
- GE Healthcare
- Zimmer Biomet
- Olympus Corporation
- Abbott Laboratories
Significant Brazil Minimally Invasive Surgery Devices Market Industry Milestones
- June 2022: GC Aesthetics, Inc. announced expansion goals for the Brazilian market, planning to commercialize all its women's healthcare aesthetic and reconstructive solutions.
- January 2022: Spinologics Inc. and Importek launched Cervision, an upper-extremity patient positioning device for cervical spine surgery, in Brazil.
- January 2022: Cervision received approval from Brazil's medical device regulatory body, Agência Nacional de Vigilância Sanitária (ANVISA).
Future Outlook for Brazil Minimally Invasive Surgery Devices Market Market
The future outlook for the Brazil Minimally Invasive Surgery Devices Market is exceptionally promising, driven by sustained technological innovation and a growing acceptance of less invasive surgical approaches. Strategic opportunities lie in the further development and adoption of robotic-assisted surgery, AI-driven diagnostic and surgical planning tools, and advanced endoscopic technologies. The market's potential is also boosted by the increasing focus on value-based healthcare, where MIS devices offer significant long-term cost savings through reduced hospital stays and faster patient recovery. Continued investment in healthcare infrastructure, coupled with supportive government initiatives and an expanding private healthcare sector, will further catalyze market expansion. The increasing demand for specialized procedures in areas like cardiology, orthopedics, and oncology will create a robust demand for sophisticated MIS solutions, positioning Brazil as a key growth market in the global minimally invasive surgery landscape.
Brazil Minimally Invasive Surgery Devices Market Segmentation
-
1. Products
- 1.1. Handheld Instruments
- 1.2. Guiding Devices
- 1.3. Electrosurgical Devices
- 1.4. Endoscopic and Laproscopic Devices
- 1.5. Monitoring and Visualization Devices
- 1.6. Ablation and Laser Based Devices
- 1.7. Others
-
2. Application
- 2.1. Aesthetic
- 2.2. Cardiovascular
- 2.3. Gastrointestinal
- 2.4. Gynecological
- 2.5. Orthopedic
- 2.6. Urological
- 2.7. Other Applications
Brazil Minimally Invasive Surgery Devices Market Segmentation By Geography
- 1. Brazil
Brazil Minimally Invasive Surgery Devices Market Regional Market Share

Geographic Coverage of Brazil Minimally Invasive Surgery Devices Market
Brazil Minimally Invasive Surgery Devices Market REPORT HIGHLIGHTS
| Aspects | Details |
|---|---|
| Study Period | 2020-2034 |
| Base Year | 2025 |
| Estimated Year | 2026 |
| Forecast Period | 2026-2034 |
| Historical Period | 2020-2025 |
| Growth Rate | CAGR of 16.1% from 2020-2034 |
| Segmentation |
|
Table of Contents
- 1. Introduction
- 1.1. Research Scope
- 1.2. Market Segmentation
- 1.3. Research Objective
- 1.4. Definitions and Assumptions
- 2. Executive Summary
- 2.1. Market Snapshot
- 3. Market Dynamics
- 3.1. Market Drivers
- 3.2. Market Restrains
- 3.3. Market Trends
- 3.4. Market Opportunities
- 4. Market Factor Analysis
- 4.1. Porters Five Forces
- 4.1.1. Bargaining Power of Suppliers
- 4.1.2. Bargaining Power of Buyers
- 4.1.3. Threat of New Entrants
- 4.1.4. Threat of Substitutes
- 4.1.5. Competitive Rivalry
- 4.2. PESTEL analysis
- 4.3. BCG Analysis
- 4.3.1. Stars (High Growth, High Market Share)
- 4.3.2. Cash Cows (Low Growth, High Market Share)
- 4.3.3. Question Mark (High Growth, Low Market Share)
- 4.3.4. Dogs (Low Growth, Low Market Share)
- 4.4. Ansoff Matrix Analysis
- 4.5. Supply Chain Analysis
- 4.6. Regulatory Landscape
- 4.7. Current Market Potential and Opportunity Assessment (TAM–SAM–SOM Framework)
- 4.8. TIR Analyst Note
- 4.1. Porters Five Forces
- 5. Market Analysis, Insights and Forecast 2021-2033
- 5.1. Market Analysis, Insights and Forecast - by Products
- 5.1.1. Handheld Instruments
- 5.1.2. Guiding Devices
- 5.1.3. Electrosurgical Devices
- 5.1.4. Endoscopic and Laproscopic Devices
- 5.1.5. Monitoring and Visualization Devices
- 5.1.6. Ablation and Laser Based Devices
- 5.1.7. Others
- 5.2. Market Analysis, Insights and Forecast - by Application
- 5.2.1. Aesthetic
- 5.2.2. Cardiovascular
- 5.2.3. Gastrointestinal
- 5.2.4. Gynecological
- 5.2.5. Orthopedic
- 5.2.6. Urological
- 5.2.7. Other Applications
- 5.3. Market Analysis, Insights and Forecast - by Region
- 5.3.1. Brazil
- 5.1. Market Analysis, Insights and Forecast - by Products
- 6. Brazil Minimally Invasive Surgery Devices Market Analysis, Insights and Forecast, 2021-2033
- 6.1. Market Analysis, Insights and Forecast - by Products
- 6.1.1. Handheld Instruments
- 6.1.2. Guiding Devices
- 6.1.3. Electrosurgical Devices
- 6.1.4. Endoscopic and Laproscopic Devices
- 6.1.5. Monitoring and Visualization Devices
- 6.1.6. Ablation and Laser Based Devices
- 6.1.7. Others
- 6.2. Market Analysis, Insights and Forecast - by Application
- 6.2.1. Aesthetic
- 6.2.2. Cardiovascular
- 6.2.3. Gastrointestinal
- 6.2.4. Gynecological
- 6.2.5. Orthopedic
- 6.2.6. Urological
- 6.2.7. Other Applications
- 6.1. Market Analysis, Insights and Forecast - by Products
- 7. Competitive Analysis
- 7.1. Company Profiles
- 7.1.1 Smith & Nephew
- 7.1.1.1. Company Overview
- 7.1.1.2. Products
- 7.1.1.3. Company Financials
- 7.1.1.4. SWOT Analysis
- 7.1.2 Stryker Corporation
- 7.1.2.1. Company Overview
- 7.1.2.2. Products
- 7.1.2.3. Company Financials
- 7.1.2.4. SWOT Analysis
- 7.1.3 Koninklijke Philips NV
- 7.1.3.1. Company Overview
- 7.1.3.2. Products
- 7.1.3.3. Company Financials
- 7.1.3.4. SWOT Analysis
- 7.1.4 Intuitive Surgical Inc
- 7.1.4.1. Company Overview
- 7.1.4.2. Products
- 7.1.4.3. Company Financials
- 7.1.4.4. SWOT Analysis
- 7.1.5 Medtronic Plc
- 7.1.5.1. Company Overview
- 7.1.5.2. Products
- 7.1.5.3. Company Financials
- 7.1.5.4. SWOT Analysis
- 7.1.6 Siemens Healthineers
- 7.1.6.1. Company Overview
- 7.1.6.2. Products
- 7.1.6.3. Company Financials
- 7.1.6.4. SWOT Analysis
- 7.1.7 GE Healthcare
- 7.1.7.1. Company Overview
- 7.1.7.2. Products
- 7.1.7.3. Company Financials
- 7.1.7.4. SWOT Analysis
- 7.1.8 Zimmer Biomet
- 7.1.8.1. Company Overview
- 7.1.8.2. Products
- 7.1.8.3. Company Financials
- 7.1.8.4. SWOT Analysis
- 7.1.9 Olympus Corporation
- 7.1.9.1. Company Overview
- 7.1.9.2. Products
- 7.1.9.3. Company Financials
- 7.1.9.4. SWOT Analysis
- 7.1.10 Abbott Laboratories
- 7.1.10.1. Company Overview
- 7.1.10.2. Products
- 7.1.10.3. Company Financials
- 7.1.10.4. SWOT Analysis
- 7.1.1 Smith & Nephew
- 7.2. Market Entropy
- 7.2.1 Company's Key Areas Served
- 7.2.2 Recent Developments
- 7.3. Company Market Share Analysis 2025
- 7.3.1 Top 5 Companies Market Share Analysis
- 7.3.2 Top 3 Companies Market Share Analysis
- 7.4. List of Potential Customers
- 8. Research Methodology
List of Figures
- Figure 1: Brazil Minimally Invasive Surgery Devices Market Revenue Breakdown (billion, %) by Product 2025 & 2033
- Figure 2: Brazil Minimally Invasive Surgery Devices Market Share (%) by Company 2025
List of Tables
- Table 1: Brazil Minimally Invasive Surgery Devices Market Revenue billion Forecast, by Products 2020 & 2033
- Table 2: Brazil Minimally Invasive Surgery Devices Market Volume K units Forecast, by Products 2020 & 2033
- Table 3: Brazil Minimally Invasive Surgery Devices Market Revenue billion Forecast, by Application 2020 & 2033
- Table 4: Brazil Minimally Invasive Surgery Devices Market Volume K units Forecast, by Application 2020 & 2033
- Table 5: Brazil Minimally Invasive Surgery Devices Market Revenue billion Forecast, by Region 2020 & 2033
- Table 6: Brazil Minimally Invasive Surgery Devices Market Volume K units Forecast, by Region 2020 & 2033
- Table 7: Brazil Minimally Invasive Surgery Devices Market Revenue billion Forecast, by Products 2020 & 2033
- Table 8: Brazil Minimally Invasive Surgery Devices Market Volume K units Forecast, by Products 2020 & 2033
- Table 9: Brazil Minimally Invasive Surgery Devices Market Revenue billion Forecast, by Application 2020 & 2033
- Table 10: Brazil Minimally Invasive Surgery Devices Market Volume K units Forecast, by Application 2020 & 2033
- Table 11: Brazil Minimally Invasive Surgery Devices Market Revenue billion Forecast, by Country 2020 & 2033
- Table 12: Brazil Minimally Invasive Surgery Devices Market Volume K units Forecast, by Country 2020 & 2033
Frequently Asked Questions
1. What is the projected Compound Annual Growth Rate (CAGR) of the Brazil Minimally Invasive Surgery Devices Market?
The projected CAGR is approximately 16.1%.
2. Which companies are prominent players in the Brazil Minimally Invasive Surgery Devices Market?
Key companies in the market include Smith & Nephew, Stryker Corporation, Koninklijke Philips NV, Intuitive Surgical Inc, Medtronic Plc, Siemens Healthineers, GE Healthcare, Zimmer Biomet, Olympus Corporation, Abbott Laboratories.
3. What are the main segments of the Brazil Minimally Invasive Surgery Devices Market?
The market segments include Products, Application.
4. Can you provide details about the market size?
The market size is estimated to be USD 94.45 billion as of 2022.
5. What are some drivers contributing to market growth?
Higher Acceptance Rate of Minimally-invasive Surgeries over Traditional Surgeries; Increasing Prevalence of Lifestyle-related and Chronic Disorders; Technological Advancements.
6. What are the notable trends driving market growth?
Aesthetics Segment is Expected to Witness Growth Over the Forecast Period.
7. Are there any restraints impacting market growth?
Lack of Experienced Professionals; Uncertain Regulatory Framework.
8. Can you provide examples of recent developments in the market?
In June 2022, GC Aesthetics, Inc., a privately-held medical technology company offering women's healthcare aesthetic and reconstructive solutions, announced its expansion goals for the Brazilian market. The company planned to commercialize all GC Aesthetics products and solutions in Brazil.
9. What pricing options are available for accessing the report?
Pricing options include single-user, multi-user, and enterprise licenses priced at USD 3800, USD 4500, and USD 5800 respectively.
10. Is the market size provided in terms of value or volume?
The market size is provided in terms of value, measured in billion and volume, measured in K units.
11. Are there any specific market keywords associated with the report?
Yes, the market keyword associated with the report is "Brazil Minimally Invasive Surgery Devices Market," which aids in identifying and referencing the specific market segment covered.
12. How do I determine which pricing option suits my needs best?
The pricing options vary based on user requirements and access needs. Individual users may opt for single-user licenses, while businesses requiring broader access may choose multi-user or enterprise licenses for cost-effective access to the report.
13. Are there any additional resources or data provided in the Brazil Minimally Invasive Surgery Devices Market report?
While the report offers comprehensive insights, it's advisable to review the specific contents or supplementary materials provided to ascertain if additional resources or data are available.
14. How can I stay updated on further developments or reports in the Brazil Minimally Invasive Surgery Devices Market?
To stay informed about further developments, trends, and reports in the Brazil Minimally Invasive Surgery Devices Market, consider subscribing to industry newsletters, following relevant companies and organizations, or regularly checking reputable industry news sources and publications.
Methodology
Step 1 - Identification of Relevant Samples Size from Population Database



Step 2 - Approaches for Defining Global Market Size (Value, Volume* & Price*)

Note*: In applicable scenarios
Step 3 - Data Sources
Primary Research
- Web Analytics
- Survey Reports
- Research Institute
- Latest Research Reports
- Opinion Leaders
Secondary Research
- Annual Reports
- White Paper
- Latest Press Release
- Industry Association
- Paid Database
- Investor Presentations

Step 4 - Data Triangulation
Involves using different sources of information in order to increase the validity of a study
These sources are likely to be stakeholders in a program - participants, other researchers, program staff, other community members, and so on.
Then we put all data in single framework & apply various statistical tools to find out the dynamic on the market.
During the analysis stage, feedback from the stakeholder groups would be compared to determine areas of agreement as well as areas of divergence


