Key Insights
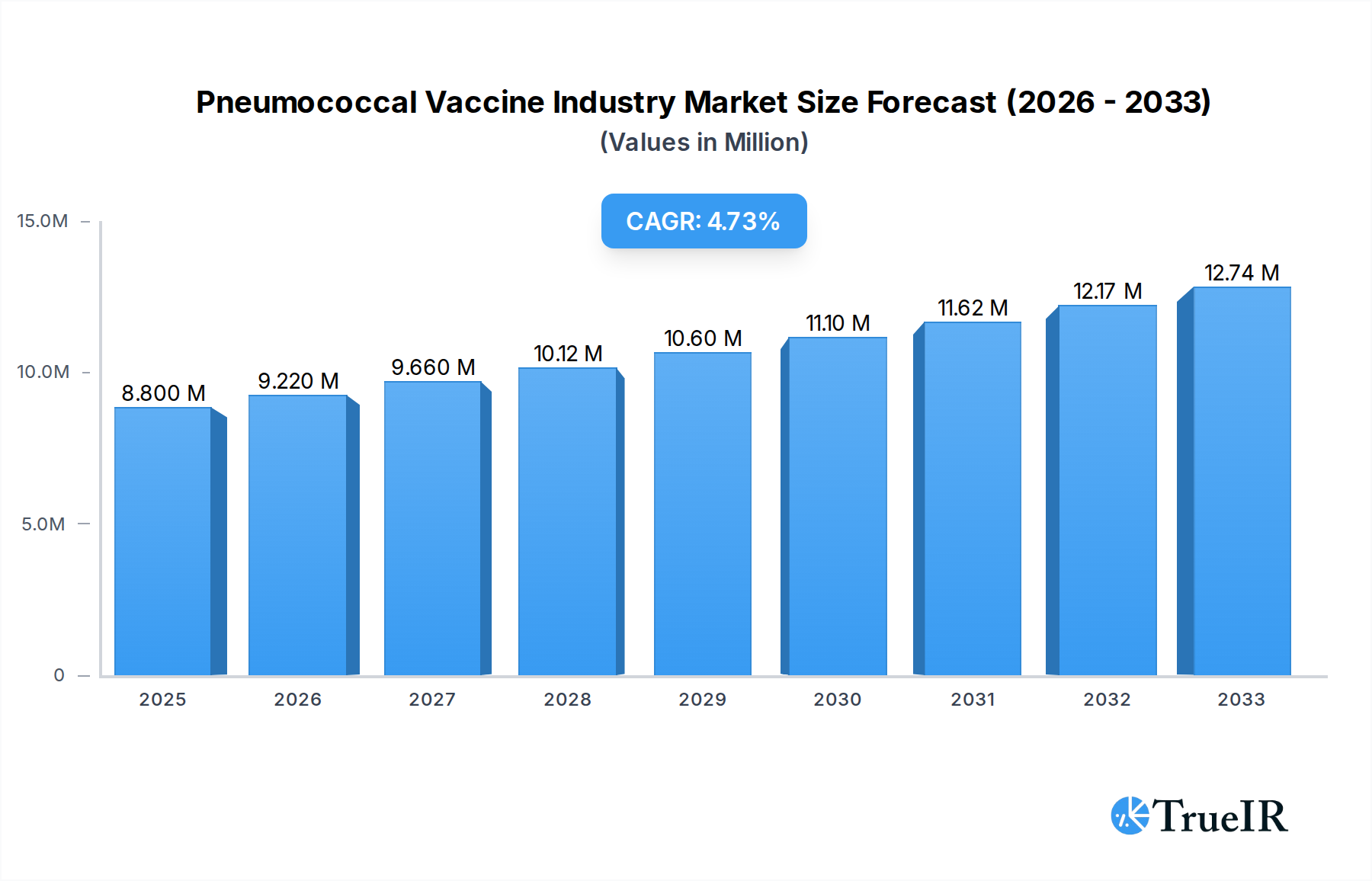
The global Pneumococcal Vaccine market is poised for significant expansion, currently valued at approximately $8.80 million in 2025 and projected to grow at a robust Compound Annual Growth Rate (CAGR) of 4.83% through 2033. This growth is primarily propelled by a confluence of factors, including the increasing global burden of pneumococcal diseases, particularly among vulnerable populations like infants, the elderly, and immunocompromised individuals. Rising healthcare expenditure, enhanced public health initiatives, and government-led vaccination campaigns play a pivotal role in driving market penetration. Furthermore, continuous advancements in vaccine technology, leading to the development of more effective and broader-spectrum vaccines, are fueling market demand. The market is segmented by vaccine type, with pneumococcal conjugate vaccines holding a dominant share due to their superior efficacy against a wider range of serotypes compared to polysaccharide vaccines. Key product types such as Prevnar 13 and Synflorix are leading the market, while established players like Pfizer, GSK, and Sanofi are at the forefront of innovation and supply. Distribution channels, including partnerships with distribution companies, NGOs, and government authorities, are crucial for ensuring widespread access to these life-saving vaccines.
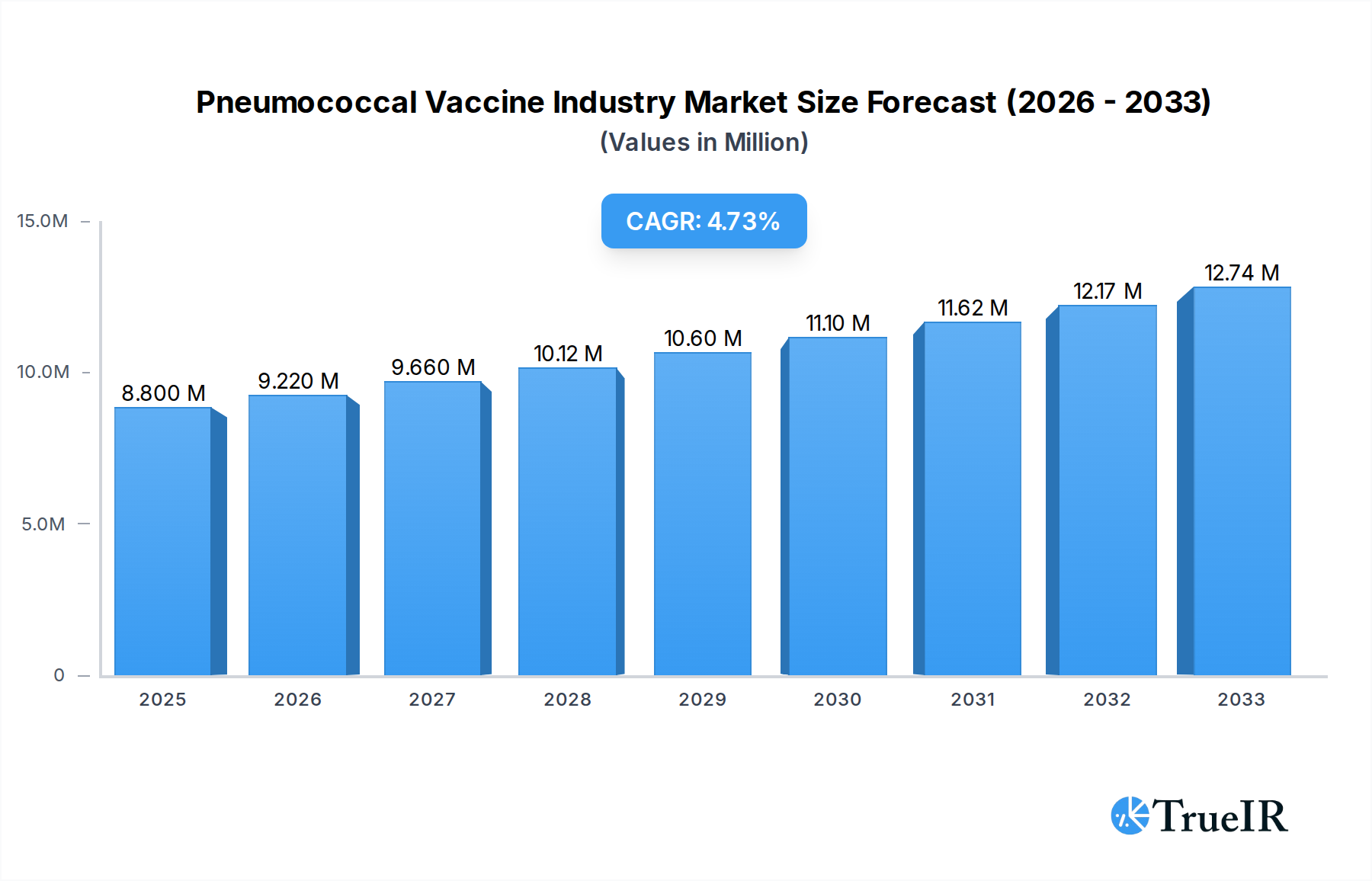
Pneumococcal Vaccine Industry Market Size (In Million)

The market's growth trajectory is further supported by the increasing awareness regarding the preventive benefits of pneumococcal vaccination in reducing morbidity and mortality associated with pneumonia, meningitis, and bacteremia. Emerging economies, particularly in the Asia Pacific region, are expected to witness substantial growth owing to expanding healthcare infrastructure, rising disposable incomes, and proactive government policies aimed at improving immunization rates. However, challenges such as the high cost of certain advanced vaccines, stringent regulatory approvals, and the need for robust cold-chain infrastructure in remote areas could pose restraints. Despite these hurdles, the unwavering focus on disease prevention and the continuous efforts by leading pharmaceutical companies to develop and expand access to pneumococcal vaccines underscore a positive and promising outlook for the global market. The market's strategic importance is amplified by the presence of key players like Walvax Biotechnology, CSL, and Merck KGaA, who are actively investing in research and development to address unmet medical needs and broaden vaccine portfolios.
Pneumococcal Vaccine Industry Company Market Share

Pneumococcal Vaccine Industry Market Analysis: Forecast to 2033
This comprehensive report delves into the dynamic pneumococcal vaccine market, a critical segment of the global immunization landscape. Analyzing historical data from 2019-2024 and providing robust forecasts through 2033, this study offers unparalleled insights into market size, trends, and opportunities for pneumococcal conjugate vaccine (PCV) and pneumococcal polysaccharide vaccine (PPSV). Our analysis covers key product segments like Prevnar 13, Synflorix, and Pneumovax23, and examines distribution channels including distribution partner companies, non-governmental organizations (NGOs), and government authorities. Discover the competitive strategies of leading players such as Pfizer Inc, GSK PLC, Sanofi S.A., CSL Ltd, Merck KGaA, Serum Institute of India Pvt Ltd, Walvax Biotechnology Co Ltd, and Beijing Minhai Biotechnology Co Ltd. This report is essential for stakeholders seeking to navigate the evolving pneumococcal disease prevention market.
Pneumococcal Vaccine Industry Market Structure & Competitive Landscape
The pneumococcal vaccine industry is characterized by a moderately concentrated market structure, dominated by a few key multinational pharmaceutical corporations with extensive research and development capabilities and established distribution networks. Innovation drivers are primarily focused on developing vaccines with broader serotype coverage, improved immunogenicity, and enhanced safety profiles, aiming to address the persistent burden of invasive pneumococcal disease (IPD). Regulatory impacts, driven by public health initiatives and national immunization programs, significantly influence market access and demand. Product substitutes, while limited in the context of highly effective vaccines, can emerge from advancements in alternative antimicrobial strategies or novel therapeutic approaches to IPD treatment, although vaccines remain the primary preventive measure. End-user segmentation largely revolves around pediatric and adult populations, with specific formulations tailored to each group's immunological needs and risk profiles. Mergers and acquisitions (M&A) trends, while not as frequent as in some other pharmaceutical sectors, are driven by strategic imperatives to expand product portfolios, gain access to new technologies, or consolidate market share. For instance, a recent acquisition might involve a smaller biotechnology firm with a promising pipeline targeting novel pneumococcal serotypes being integrated into a larger player’s vaccine division, thereby enhancing the latter’s competitive standing. The market concentration ratio for the top three players is estimated to be around 70%, reflecting their significant market share. M&A volumes have averaged approximately $500 Million annually over the historical period, indicating strategic consolidation rather than aggressive expansion through acquisition.
Pneumococcal Vaccine Industry Market Trends & Opportunities
The global pneumococcal vaccine market is poised for substantial growth, projected to expand at a robust Compound Annual Growth Rate (CAGR) of approximately 7.8% from the base year of 2025 through 2033. This upward trajectory is fueled by a confluence of factors, including rising global vaccination rates, increasing awareness of pneumococcal disease prevention, and the continuous development of next-generation vaccines. The market size, estimated to be in the range of $12,000 Million to $15,000 Million for 2025, is expected to witness significant expansion due to the persistent threat of Streptococcus pneumoniae infections, which cause pneumonia, meningitis, and sepsis. Technological shifts are a major driving force, with ongoing research and development focused on expanding serotype coverage in pneumococcal conjugate vaccines (PCVs) beyond the current 13 or 15 serotypes, aiming to combat emerging strains and reduce the incidence of antibiotic-resistant infections. This includes advancements in vaccine platforms, such as the development of novel adjuvants and conjugation techniques, to enhance immune responses, particularly in vulnerable populations like the elderly and immunocompromised individuals.
Consumer preferences are increasingly leaning towards vaccines offering broader protection and improved efficacy. Public health recommendations and the inclusion of pneumococcal vaccines in routine immunization schedules by major health organizations like the World Health Organization (WHO) and national health agencies play a pivotal role in shaping these preferences and driving market demand. For example, the widespread adoption of PCVs in infant immunization programs has become a cornerstone of global child health. Competitive dynamics are characterized by intense innovation and strategic partnerships among leading pharmaceutical companies. The pursuit of differentiated products with superior serotype coverage, such as investigational 20-valent and 21-valent PCVs, is a key competitive strategy. The market penetration rate for PCVs in developed countries is already high, exceeding 90% for infant immunization, while emerging markets present significant growth opportunities as vaccination programs expand and healthcare infrastructure improves. Opportunities also lie in the development of vaccines for specific high-risk adult populations and the exploration of novel delivery systems to improve vaccine accessibility and compliance. The increasing prevalence of co-morbidities in aging populations further amplifies the demand for effective pneumococcal disease prevention.
Dominant Markets & Segments in Pneumococcal Vaccine Industry
The Pneumococcal Vaccine Industry exhibits distinct regional and segment dominance, driven by public health priorities, economic development, and healthcare infrastructure. Globally, the Pneumococcal conjugate vaccine (PCV) segment is the dominant force, accounting for an estimated 85% of the total market value in 2025. This dominance is attributed to its critical role in pediatric immunization programs, which have demonstrably reduced the incidence of invasive pneumococcal disease in children under five. Within the PCV segment, Prevnar 13 by Pfizer Inc. and Synflorix by GSK PLC have historically held significant market share, although newer PCVs with broader serotype coverage are increasingly gaining traction.
Dominant Regions & Countries: North America and Europe currently represent the largest markets for pneumococcal vaccines, driven by well-established national immunization programs, high healthcare expenditure, and a strong focus on preventive healthcare. The United States, in particular, is a key market due to its large population and robust vaccination policies. However, the Asia-Pacific region is emerging as a significant growth engine, fueled by increasing government investments in public health, expanding vaccination coverage in populous countries like China and India, and a growing middle class with greater access to healthcare.
Key Growth Drivers & Market Dominance:
- Government Authorities: These are the primary purchasers and policymakers for pneumococcal vaccines, dictating their inclusion in national immunization schedules. Government mandates and procurement programs are critical for market penetration, especially in developing economies.
- Distribution Partner Companies: These entities play a crucial role in ensuring the efficient and widespread distribution of vaccines, particularly in remote areas or where direct government distribution may be challenging. Their logistics and supply chain expertise are vital for market access.
- Non-governmental Organizations (NGOs): NGOs often collaborate with governments and international health organizations to implement vaccination campaigns in underserved populations, thereby expanding the market reach of pneumococcal vaccines.
The Pneumococcal polysaccharide vaccine (PPSV), primarily represented by Pneumovax23, holds a significant share in the adult vaccination market, particularly for individuals aged 65 and older and those with chronic health conditions. While PCVs are increasingly being recommended for older adults, PPSV remains a vital tool for broader serotype coverage in this demographic.
Market Penetration and Future Outlook: The market penetration for PCVs in pediatric populations in developed nations is nearly saturated. However, opportunities exist for newer PCVs with expanded serotype coverage to gain market share and address remaining disease burdens. In emerging markets, significant growth potential remains as vaccination programs mature. The ongoing development of novel pneumococcal vaccines targeting an expanded range of serotypes, coupled with increasing public health awareness and governmental initiatives, will continue to shape the dominant segments and drive market expansion.
Pneumococcal Vaccine Industry Product Analysis
Product innovation in the pneumococcal vaccine industry is primarily driven by the pursuit of broader serotype coverage and improved immunogenicity. Advancements in conjugation technologies and adjuvant formulations are enabling the development of vaccines that target a greater number of Streptococcus pneumoniae serotypes, thereby offering enhanced protection against a wider spectrum of pneumococcal diseases. Products like Prevnar 20 and emerging investigational vaccines demonstrate this trend, aiming to combat both common and less frequent but severe serotypes. The competitive advantage lies in offering superior protection against invasive pneumococcal disease (IPD) and pneumococcal pneumonia, particularly in vulnerable populations such as infants, young children, and older adults. These technological advancements directly address unmet medical needs and align with global public health objectives for disease prevention.
Key Drivers, Barriers & Challenges in Pneumococcal Vaccine Industry
Key Drivers: The primary forces propelling the pneumococcal vaccine market include the persistent global burden of pneumococcal diseases, leading to significant morbidity and mortality, particularly in children and the elderly. Technological advancements in vaccine development, enabling broader serotype coverage and improved efficacy, are crucial. Furthermore, supportive government policies, national immunization programs, and initiatives by organizations like the WHO to promote widespread vaccination coverage are significant drivers. Economic factors, such as increasing healthcare expenditure in emerging economies and the cost-effectiveness of vaccination programs in preventing disease complications and reducing healthcare costs, also contribute to market growth.
Barriers & Challenges: Despite strong growth, the industry faces challenges. Regulatory hurdles and the stringent approval processes for new vaccines can extend time-to-market. Supply chain complexities, ensuring consistent availability and cold chain integrity, are critical, especially in remote or resource-limited settings. Competitive pressures among established players and emerging manufacturers, along with pricing sensitivities, can impact market dynamics. The emergence of antibiotic-resistant strains of Streptococcus pneumoniae presents an ongoing challenge, necessitating continuous vaccine innovation to maintain efficacy. Vaccine hesitancy, though less pronounced for pneumococcal vaccines compared to some others, can still pose a barrier in certain regions.
Growth Drivers in the Pneumococcal Vaccine Industry Market
Several key growth drivers are shaping the pneumococcal vaccine industry. The persistent and significant global burden of pneumococcal disease, including pneumonia, meningitis, and sepsis, remains a primary impetus for demand. Technological advancements in vaccine development are crucial; researchers are continuously innovating to introduce vaccines with broader serotype coverage, addressing a wider range of Streptococcus pneumoniae strains. This is particularly important in combating antibiotic-resistant bacteria. Economic growth in emerging markets is also a significant factor, leading to increased healthcare spending, expanding access to vaccination programs, and a growing demand for advanced healthcare solutions. Government mandates and the inclusion of pneumococcal vaccines in routine immunization schedules by national health authorities worldwide are pivotal, ensuring consistent demand and market penetration. International organizations like the WHO continue to advocate for and support vaccination initiatives, further bolstering growth.
Challenges Impacting Pneumococcal Vaccine Industry Growth
The pneumococcal vaccine industry is not without its challenges. Regulatory complexities and the lengthy, rigorous approval processes for new vaccines can delay market entry and increase development costs. Ensuring robust and resilient supply chains is paramount, as disruptions can lead to vaccine shortages, particularly in remote or underserved regions, impacting global health equity. Competitive pressures among established pharmaceutical giants and emerging players can lead to pricing challenges and intense marketing efforts. The potential for vaccine hesitancy, while perhaps less prevalent for pneumococcal vaccines than some others, can still hinder optimal uptake in certain populations. Furthermore, the continuous evolution of Streptococcus pneumoniae strains and the rise of antibiotic resistance necessitate ongoing research and development to maintain the effectiveness of existing vaccines and develop next-generation formulations, presenting a sustained challenge for the industry.
Key Players Shaping the Pneumococcal Vaccine Industry Market
- Pfizer Inc
- GSK PLC
- Sanofi S.A.
- CSL Ltd
- Merck KGaA
- Serum Institute of India Pvt Ltd
- Walvax Biotechnology Co Ltd
- Beijing Minhai Biotechnology Co Ltd
Significant Pneumococcal Vaccine Industry Industry Milestones
- April 2024: Merck presented the results of the phase 3 clinical trial STRIDE-10 for an investigational 21-valent pneumococcal conjugate vaccine developed for adults.
- March 2024: Vaxcyte completed enrollment for the phase-2 clinical trial to evaluate the 24-valent pneumococcal conjugate vaccine (PCV) candidate VAX-24 to eradicate invasive pneumococcal disease (IPD) in children.
Future Outlook for Pneumococcal Vaccine Industry Market
The future outlook for the pneumococcal vaccine industry is exceptionally promising, driven by sustained global demand for effective pneumococcal disease prevention. Continued innovation in vaccine technology, particularly the development of vaccines with broader serotype coverage and enhanced immunogenicity, will be a key growth catalyst. Emerging markets, with their expanding healthcare infrastructure and increasing vaccination rates, represent significant untapped potential. Furthermore, the growing emphasis on adult immunization and the need for robust protection against pneumococcal infections in aging and immunocompromised populations will fuel market expansion. Strategic collaborations, investment in R&D, and supportive government policies will further solidify the industry's trajectory, ensuring its vital role in global public health for the foreseeable future.
Pneumococcal Vaccine Industry Segmentation
-
1. Vaccine Type
- 1.1. Pneumococcal conjugate vaccine
- 1.2. Pneumococcal polysaccharide vaccine
-
2. Product Type
- 2.1. Prevnar 13
- 2.2. Synflorix
- 2.3. Pneumovax23
-
3. Distribution Channel
- 3.1. Distribution partner companies
- 3.2. Non-governmental Organizations
- 3.3. Government Authorities
Pneumococcal Vaccine Industry Segmentation By Geography
-
1. North America
- 1.1. United States
- 1.2. Canada
- 1.3. Mexico
-
2. Europe
- 2.1. Germany
- 2.2. United Kingdom
- 2.3. France
- 2.4. Italy
- 2.5. Spain
- 2.6. Rest of Europe
-
3. Asia Pacific
- 3.1. China
- 3.2. Japan
- 3.3. India
- 3.4. Australia
- 3.5. South Korea
- 3.6. Rest of Asia Pacific
-
4. Middle East and Africa
- 4.1. GCC
- 4.2. South Africa
- 4.3. Rest of Middle East and Africa
-
5. South America
- 5.1. Brazil
- 5.2. Argentina
- 5.3. Rest of South America
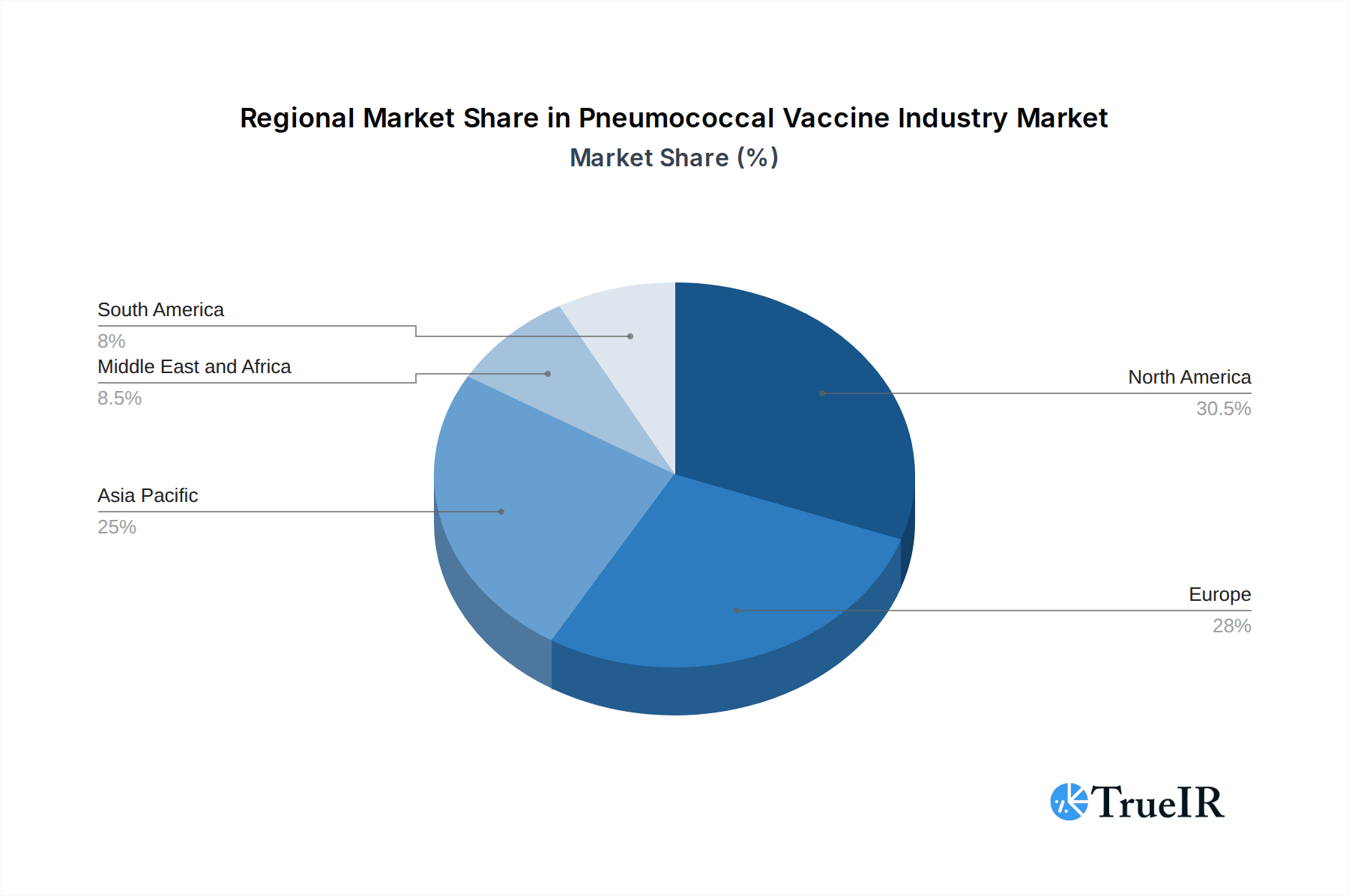
Pneumococcal Vaccine Industry Regional Market Share

Geographic Coverage of Pneumococcal Vaccine Industry
Pneumococcal Vaccine Industry REPORT HIGHLIGHTS
| Aspects | Details |
|---|---|
| Study Period | 2020-2034 |
| Base Year | 2025 |
| Estimated Year | 2026 |
| Forecast Period | 2026-2034 |
| Historical Period | 2020-2025 |
| Growth Rate | CAGR of 4.83% from 2020-2034 |
| Segmentation |
|
Table of Contents
- 1. Introduction
- 1.1. Research Scope
- 1.2. Market Segmentation
- 1.3. Research Objective
- 1.4. Definitions and Assumptions
- 2. Executive Summary
- 2.1. Market Snapshot
- 3. Market Dynamics
- 3.1. Market Drivers
- 3.2. Market Restrains
- 3.3. Market Trends
- 3.4. Market Opportunities
- 4. Market Factor Analysis
- 4.1. Porters Five Forces
- 4.1.1. Bargaining Power of Suppliers
- 4.1.2. Bargaining Power of Buyers
- 4.1.3. Threat of New Entrants
- 4.1.4. Threat of Substitutes
- 4.1.5. Competitive Rivalry
- 4.2. PESTEL analysis
- 4.3. BCG Analysis
- 4.3.1. Stars (High Growth, High Market Share)
- 4.3.2. Cash Cows (Low Growth, High Market Share)
- 4.3.3. Question Mark (High Growth, Low Market Share)
- 4.3.4. Dogs (Low Growth, Low Market Share)
- 4.4. Ansoff Matrix Analysis
- 4.5. Supply Chain Analysis
- 4.6. Regulatory Landscape
- 4.7. Current Market Potential and Opportunity Assessment (TAM–SAM–SOM Framework)
- 4.8. TIR Analyst Note
- 4.1. Porters Five Forces
- 5. Market Analysis, Insights and Forecast 2021-2033
- 5.1. Market Analysis, Insights and Forecast - by Vaccine Type
- 5.1.1. Pneumococcal conjugate vaccine
- 5.1.2. Pneumococcal polysaccharide vaccine
- 5.2. Market Analysis, Insights and Forecast - by Product Type
- 5.2.1. Prevnar 13
- 5.2.2. Synflorix
- 5.2.3. Pneumovax23
- 5.3. Market Analysis, Insights and Forecast - by Distribution Channel
- 5.3.1. Distribution partner companies
- 5.3.2. Non-governmental Organizations
- 5.3.3. Government Authorities
- 5.4. Market Analysis, Insights and Forecast - by Region
- 5.4.1. North America
- 5.4.2. Europe
- 5.4.3. Asia Pacific
- 5.4.4. Middle East and Africa
- 5.4.5. South America
- 5.1. Market Analysis, Insights and Forecast - by Vaccine Type
- 6. Global Pneumococcal Vaccine Industry Analysis, Insights and Forecast, 2021-2033
- 6.1. Market Analysis, Insights and Forecast - by Vaccine Type
- 6.1.1. Pneumococcal conjugate vaccine
- 6.1.2. Pneumococcal polysaccharide vaccine
- 6.2. Market Analysis, Insights and Forecast - by Product Type
- 6.2.1. Prevnar 13
- 6.2.2. Synflorix
- 6.2.3. Pneumovax23
- 6.3. Market Analysis, Insights and Forecast - by Distribution Channel
- 6.3.1. Distribution partner companies
- 6.3.2. Non-governmental Organizations
- 6.3.3. Government Authorities
- 6.1. Market Analysis, Insights and Forecast - by Vaccine Type
- 7. North America Pneumococcal Vaccine Industry Analysis, Insights and Forecast, 2020-2032
- 7.1. Market Analysis, Insights and Forecast - by Vaccine Type
- 7.1.1. Pneumococcal conjugate vaccine
- 7.1.2. Pneumococcal polysaccharide vaccine
- 7.2. Market Analysis, Insights and Forecast - by Product Type
- 7.2.1. Prevnar 13
- 7.2.2. Synflorix
- 7.2.3. Pneumovax23
- 7.3. Market Analysis, Insights and Forecast - by Distribution Channel
- 7.3.1. Distribution partner companies
- 7.3.2. Non-governmental Organizations
- 7.3.3. Government Authorities
- 7.1. Market Analysis, Insights and Forecast - by Vaccine Type
- 8. Europe Pneumococcal Vaccine Industry Analysis, Insights and Forecast, 2020-2032
- 8.1. Market Analysis, Insights and Forecast - by Vaccine Type
- 8.1.1. Pneumococcal conjugate vaccine
- 8.1.2. Pneumococcal polysaccharide vaccine
- 8.2. Market Analysis, Insights and Forecast - by Product Type
- 8.2.1. Prevnar 13
- 8.2.2. Synflorix
- 8.2.3. Pneumovax23
- 8.3. Market Analysis, Insights and Forecast - by Distribution Channel
- 8.3.1. Distribution partner companies
- 8.3.2. Non-governmental Organizations
- 8.3.3. Government Authorities
- 8.1. Market Analysis, Insights and Forecast - by Vaccine Type
- 9. Asia Pacific Pneumococcal Vaccine Industry Analysis, Insights and Forecast, 2020-2032
- 9.1. Market Analysis, Insights and Forecast - by Vaccine Type
- 9.1.1. Pneumococcal conjugate vaccine
- 9.1.2. Pneumococcal polysaccharide vaccine
- 9.2. Market Analysis, Insights and Forecast - by Product Type
- 9.2.1. Prevnar 13
- 9.2.2. Synflorix
- 9.2.3. Pneumovax23
- 9.3. Market Analysis, Insights and Forecast - by Distribution Channel
- 9.3.1. Distribution partner companies
- 9.3.2. Non-governmental Organizations
- 9.3.3. Government Authorities
- 9.1. Market Analysis, Insights and Forecast - by Vaccine Type
- 10. Middle East and Africa Pneumococcal Vaccine Industry Analysis, Insights and Forecast, 2020-2032
- 10.1. Market Analysis, Insights and Forecast - by Vaccine Type
- 10.1.1. Pneumococcal conjugate vaccine
- 10.1.2. Pneumococcal polysaccharide vaccine
- 10.2. Market Analysis, Insights and Forecast - by Product Type
- 10.2.1. Prevnar 13
- 10.2.2. Synflorix
- 10.2.3. Pneumovax23
- 10.3. Market Analysis, Insights and Forecast - by Distribution Channel
- 10.3.1. Distribution partner companies
- 10.3.2. Non-governmental Organizations
- 10.3.3. Government Authorities
- 10.1. Market Analysis, Insights and Forecast - by Vaccine Type
- 11. South America Pneumococcal Vaccine Industry Analysis, Insights and Forecast, 2020-2032
- 11.1. Market Analysis, Insights and Forecast - by Vaccine Type
- 11.1.1. Pneumococcal conjugate vaccine
- 11.1.2. Pneumococcal polysaccharide vaccine
- 11.2. Market Analysis, Insights and Forecast - by Product Type
- 11.2.1. Prevnar 13
- 11.2.2. Synflorix
- 11.2.3. Pneumovax23
- 11.3. Market Analysis, Insights and Forecast - by Distribution Channel
- 11.3.1. Distribution partner companies
- 11.3.2. Non-governmental Organizations
- 11.3.3. Government Authorities
- 11.1. Market Analysis, Insights and Forecast - by Vaccine Type
- 12. Competitive Analysis
- 12.1. Company Profiles
- 12.1.1 Walvax Biotechnology Co Ltd
- 12.1.1.1. Company Overview
- 12.1.1.2. Products
- 12.1.1.3. Company Financials
- 12.1.1.4. SWOT Analysis
- 12.1.2 CSL Ltd
- 12.1.2.1. Company Overview
- 12.1.2.2. Products
- 12.1.2.3. Company Financials
- 12.1.2.4. SWOT Analysis
- 12.1.3 Merck KGaA
- 12.1.3.1. Company Overview
- 12.1.3.2. Products
- 12.1.3.3. Company Financials
- 12.1.3.4. SWOT Analysis
- 12.1.4 Beijing Minhai Biotechnology Co Ltd*List Not Exhaustive
- 12.1.4.1. Company Overview
- 12.1.4.2. Products
- 12.1.4.3. Company Financials
- 12.1.4.4. SWOT Analysis
- 12.1.5 GSK PLC
- 12.1.5.1. Company Overview
- 12.1.5.2. Products
- 12.1.5.3. Company Financials
- 12.1.5.4. SWOT Analysis
- 12.1.6 Serum Institute of India Pvt Ltd
- 12.1.6.1. Company Overview
- 12.1.6.2. Products
- 12.1.6.3. Company Financials
- 12.1.6.4. SWOT Analysis
- 12.1.7 Sanofi S A
- 12.1.7.1. Company Overview
- 12.1.7.2. Products
- 12.1.7.3. Company Financials
- 12.1.7.4. SWOT Analysis
- 12.1.8 Pfizer Inc
- 12.1.8.1. Company Overview
- 12.1.8.2. Products
- 12.1.8.3. Company Financials
- 12.1.8.4. SWOT Analysis
- 12.1.1 Walvax Biotechnology Co Ltd
- 12.2. Market Entropy
- 12.2.1 Company's Key Areas Served
- 12.2.2 Recent Developments
- 12.3. Company Market Share Analysis 2025
- 12.3.1 Top 5 Companies Market Share Analysis
- 12.3.2 Top 3 Companies Market Share Analysis
- 12.4. List of Potential Customers
- 13. Research Methodology
List of Figures
- Figure 1: Global Pneumococcal Vaccine Industry Revenue Breakdown (Million, %) by Region 2025 & 2033
- Figure 2: North America Pneumococcal Vaccine Industry Revenue (Million), by Vaccine Type 2025 & 2033
- Figure 3: North America Pneumococcal Vaccine Industry Revenue Share (%), by Vaccine Type 2025 & 2033
- Figure 4: North America Pneumococcal Vaccine Industry Revenue (Million), by Product Type 2025 & 2033
- Figure 5: North America Pneumococcal Vaccine Industry Revenue Share (%), by Product Type 2025 & 2033
- Figure 6: North America Pneumococcal Vaccine Industry Revenue (Million), by Distribution Channel 2025 & 2033
- Figure 7: North America Pneumococcal Vaccine Industry Revenue Share (%), by Distribution Channel 2025 & 2033
- Figure 8: North America Pneumococcal Vaccine Industry Revenue (Million), by Country 2025 & 2033
- Figure 9: North America Pneumococcal Vaccine Industry Revenue Share (%), by Country 2025 & 2033
- Figure 10: Europe Pneumococcal Vaccine Industry Revenue (Million), by Vaccine Type 2025 & 2033
- Figure 11: Europe Pneumococcal Vaccine Industry Revenue Share (%), by Vaccine Type 2025 & 2033
- Figure 12: Europe Pneumococcal Vaccine Industry Revenue (Million), by Product Type 2025 & 2033
- Figure 13: Europe Pneumococcal Vaccine Industry Revenue Share (%), by Product Type 2025 & 2033
- Figure 14: Europe Pneumococcal Vaccine Industry Revenue (Million), by Distribution Channel 2025 & 2033
- Figure 15: Europe Pneumococcal Vaccine Industry Revenue Share (%), by Distribution Channel 2025 & 2033
- Figure 16: Europe Pneumococcal Vaccine Industry Revenue (Million), by Country 2025 & 2033
- Figure 17: Europe Pneumococcal Vaccine Industry Revenue Share (%), by Country 2025 & 2033
- Figure 18: Asia Pacific Pneumococcal Vaccine Industry Revenue (Million), by Vaccine Type 2025 & 2033
- Figure 19: Asia Pacific Pneumococcal Vaccine Industry Revenue Share (%), by Vaccine Type 2025 & 2033
- Figure 20: Asia Pacific Pneumococcal Vaccine Industry Revenue (Million), by Product Type 2025 & 2033
- Figure 21: Asia Pacific Pneumococcal Vaccine Industry Revenue Share (%), by Product Type 2025 & 2033
- Figure 22: Asia Pacific Pneumococcal Vaccine Industry Revenue (Million), by Distribution Channel 2025 & 2033
- Figure 23: Asia Pacific Pneumococcal Vaccine Industry Revenue Share (%), by Distribution Channel 2025 & 2033
- Figure 24: Asia Pacific Pneumococcal Vaccine Industry Revenue (Million), by Country 2025 & 2033
- Figure 25: Asia Pacific Pneumococcal Vaccine Industry Revenue Share (%), by Country 2025 & 2033
- Figure 26: Middle East and Africa Pneumococcal Vaccine Industry Revenue (Million), by Vaccine Type 2025 & 2033
- Figure 27: Middle East and Africa Pneumococcal Vaccine Industry Revenue Share (%), by Vaccine Type 2025 & 2033
- Figure 28: Middle East and Africa Pneumococcal Vaccine Industry Revenue (Million), by Product Type 2025 & 2033
- Figure 29: Middle East and Africa Pneumococcal Vaccine Industry Revenue Share (%), by Product Type 2025 & 2033
- Figure 30: Middle East and Africa Pneumococcal Vaccine Industry Revenue (Million), by Distribution Channel 2025 & 2033
- Figure 31: Middle East and Africa Pneumococcal Vaccine Industry Revenue Share (%), by Distribution Channel 2025 & 2033
- Figure 32: Middle East and Africa Pneumococcal Vaccine Industry Revenue (Million), by Country 2025 & 2033
- Figure 33: Middle East and Africa Pneumococcal Vaccine Industry Revenue Share (%), by Country 2025 & 2033
- Figure 34: South America Pneumococcal Vaccine Industry Revenue (Million), by Vaccine Type 2025 & 2033
- Figure 35: South America Pneumococcal Vaccine Industry Revenue Share (%), by Vaccine Type 2025 & 2033
- Figure 36: South America Pneumococcal Vaccine Industry Revenue (Million), by Product Type 2025 & 2033
- Figure 37: South America Pneumococcal Vaccine Industry Revenue Share (%), by Product Type 2025 & 2033
- Figure 38: South America Pneumococcal Vaccine Industry Revenue (Million), by Distribution Channel 2025 & 2033
- Figure 39: South America Pneumococcal Vaccine Industry Revenue Share (%), by Distribution Channel 2025 & 2033
- Figure 40: South America Pneumococcal Vaccine Industry Revenue (Million), by Country 2025 & 2033
- Figure 41: South America Pneumococcal Vaccine Industry Revenue Share (%), by Country 2025 & 2033
List of Tables
- Table 1: Global Pneumococcal Vaccine Industry Revenue Million Forecast, by Vaccine Type 2020 & 2033
- Table 2: Global Pneumococcal Vaccine Industry Revenue Million Forecast, by Product Type 2020 & 2033
- Table 3: Global Pneumococcal Vaccine Industry Revenue Million Forecast, by Distribution Channel 2020 & 2033
- Table 4: Global Pneumococcal Vaccine Industry Revenue Million Forecast, by Region 2020 & 2033
- Table 5: Global Pneumococcal Vaccine Industry Revenue Million Forecast, by Vaccine Type 2020 & 2033
- Table 6: Global Pneumococcal Vaccine Industry Revenue Million Forecast, by Product Type 2020 & 2033
- Table 7: Global Pneumococcal Vaccine Industry Revenue Million Forecast, by Distribution Channel 2020 & 2033
- Table 8: Global Pneumococcal Vaccine Industry Revenue Million Forecast, by Country 2020 & 2033
- Table 9: United States Pneumococcal Vaccine Industry Revenue (Million) Forecast, by Application 2020 & 2033
- Table 10: Canada Pneumococcal Vaccine Industry Revenue (Million) Forecast, by Application 2020 & 2033
- Table 11: Mexico Pneumococcal Vaccine Industry Revenue (Million) Forecast, by Application 2020 & 2033
- Table 12: Global Pneumococcal Vaccine Industry Revenue Million Forecast, by Vaccine Type 2020 & 2033
- Table 13: Global Pneumococcal Vaccine Industry Revenue Million Forecast, by Product Type 2020 & 2033
- Table 14: Global Pneumococcal Vaccine Industry Revenue Million Forecast, by Distribution Channel 2020 & 2033
- Table 15: Global Pneumococcal Vaccine Industry Revenue Million Forecast, by Country 2020 & 2033
- Table 16: Germany Pneumococcal Vaccine Industry Revenue (Million) Forecast, by Application 2020 & 2033
- Table 17: United Kingdom Pneumococcal Vaccine Industry Revenue (Million) Forecast, by Application 2020 & 2033
- Table 18: France Pneumococcal Vaccine Industry Revenue (Million) Forecast, by Application 2020 & 2033
- Table 19: Italy Pneumococcal Vaccine Industry Revenue (Million) Forecast, by Application 2020 & 2033
- Table 20: Spain Pneumococcal Vaccine Industry Revenue (Million) Forecast, by Application 2020 & 2033
- Table 21: Rest of Europe Pneumococcal Vaccine Industry Revenue (Million) Forecast, by Application 2020 & 2033
- Table 22: Global Pneumococcal Vaccine Industry Revenue Million Forecast, by Vaccine Type 2020 & 2033
- Table 23: Global Pneumococcal Vaccine Industry Revenue Million Forecast, by Product Type 2020 & 2033
- Table 24: Global Pneumococcal Vaccine Industry Revenue Million Forecast, by Distribution Channel 2020 & 2033
- Table 25: Global Pneumococcal Vaccine Industry Revenue Million Forecast, by Country 2020 & 2033
- Table 26: China Pneumococcal Vaccine Industry Revenue (Million) Forecast, by Application 2020 & 2033
- Table 27: Japan Pneumococcal Vaccine Industry Revenue (Million) Forecast, by Application 2020 & 2033
- Table 28: India Pneumococcal Vaccine Industry Revenue (Million) Forecast, by Application 2020 & 2033
- Table 29: Australia Pneumococcal Vaccine Industry Revenue (Million) Forecast, by Application 2020 & 2033
- Table 30: South Korea Pneumococcal Vaccine Industry Revenue (Million) Forecast, by Application 2020 & 2033
- Table 31: Rest of Asia Pacific Pneumococcal Vaccine Industry Revenue (Million) Forecast, by Application 2020 & 2033
- Table 32: Global Pneumococcal Vaccine Industry Revenue Million Forecast, by Vaccine Type 2020 & 2033
- Table 33: Global Pneumococcal Vaccine Industry Revenue Million Forecast, by Product Type 2020 & 2033
- Table 34: Global Pneumococcal Vaccine Industry Revenue Million Forecast, by Distribution Channel 2020 & 2033
- Table 35: Global Pneumococcal Vaccine Industry Revenue Million Forecast, by Country 2020 & 2033
- Table 36: GCC Pneumococcal Vaccine Industry Revenue (Million) Forecast, by Application 2020 & 2033
- Table 37: South Africa Pneumococcal Vaccine Industry Revenue (Million) Forecast, by Application 2020 & 2033
- Table 38: Rest of Middle East and Africa Pneumococcal Vaccine Industry Revenue (Million) Forecast, by Application 2020 & 2033
- Table 39: Global Pneumococcal Vaccine Industry Revenue Million Forecast, by Vaccine Type 2020 & 2033
- Table 40: Global Pneumococcal Vaccine Industry Revenue Million Forecast, by Product Type 2020 & 2033
- Table 41: Global Pneumococcal Vaccine Industry Revenue Million Forecast, by Distribution Channel 2020 & 2033
- Table 42: Global Pneumococcal Vaccine Industry Revenue Million Forecast, by Country 2020 & 2033
- Table 43: Brazil Pneumococcal Vaccine Industry Revenue (Million) Forecast, by Application 2020 & 2033
- Table 44: Argentina Pneumococcal Vaccine Industry Revenue (Million) Forecast, by Application 2020 & 2033
- Table 45: Rest of South America Pneumococcal Vaccine Industry Revenue (Million) Forecast, by Application 2020 & 2033
Frequently Asked Questions
1. What is the projected Compound Annual Growth Rate (CAGR) of the Pneumococcal Vaccine Industry?
The projected CAGR is approximately 4.83%.
2. Which companies are prominent players in the Pneumococcal Vaccine Industry?
Key companies in the market include Walvax Biotechnology Co Ltd, CSL Ltd, Merck KGaA, Beijing Minhai Biotechnology Co Ltd*List Not Exhaustive, GSK PLC, Serum Institute of India Pvt Ltd, Sanofi S A, Pfizer Inc.
3. What are the main segments of the Pneumococcal Vaccine Industry?
The market segments include Vaccine Type, Product Type, Distribution Channel.
4. Can you provide details about the market size?
The market size is estimated to be USD 8.80 Million as of 2022.
5. What are some drivers contributing to market growth?
Increasing Government Awareness Programs Regarding Pneumonia Immunization; Rising Prevalence Of Pneumonia; Introduction of Novel Pneumococcal Vaccines.
6. What are the notable trends driving market growth?
The Pneumococcal Conjugate Vaccine Segment is Anticipated to Witness Growth Over the Forecast Period.
7. Are there any restraints impacting market growth?
Long Duration for the Production; High Cost of Production.
8. Can you provide examples of recent developments in the market?
April 2024: Merck presented the results of the phase 3 clinical trial STRIDE-10 for an investigational 21-valent pneumococcal conjugate vaccine developed for adults.
9. What pricing options are available for accessing the report?
Pricing options include single-user, multi-user, and enterprise licenses priced at USD 4750, USD 5250, and USD 8750 respectively.
10. Is the market size provided in terms of value or volume?
The market size is provided in terms of value, measured in Million.
11. Are there any specific market keywords associated with the report?
Yes, the market keyword associated with the report is "Pneumococcal Vaccine Industry," which aids in identifying and referencing the specific market segment covered.
12. How do I determine which pricing option suits my needs best?
The pricing options vary based on user requirements and access needs. Individual users may opt for single-user licenses, while businesses requiring broader access may choose multi-user or enterprise licenses for cost-effective access to the report.
13. Are there any additional resources or data provided in the Pneumococcal Vaccine Industry report?
While the report offers comprehensive insights, it's advisable to review the specific contents or supplementary materials provided to ascertain if additional resources or data are available.
14. How can I stay updated on further developments or reports in the Pneumococcal Vaccine Industry?
To stay informed about further developments, trends, and reports in the Pneumococcal Vaccine Industry, consider subscribing to industry newsletters, following relevant companies and organizations, or regularly checking reputable industry news sources and publications.
Methodology
Step 1 - Identification of Relevant Samples Size from Population Database



Step 2 - Approaches for Defining Global Market Size (Value, Volume* & Price*)

Note*: In applicable scenarios
Step 3 - Data Sources
Primary Research
- Web Analytics
- Survey Reports
- Research Institute
- Latest Research Reports
- Opinion Leaders
Secondary Research
- Annual Reports
- White Paper
- Latest Press Release
- Industry Association
- Paid Database
- Investor Presentations

Step 4 - Data Triangulation
Involves using different sources of information in order to increase the validity of a study
These sources are likely to be stakeholders in a program - participants, other researchers, program staff, other community members, and so on.
Then we put all data in single framework & apply various statistical tools to find out the dynamic on the market.
During the analysis stage, feedback from the stakeholder groups would be compared to determine areas of agreement as well as areas of divergence


