Key Insights
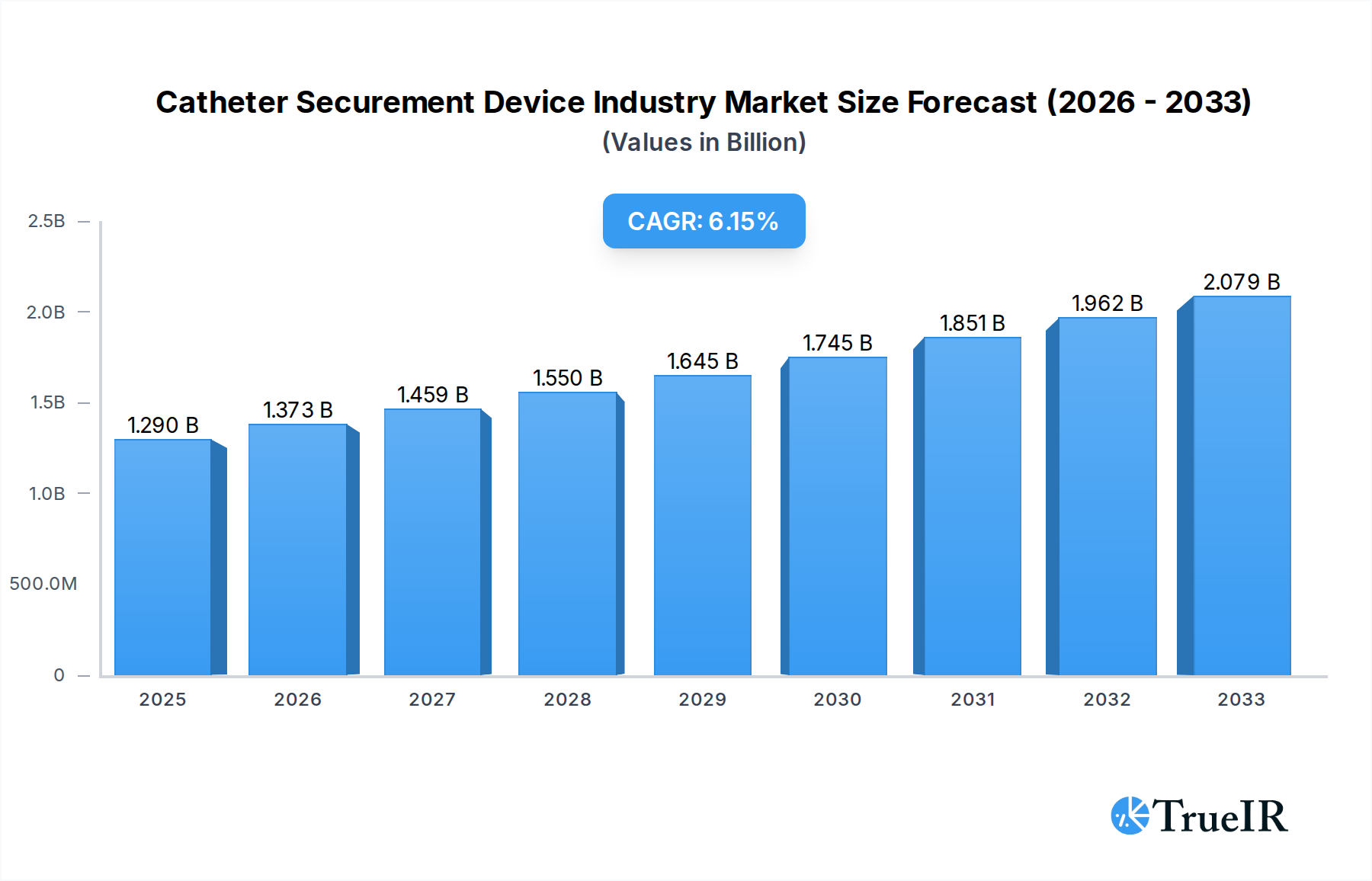
The global Catheter Securement Device market is poised for robust growth, projected to reach USD 1.29 billion in 2025, with a compelling CAGR of 6.4% anticipated over the forecast period extending to 2033. This expansion is driven by an increasing prevalence of chronic diseases, a rising aging population susceptible to infections and requiring long-term catheterization, and the growing demand for minimally invasive procedures. Advancements in securement technologies, offering enhanced patient comfort, reduced risk of dislodgement, and improved infection control, are also significant contributors. Hospitals remain the dominant end-user segment due to the high volume of catheterization procedures performed, while the expanding homecare sector presents a notable growth opportunity. The market is characterized by a competitive landscape with major players focusing on product innovation, strategic partnerships, and geographical expansion to capture market share.
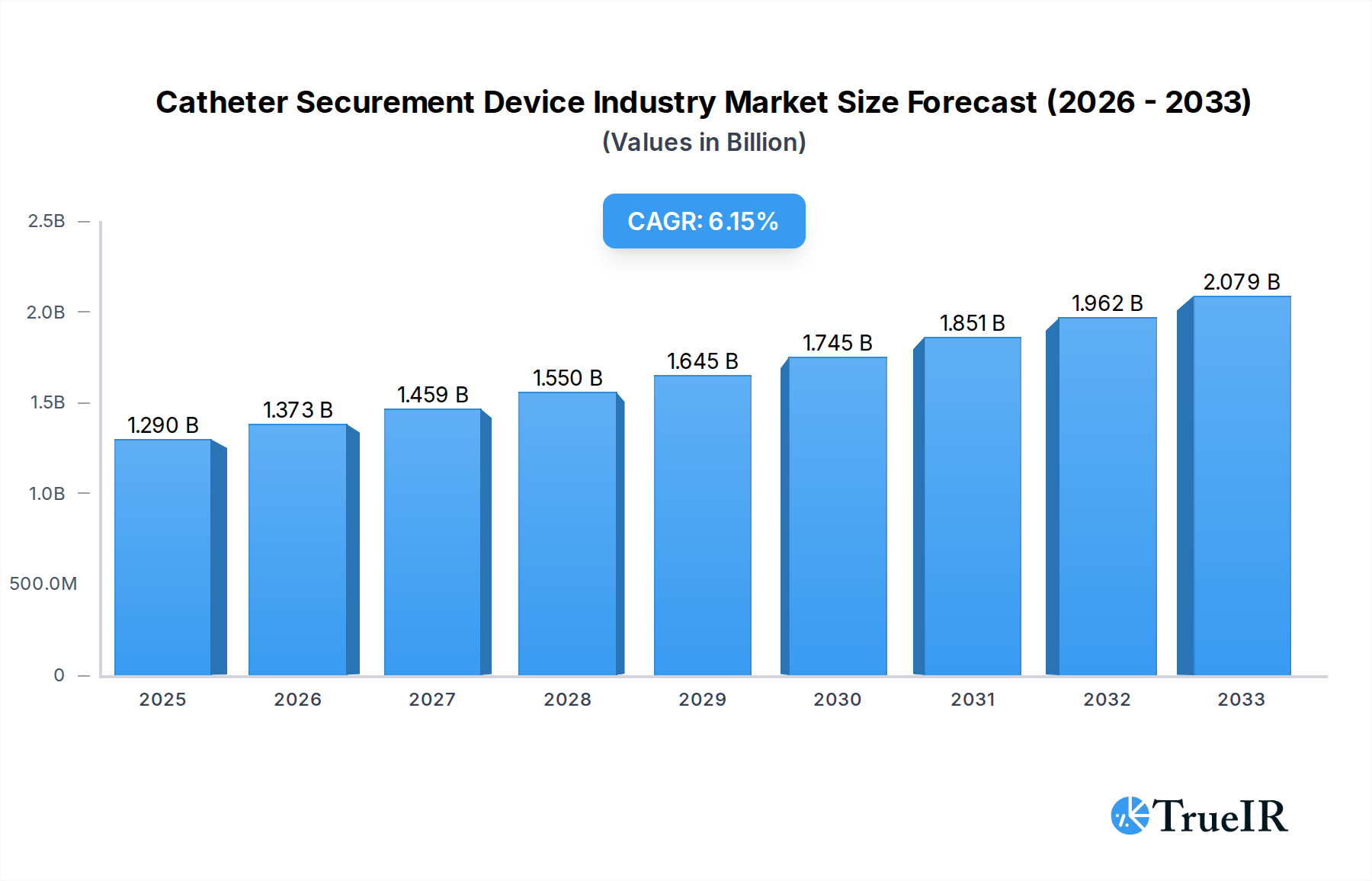
Catheter Securement Device Industry Market Size (In Billion)

The market's trajectory is further shaped by key trends such as the development of antimicrobial-coated securement devices to combat catheter-associated infections, and the increasing adoption of adhesive-based and integrated securement solutions that offer ease of use and improved patient adherence. While the market enjoys substantial growth drivers, certain restraints, such as the cost-effectiveness concerns in certain healthcare settings and the need for greater awareness and adoption of advanced securement devices in developing regions, warrant attention. The product segment is led by Arterial Securement Devices and Central Venous Catheter Securement Devices, reflecting their widespread application in critical care and long-term venous access. North America and Europe currently hold the largest market shares, driven by advanced healthcare infrastructure and a higher incidence of conditions requiring catheterization. However, the Asia Pacific region is expected to witness the fastest growth due to rapid improvements in healthcare facilities and increasing disposable incomes.
Catheter Securement Device Industry Company Market Share

Here is a dynamic, SEO-optimized report description for the Catheter Securement Device Industry, designed for immediate use without modification.
Catheter Securement Device Industry Market Structure & Competitive Landscape
The global Catheter Securement Device market is characterized by a moderately concentrated structure, with a significant share held by a few key players, alongside a growing number of specialized manufacturers. Innovation serves as a primary driver, fueled by the continuous need for enhanced patient safety, reduced healthcare-associated infections, and improved clinical outcomes. Regulatory frameworks, including FDA approvals and CE marking, play a crucial role in market entry and product development, ensuring adherence to stringent safety and efficacy standards. The threat of product substitutes, such as advanced dressing techniques or alternative fixation methods, exists but is often mitigated by the specialized design and proven reliability of dedicated securement devices. End-user segmentation, primarily driven by hospital procurement and the burgeoning home care sector, significantly influences market dynamics. Mergers and acquisitions (M&A) are strategic maneuvers employed by larger entities to expand their product portfolios, geographic reach, and market share. We anticipate the volume of M&A activities to reach approximately 500 million USD annually during the forecast period. Key players like BD (C R Bard), 3M, and Medtronic PLC demonstrate a strong presence due to their extensive product lines and global distribution networks.
Catheter Securement Device Industry Market Trends & Opportunities
The Catheter Securement Device industry is poised for substantial growth, driven by an increasing prevalence of chronic diseases, an aging global population, and a growing emphasis on preventing catheter-related complications. The global market is projected to reach an estimated 15.8 billion USD by 2025, exhibiting a robust Compound Annual Growth Rate (CAGR) of 7.2% from 2019 to 2033. This expansion is underpinned by several key trends. Firstly, technological advancements are leading to the development of innovative securement devices that offer superior adhesion, reduced skin irritation, and enhanced patient comfort. Features such as antimicrobial coatings, integrated monitoring capabilities, and user-friendly designs are becoming increasingly prevalent. Secondly, a rising awareness among healthcare professionals and institutions regarding the economic and clinical burden of catheter-related bloodstream infections (CRBSIs) and dislodgement events is propelling the adoption of advanced securement solutions. The direct and indirect costs associated with these complications, including extended hospital stays, increased treatment expenses, and potential litigation, underscore the value proposition of effective catheter securement.
Furthermore, the expanding healthcare infrastructure in emerging economies presents significant opportunities for market penetration. As access to advanced medical care improves in regions like Asia-Pacific and Latin America, the demand for sophisticated medical devices, including catheter securement solutions, is expected to surge. The shift towards home healthcare settings also contributes to market expansion. With an increasing number of patients receiving medical treatment outside traditional hospital environments, there is a growing need for secure and easy-to-use securement devices that can be managed by patients or their caregivers. This trend necessitates the development of user-friendly, disposable, and cost-effective solutions. Competitive dynamics are evolving, with both established players and new entrants vying for market share through product differentiation, strategic partnerships, and aggressive marketing strategies. The market penetration rate for advanced catheter securement devices is currently estimated at 65% in developed nations, with significant room for growth in developing regions.
Dominant Markets & Segments in Catheter Securement Device Industry
The Catheter Securement Device market exhibits clear dominance across specific regions and product segments, driven by a confluence of healthcare infrastructure, patient demographics, and regulatory environments. North America currently leads the market, accounting for an estimated 35% of the global revenue in 2025. This leadership is attributed to its advanced healthcare system, high expenditure on medical devices, widespread adoption of sophisticated medical technologies, and a high prevalence of conditions requiring catheterization. Within North America, the United States represents the single largest country market, driven by its extensive hospital network and a strong focus on patient safety initiatives.
The Hospitals end-user segment is the largest contributor to market revenue, representing approximately 60% of the total market value in 2025. This dominance stems from the high volume of catheterizations performed in inpatient settings for various medical procedures and treatments. Hospitals are also at the forefront of adopting advanced securement devices due to stringent infection control protocols and the financial implications of catheter-related complications.
In terms of product segments, Central Venous Catheter Securement Devices are a major revenue generator, estimated to capture 25% of the market by 2025. This is due to the critical nature of central venous catheters in managing critically ill patients, requiring robust and reliable securement to prevent dislodgement and infection. Arterial Securement Devices also represent a significant segment, driven by the widespread use of arterial lines for hemodynamic monitoring and blood sampling. The Urinary Catheters Securement Devices segment is also substantial, catering to a vast patient population requiring long-term urinary catheterization, where preventing dislodgement and associated discomfort is paramount.
Key growth drivers for these dominant segments include:
- Advanced Healthcare Infrastructure: Well-equipped hospitals with a high patient turnover and access to cutting-edge medical technology.
- Favorable Reimbursement Policies: Policies that encourage the use of advanced securement devices due to their proven efficacy in reducing complications and associated costs.
- High Prevalence of Catheter-Related Complications: A growing understanding and tracking of CRBSIs and catheter dislodgements, leading to increased demand for preventive solutions.
- Aging Population: An increasing elderly population susceptible to chronic conditions requiring prolonged catheterization.
- Technological Innovations: Continuous development of securement devices with enhanced features and improved patient outcomes.
Catheter Securement Device Industry Product Analysis
Product innovation in the Catheter Securement Device industry is primarily focused on enhancing patient safety, comfort, and ease of use. Manufacturers are developing adhesive-based securement devices with hypoallergenic materials to minimize skin irritation, particularly for long-term applications. Antimicrobial coatings are being integrated into securement devices to actively combat bacterial growth and reduce the risk of catheter-related infections. Smart securement devices with integrated sensors for monitoring catheter position or fluid flow are emerging, offering advanced clinical insights. The competitive advantage lies in the ability to offer a comprehensive range of securement solutions tailored to specific catheter types and clinical applications, alongside proven clinical efficacy and cost-effectiveness.
Key Drivers, Barriers & Challenges in Catheter Securement Device Industry
Key Drivers:
- Technological Advancements: Development of innovative materials, improved adhesion, antimicrobial properties, and user-friendly designs.
- Increasing Healthcare Expenditure: Growing investment in medical devices and patient safety initiatives globally.
- Rising Prevalence of Chronic Diseases: An aging population and increased incidence of conditions requiring long-term catheterization.
- Growing Awareness of Catheter-Related Complications: Emphasis on preventing infections and dislodgements to improve patient outcomes and reduce healthcare costs.
- Expansion of Home Healthcare: Increasing demand for securement solutions suitable for non-clinical settings.
Barriers and Challenges:
- Regulatory Hurdles: Stringent approval processes and evolving standards can delay product launches and increase development costs.
- Price Sensitivity: In some healthcare systems, cost remains a significant factor influencing purchasing decisions, potentially limiting adoption of premium securement devices.
- Supply Chain Disruptions: Global geopolitical events and logistics challenges can impact raw material availability and product distribution, potentially leading to stockouts.
- Competition from Traditional Methods: Persistence of less advanced but cheaper methods in certain markets.
- Limited Physician Awareness: In some regions, a lack of comprehensive training and awareness regarding the benefits of advanced securement devices can hinder adoption. The estimated impact of supply chain disruptions on market growth is approximately 5% annually.
Growth Drivers in the Catheter Securement Device Industry Market
The Catheter Securement Device market is propelled by several key growth drivers. Technological innovation stands out, with advancements in materials science leading to more skin-friendly adhesives and integrated antimicrobial properties that significantly reduce infection risks. The increasing global prevalence of chronic diseases and an aging population necessitate longer-term catheterization, directly boosting demand for reliable securement solutions. Furthermore, heightened awareness among healthcare providers and payers about the substantial costs associated with catheter-related complications, such as bloodstream infections and accidental dislodgements, is driving the adoption of preventive measures. Favorable reimbursement policies in many developed nations also incentivize the use of advanced securement devices. The expansion of home healthcare services further broadens the market, creating a need for user-friendly and effective devices for non-clinical environments.
Challenges Impacting Catheter Securement Device Industry Growth
Despite robust growth prospects, the Catheter Securement Device industry faces several challenges. Navigating complex and evolving regulatory landscapes across different geographies can be a significant hurdle, leading to extended product approval timelines and increased compliance costs. Supply chain vulnerabilities, exacerbated by geopolitical instability and global trade dynamics, pose a risk to the consistent availability of raw materials and finished products. Competitive pressures are intense, with a mix of established players and emerging companies vying for market share, often leading to price erosion in certain segments. Furthermore, overcoming inertia and encouraging the widespread adoption of advanced securement devices over traditional, less effective methods requires sustained education and demonstration of economic benefits. The estimated impact of regulatory hurdles on new product launches is a delay of 6-18 months.
Key Players Shaping the Catheter Securement Device Industry Market
- Argon Medical Devices
- Merit Medical Systems
- Tractus Vascular
- Levity Products Incorporated
- BD (C R Bard)
- 3M
- Baxter
- ConvaTec Group PLC
- Medtronic PLC
- TIDI Products LLC
- B Braun SE
- Medline Industries Inc
- CATHETRIX
Significant Catheter Securement Device Industry Industry Milestones
- April 2022: Bedal and Gulf Medical Co. Ltd. (GMC) signed a distribution agreement for Saudi Arabia and Bahrain. The Bedal products will be distributed by GMC in the Saudi Arabian and Bahraini markets, representing a major part of the market for the usage of catheter stabilization devices in the Gulf region.
- March 2022: Tractus Vascular reported the first human use of its Tractus Crossing Support Catheter.
Future Outlook for Catheter Securement Device Industry Market
The future outlook for the Catheter Securement Device industry is exceptionally bright, driven by persistent demand and ongoing innovation. The projected market size of over 30 billion USD by 2033 signals significant growth potential. Key growth catalysts include the continued development of smart securement devices with integrated monitoring capabilities, further advancements in antimicrobial technologies to combat escalating infection rates, and the expanding use of these devices in novel applications beyond traditional catheters. Strategic partnerships and acquisitions will likely continue to shape the competitive landscape, with companies seeking to broaden their product portfolios and geographic reach. The increasing focus on value-based healthcare and patient safety will further bolster the adoption of advanced securement solutions, making this a dynamic and essential segment of the medical device market.
Catheter Securement Device Industry Segmentation
-
1. Product
- 1.1. Arterial Securement Devices
- 1.2. Central Venous Catheter Securement Devices
- 1.3. Peripheral Securement Devices
- 1.4. Urinary Catheters Securement Devices
- 1.5. Chest Drainage Tube Securement Devices
- 1.6. Other Securement Devices
-
2. End-User
- 2.1. Hospitals
- 2.2. Home care Settings
- 2.3. Other End-Users
Catheter Securement Device Industry Segmentation By Geography
-
1. North America
- 1.1. United States
- 1.2. Canada
- 1.3. Mexico
-
2. Europe
- 2.1. France
- 2.2. Germany
- 2.3. United Kingdom
- 2.4. Italy
- 2.5. Spain
- 2.6. Rest of Europe
-
3. Asia Pacific
- 3.1. China
- 3.2. Japan
- 3.3. India
- 3.4. Australia
- 3.5. South Korea
- 3.6. Rest of Asia Pacific
-
4. Middle East and Africa
- 4.1. GCC
- 4.2. South Africa
- 4.3. Rest of Middle East and Africa
-
5. South America
- 5.1. Brazil
- 5.2. Argentina
- 5.3. Rest of South America
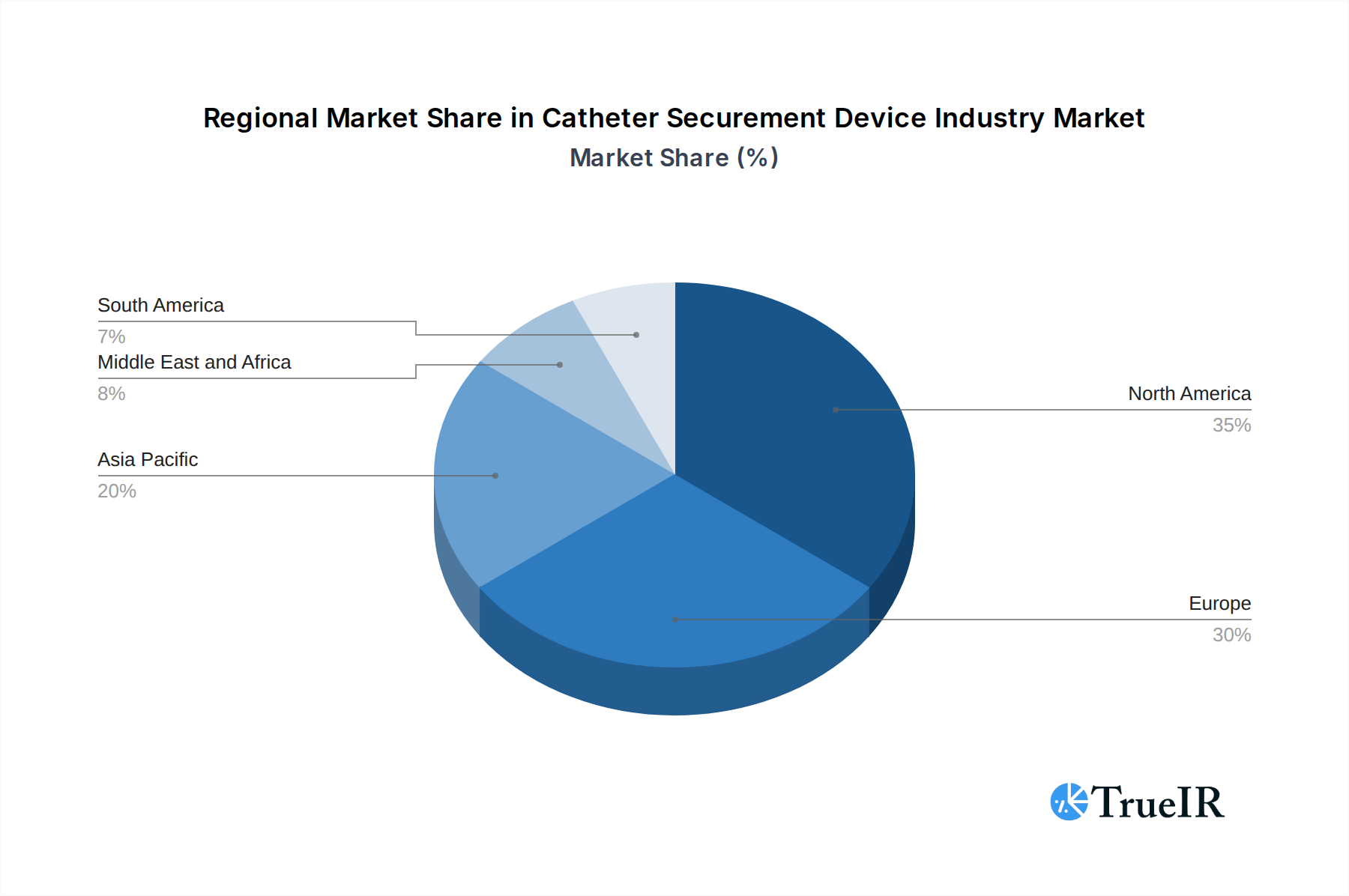
Catheter Securement Device Industry Regional Market Share

Geographic Coverage of Catheter Securement Device Industry
Catheter Securement Device Industry REPORT HIGHLIGHTS
| Aspects | Details |
|---|---|
| Study Period | 2020-2034 |
| Base Year | 2025 |
| Estimated Year | 2026 |
| Forecast Period | 2026-2034 |
| Historical Period | 2020-2025 |
| Growth Rate | CAGR of 6.4% from 2020-2034 |
| Segmentation |
|
Table of Contents
- 1. Introduction
- 1.1. Research Scope
- 1.2. Market Segmentation
- 1.3. Research Objective
- 1.4. Definitions and Assumptions
- 2. Executive Summary
- 2.1. Market Snapshot
- 3. Market Dynamics
- 3.1. Market Drivers
- 3.2. Market Restrains
- 3.3. Market Trends
- 3.4. Market Opportunities
- 4. Market Factor Analysis
- 4.1. Porters Five Forces
- 4.1.1. Bargaining Power of Suppliers
- 4.1.2. Bargaining Power of Buyers
- 4.1.3. Threat of New Entrants
- 4.1.4. Threat of Substitutes
- 4.1.5. Competitive Rivalry
- 4.2. PESTEL analysis
- 4.3. BCG Analysis
- 4.3.1. Stars (High Growth, High Market Share)
- 4.3.2. Cash Cows (Low Growth, High Market Share)
- 4.3.3. Question Mark (High Growth, Low Market Share)
- 4.3.4. Dogs (Low Growth, Low Market Share)
- 4.4. Ansoff Matrix Analysis
- 4.5. Supply Chain Analysis
- 4.6. Regulatory Landscape
- 4.7. Current Market Potential and Opportunity Assessment (TAM–SAM–SOM Framework)
- 4.8. TIR Analyst Note
- 4.1. Porters Five Forces
- 5. Market Analysis, Insights and Forecast 2021-2033
- 5.1. Market Analysis, Insights and Forecast - by Product
- 5.1.1. Arterial Securement Devices
- 5.1.2. Central Venous Catheter Securement Devices
- 5.1.3. Peripheral Securement Devices
- 5.1.4. Urinary Catheters Securement Devices
- 5.1.5. Chest Drainage Tube Securement Devices
- 5.1.6. Other Securement Devices
- 5.2. Market Analysis, Insights and Forecast - by End-User
- 5.2.1. Hospitals
- 5.2.2. Home care Settings
- 5.2.3. Other End-Users
- 5.3. Market Analysis, Insights and Forecast - by Region
- 5.3.1. North America
- 5.3.2. Europe
- 5.3.3. Asia Pacific
- 5.3.4. Middle East and Africa
- 5.3.5. South America
- 5.1. Market Analysis, Insights and Forecast - by Product
- 6. Global Catheter Securement Device Industry Analysis, Insights and Forecast, 2021-2033
- 6.1. Market Analysis, Insights and Forecast - by Product
- 6.1.1. Arterial Securement Devices
- 6.1.2. Central Venous Catheter Securement Devices
- 6.1.3. Peripheral Securement Devices
- 6.1.4. Urinary Catheters Securement Devices
- 6.1.5. Chest Drainage Tube Securement Devices
- 6.1.6. Other Securement Devices
- 6.2. Market Analysis, Insights and Forecast - by End-User
- 6.2.1. Hospitals
- 6.2.2. Home care Settings
- 6.2.3. Other End-Users
- 6.1. Market Analysis, Insights and Forecast - by Product
- 7. North America Catheter Securement Device Industry Analysis, Insights and Forecast, 2020-2032
- 7.1. Market Analysis, Insights and Forecast - by Product
- 7.1.1. Arterial Securement Devices
- 7.1.2. Central Venous Catheter Securement Devices
- 7.1.3. Peripheral Securement Devices
- 7.1.4. Urinary Catheters Securement Devices
- 7.1.5. Chest Drainage Tube Securement Devices
- 7.1.6. Other Securement Devices
- 7.2. Market Analysis, Insights and Forecast - by End-User
- 7.2.1. Hospitals
- 7.2.2. Home care Settings
- 7.2.3. Other End-Users
- 7.1. Market Analysis, Insights and Forecast - by Product
- 8. Europe Catheter Securement Device Industry Analysis, Insights and Forecast, 2020-2032
- 8.1. Market Analysis, Insights and Forecast - by Product
- 8.1.1. Arterial Securement Devices
- 8.1.2. Central Venous Catheter Securement Devices
- 8.1.3. Peripheral Securement Devices
- 8.1.4. Urinary Catheters Securement Devices
- 8.1.5. Chest Drainage Tube Securement Devices
- 8.1.6. Other Securement Devices
- 8.2. Market Analysis, Insights and Forecast - by End-User
- 8.2.1. Hospitals
- 8.2.2. Home care Settings
- 8.2.3. Other End-Users
- 8.1. Market Analysis, Insights and Forecast - by Product
- 9. Asia Pacific Catheter Securement Device Industry Analysis, Insights and Forecast, 2020-2032
- 9.1. Market Analysis, Insights and Forecast - by Product
- 9.1.1. Arterial Securement Devices
- 9.1.2. Central Venous Catheter Securement Devices
- 9.1.3. Peripheral Securement Devices
- 9.1.4. Urinary Catheters Securement Devices
- 9.1.5. Chest Drainage Tube Securement Devices
- 9.1.6. Other Securement Devices
- 9.2. Market Analysis, Insights and Forecast - by End-User
- 9.2.1. Hospitals
- 9.2.2. Home care Settings
- 9.2.3. Other End-Users
- 9.1. Market Analysis, Insights and Forecast - by Product
- 10. Middle East and Africa Catheter Securement Device Industry Analysis, Insights and Forecast, 2020-2032
- 10.1. Market Analysis, Insights and Forecast - by Product
- 10.1.1. Arterial Securement Devices
- 10.1.2. Central Venous Catheter Securement Devices
- 10.1.3. Peripheral Securement Devices
- 10.1.4. Urinary Catheters Securement Devices
- 10.1.5. Chest Drainage Tube Securement Devices
- 10.1.6. Other Securement Devices
- 10.2. Market Analysis, Insights and Forecast - by End-User
- 10.2.1. Hospitals
- 10.2.2. Home care Settings
- 10.2.3. Other End-Users
- 10.1. Market Analysis, Insights and Forecast - by Product
- 11. South America Catheter Securement Device Industry Analysis, Insights and Forecast, 2020-2032
- 11.1. Market Analysis, Insights and Forecast - by Product
- 11.1.1. Arterial Securement Devices
- 11.1.2. Central Venous Catheter Securement Devices
- 11.1.3. Peripheral Securement Devices
- 11.1.4. Urinary Catheters Securement Devices
- 11.1.5. Chest Drainage Tube Securement Devices
- 11.1.6. Other Securement Devices
- 11.2. Market Analysis, Insights and Forecast - by End-User
- 11.2.1. Hospitals
- 11.2.2. Home care Settings
- 11.2.3. Other End-Users
- 11.1. Market Analysis, Insights and Forecast - by Product
- 12. Competitive Analysis
- 12.1. Company Profiles
- 12.1.1 Argon Medical Devices
- 12.1.1.1. Company Overview
- 12.1.1.2. Products
- 12.1.1.3. Company Financials
- 12.1.1.4. SWOT Analysis
- 12.1.2 Merit Medical Systems
- 12.1.2.1. Company Overview
- 12.1.2.2. Products
- 12.1.2.3. Company Financials
- 12.1.2.4. SWOT Analysis
- 12.1.3 Tractus Vascular
- 12.1.3.1. Company Overview
- 12.1.3.2. Products
- 12.1.3.3. Company Financials
- 12.1.3.4. SWOT Analysis
- 12.1.4 Levity Products Incorporated
- 12.1.4.1. Company Overview
- 12.1.4.2. Products
- 12.1.4.3. Company Financials
- 12.1.4.4. SWOT Analysis
- 12.1.5 BD (C R Bard)
- 12.1.5.1. Company Overview
- 12.1.5.2. Products
- 12.1.5.3. Company Financials
- 12.1.5.4. SWOT Analysis
- 12.1.6 3M
- 12.1.6.1. Company Overview
- 12.1.6.2. Products
- 12.1.6.3. Company Financials
- 12.1.6.4. SWOT Analysis
- 12.1.7 Baxter
- 12.1.7.1. Company Overview
- 12.1.7.2. Products
- 12.1.7.3. Company Financials
- 12.1.7.4. SWOT Analysis
- 12.1.8 ConvaTec Group PLC
- 12.1.8.1. Company Overview
- 12.1.8.2. Products
- 12.1.8.3. Company Financials
- 12.1.8.4. SWOT Analysis
- 12.1.9 Medtronic PLC
- 12.1.9.1. Company Overview
- 12.1.9.2. Products
- 12.1.9.3. Company Financials
- 12.1.9.4. SWOT Analysis
- 12.1.10 TIDI Products LLC
- 12.1.10.1. Company Overview
- 12.1.10.2. Products
- 12.1.10.3. Company Financials
- 12.1.10.4. SWOT Analysis
- 12.1.11 B Braun SE
- 12.1.11.1. Company Overview
- 12.1.11.2. Products
- 12.1.11.3. Company Financials
- 12.1.11.4. SWOT Analysis
- 12.1.12 Medline Industries Inc
- 12.1.12.1. Company Overview
- 12.1.12.2. Products
- 12.1.12.3. Company Financials
- 12.1.12.4. SWOT Analysis
- 12.1.13 CATHETRIX
- 12.1.13.1. Company Overview
- 12.1.13.2. Products
- 12.1.13.3. Company Financials
- 12.1.13.4. SWOT Analysis
- 12.1.1 Argon Medical Devices
- 12.2. Market Entropy
- 12.2.1 Company's Key Areas Served
- 12.2.2 Recent Developments
- 12.3. Company Market Share Analysis 2025
- 12.3.1 Top 5 Companies Market Share Analysis
- 12.3.2 Top 3 Companies Market Share Analysis
- 12.4. List of Potential Customers
- 13. Research Methodology
List of Figures
- Figure 1: Global Catheter Securement Device Industry Revenue Breakdown (billion, %) by Region 2025 & 2033
- Figure 2: Global Catheter Securement Device Industry Volume Breakdown (K Units, %) by Region 2025 & 2033
- Figure 3: North America Catheter Securement Device Industry Revenue (billion), by Product 2025 & 2033
- Figure 4: North America Catheter Securement Device Industry Volume (K Units), by Product 2025 & 2033
- Figure 5: North America Catheter Securement Device Industry Revenue Share (%), by Product 2025 & 2033
- Figure 6: North America Catheter Securement Device Industry Volume Share (%), by Product 2025 & 2033
- Figure 7: North America Catheter Securement Device Industry Revenue (billion), by End-User 2025 & 2033
- Figure 8: North America Catheter Securement Device Industry Volume (K Units), by End-User 2025 & 2033
- Figure 9: North America Catheter Securement Device Industry Revenue Share (%), by End-User 2025 & 2033
- Figure 10: North America Catheter Securement Device Industry Volume Share (%), by End-User 2025 & 2033
- Figure 11: North America Catheter Securement Device Industry Revenue (billion), by Country 2025 & 2033
- Figure 12: North America Catheter Securement Device Industry Volume (K Units), by Country 2025 & 2033
- Figure 13: North America Catheter Securement Device Industry Revenue Share (%), by Country 2025 & 2033
- Figure 14: North America Catheter Securement Device Industry Volume Share (%), by Country 2025 & 2033
- Figure 15: Europe Catheter Securement Device Industry Revenue (billion), by Product 2025 & 2033
- Figure 16: Europe Catheter Securement Device Industry Volume (K Units), by Product 2025 & 2033
- Figure 17: Europe Catheter Securement Device Industry Revenue Share (%), by Product 2025 & 2033
- Figure 18: Europe Catheter Securement Device Industry Volume Share (%), by Product 2025 & 2033
- Figure 19: Europe Catheter Securement Device Industry Revenue (billion), by End-User 2025 & 2033
- Figure 20: Europe Catheter Securement Device Industry Volume (K Units), by End-User 2025 & 2033
- Figure 21: Europe Catheter Securement Device Industry Revenue Share (%), by End-User 2025 & 2033
- Figure 22: Europe Catheter Securement Device Industry Volume Share (%), by End-User 2025 & 2033
- Figure 23: Europe Catheter Securement Device Industry Revenue (billion), by Country 2025 & 2033
- Figure 24: Europe Catheter Securement Device Industry Volume (K Units), by Country 2025 & 2033
- Figure 25: Europe Catheter Securement Device Industry Revenue Share (%), by Country 2025 & 2033
- Figure 26: Europe Catheter Securement Device Industry Volume Share (%), by Country 2025 & 2033
- Figure 27: Asia Pacific Catheter Securement Device Industry Revenue (billion), by Product 2025 & 2033
- Figure 28: Asia Pacific Catheter Securement Device Industry Volume (K Units), by Product 2025 & 2033
- Figure 29: Asia Pacific Catheter Securement Device Industry Revenue Share (%), by Product 2025 & 2033
- Figure 30: Asia Pacific Catheter Securement Device Industry Volume Share (%), by Product 2025 & 2033
- Figure 31: Asia Pacific Catheter Securement Device Industry Revenue (billion), by End-User 2025 & 2033
- Figure 32: Asia Pacific Catheter Securement Device Industry Volume (K Units), by End-User 2025 & 2033
- Figure 33: Asia Pacific Catheter Securement Device Industry Revenue Share (%), by End-User 2025 & 2033
- Figure 34: Asia Pacific Catheter Securement Device Industry Volume Share (%), by End-User 2025 & 2033
- Figure 35: Asia Pacific Catheter Securement Device Industry Revenue (billion), by Country 2025 & 2033
- Figure 36: Asia Pacific Catheter Securement Device Industry Volume (K Units), by Country 2025 & 2033
- Figure 37: Asia Pacific Catheter Securement Device Industry Revenue Share (%), by Country 2025 & 2033
- Figure 38: Asia Pacific Catheter Securement Device Industry Volume Share (%), by Country 2025 & 2033
- Figure 39: Middle East and Africa Catheter Securement Device Industry Revenue (billion), by Product 2025 & 2033
- Figure 40: Middle East and Africa Catheter Securement Device Industry Volume (K Units), by Product 2025 & 2033
- Figure 41: Middle East and Africa Catheter Securement Device Industry Revenue Share (%), by Product 2025 & 2033
- Figure 42: Middle East and Africa Catheter Securement Device Industry Volume Share (%), by Product 2025 & 2033
- Figure 43: Middle East and Africa Catheter Securement Device Industry Revenue (billion), by End-User 2025 & 2033
- Figure 44: Middle East and Africa Catheter Securement Device Industry Volume (K Units), by End-User 2025 & 2033
- Figure 45: Middle East and Africa Catheter Securement Device Industry Revenue Share (%), by End-User 2025 & 2033
- Figure 46: Middle East and Africa Catheter Securement Device Industry Volume Share (%), by End-User 2025 & 2033
- Figure 47: Middle East and Africa Catheter Securement Device Industry Revenue (billion), by Country 2025 & 2033
- Figure 48: Middle East and Africa Catheter Securement Device Industry Volume (K Units), by Country 2025 & 2033
- Figure 49: Middle East and Africa Catheter Securement Device Industry Revenue Share (%), by Country 2025 & 2033
- Figure 50: Middle East and Africa Catheter Securement Device Industry Volume Share (%), by Country 2025 & 2033
- Figure 51: South America Catheter Securement Device Industry Revenue (billion), by Product 2025 & 2033
- Figure 52: South America Catheter Securement Device Industry Volume (K Units), by Product 2025 & 2033
- Figure 53: South America Catheter Securement Device Industry Revenue Share (%), by Product 2025 & 2033
- Figure 54: South America Catheter Securement Device Industry Volume Share (%), by Product 2025 & 2033
- Figure 55: South America Catheter Securement Device Industry Revenue (billion), by End-User 2025 & 2033
- Figure 56: South America Catheter Securement Device Industry Volume (K Units), by End-User 2025 & 2033
- Figure 57: South America Catheter Securement Device Industry Revenue Share (%), by End-User 2025 & 2033
- Figure 58: South America Catheter Securement Device Industry Volume Share (%), by End-User 2025 & 2033
- Figure 59: South America Catheter Securement Device Industry Revenue (billion), by Country 2025 & 2033
- Figure 60: South America Catheter Securement Device Industry Volume (K Units), by Country 2025 & 2033
- Figure 61: South America Catheter Securement Device Industry Revenue Share (%), by Country 2025 & 2033
- Figure 62: South America Catheter Securement Device Industry Volume Share (%), by Country 2025 & 2033
List of Tables
- Table 1: Global Catheter Securement Device Industry Revenue billion Forecast, by Product 2020 & 2033
- Table 2: Global Catheter Securement Device Industry Volume K Units Forecast, by Product 2020 & 2033
- Table 3: Global Catheter Securement Device Industry Revenue billion Forecast, by End-User 2020 & 2033
- Table 4: Global Catheter Securement Device Industry Volume K Units Forecast, by End-User 2020 & 2033
- Table 5: Global Catheter Securement Device Industry Revenue billion Forecast, by Region 2020 & 2033
- Table 6: Global Catheter Securement Device Industry Volume K Units Forecast, by Region 2020 & 2033
- Table 7: Global Catheter Securement Device Industry Revenue billion Forecast, by Product 2020 & 2033
- Table 8: Global Catheter Securement Device Industry Volume K Units Forecast, by Product 2020 & 2033
- Table 9: Global Catheter Securement Device Industry Revenue billion Forecast, by End-User 2020 & 2033
- Table 10: Global Catheter Securement Device Industry Volume K Units Forecast, by End-User 2020 & 2033
- Table 11: Global Catheter Securement Device Industry Revenue billion Forecast, by Country 2020 & 2033
- Table 12: Global Catheter Securement Device Industry Volume K Units Forecast, by Country 2020 & 2033
- Table 13: United States Catheter Securement Device Industry Revenue (billion) Forecast, by Application 2020 & 2033
- Table 14: United States Catheter Securement Device Industry Volume (K Units) Forecast, by Application 2020 & 2033
- Table 15: Canada Catheter Securement Device Industry Revenue (billion) Forecast, by Application 2020 & 2033
- Table 16: Canada Catheter Securement Device Industry Volume (K Units) Forecast, by Application 2020 & 2033
- Table 17: Mexico Catheter Securement Device Industry Revenue (billion) Forecast, by Application 2020 & 2033
- Table 18: Mexico Catheter Securement Device Industry Volume (K Units) Forecast, by Application 2020 & 2033
- Table 19: Global Catheter Securement Device Industry Revenue billion Forecast, by Product 2020 & 2033
- Table 20: Global Catheter Securement Device Industry Volume K Units Forecast, by Product 2020 & 2033
- Table 21: Global Catheter Securement Device Industry Revenue billion Forecast, by End-User 2020 & 2033
- Table 22: Global Catheter Securement Device Industry Volume K Units Forecast, by End-User 2020 & 2033
- Table 23: Global Catheter Securement Device Industry Revenue billion Forecast, by Country 2020 & 2033
- Table 24: Global Catheter Securement Device Industry Volume K Units Forecast, by Country 2020 & 2033
- Table 25: France Catheter Securement Device Industry Revenue (billion) Forecast, by Application 2020 & 2033
- Table 26: France Catheter Securement Device Industry Volume (K Units) Forecast, by Application 2020 & 2033
- Table 27: Germany Catheter Securement Device Industry Revenue (billion) Forecast, by Application 2020 & 2033
- Table 28: Germany Catheter Securement Device Industry Volume (K Units) Forecast, by Application 2020 & 2033
- Table 29: United Kingdom Catheter Securement Device Industry Revenue (billion) Forecast, by Application 2020 & 2033
- Table 30: United Kingdom Catheter Securement Device Industry Volume (K Units) Forecast, by Application 2020 & 2033
- Table 31: Italy Catheter Securement Device Industry Revenue (billion) Forecast, by Application 2020 & 2033
- Table 32: Italy Catheter Securement Device Industry Volume (K Units) Forecast, by Application 2020 & 2033
- Table 33: Spain Catheter Securement Device Industry Revenue (billion) Forecast, by Application 2020 & 2033
- Table 34: Spain Catheter Securement Device Industry Volume (K Units) Forecast, by Application 2020 & 2033
- Table 35: Rest of Europe Catheter Securement Device Industry Revenue (billion) Forecast, by Application 2020 & 2033
- Table 36: Rest of Europe Catheter Securement Device Industry Volume (K Units) Forecast, by Application 2020 & 2033
- Table 37: Global Catheter Securement Device Industry Revenue billion Forecast, by Product 2020 & 2033
- Table 38: Global Catheter Securement Device Industry Volume K Units Forecast, by Product 2020 & 2033
- Table 39: Global Catheter Securement Device Industry Revenue billion Forecast, by End-User 2020 & 2033
- Table 40: Global Catheter Securement Device Industry Volume K Units Forecast, by End-User 2020 & 2033
- Table 41: Global Catheter Securement Device Industry Revenue billion Forecast, by Country 2020 & 2033
- Table 42: Global Catheter Securement Device Industry Volume K Units Forecast, by Country 2020 & 2033
- Table 43: China Catheter Securement Device Industry Revenue (billion) Forecast, by Application 2020 & 2033
- Table 44: China Catheter Securement Device Industry Volume (K Units) Forecast, by Application 2020 & 2033
- Table 45: Japan Catheter Securement Device Industry Revenue (billion) Forecast, by Application 2020 & 2033
- Table 46: Japan Catheter Securement Device Industry Volume (K Units) Forecast, by Application 2020 & 2033
- Table 47: India Catheter Securement Device Industry Revenue (billion) Forecast, by Application 2020 & 2033
- Table 48: India Catheter Securement Device Industry Volume (K Units) Forecast, by Application 2020 & 2033
- Table 49: Australia Catheter Securement Device Industry Revenue (billion) Forecast, by Application 2020 & 2033
- Table 50: Australia Catheter Securement Device Industry Volume (K Units) Forecast, by Application 2020 & 2033
- Table 51: South Korea Catheter Securement Device Industry Revenue (billion) Forecast, by Application 2020 & 2033
- Table 52: South Korea Catheter Securement Device Industry Volume (K Units) Forecast, by Application 2020 & 2033
- Table 53: Rest of Asia Pacific Catheter Securement Device Industry Revenue (billion) Forecast, by Application 2020 & 2033
- Table 54: Rest of Asia Pacific Catheter Securement Device Industry Volume (K Units) Forecast, by Application 2020 & 2033
- Table 55: Global Catheter Securement Device Industry Revenue billion Forecast, by Product 2020 & 2033
- Table 56: Global Catheter Securement Device Industry Volume K Units Forecast, by Product 2020 & 2033
- Table 57: Global Catheter Securement Device Industry Revenue billion Forecast, by End-User 2020 & 2033
- Table 58: Global Catheter Securement Device Industry Volume K Units Forecast, by End-User 2020 & 2033
- Table 59: Global Catheter Securement Device Industry Revenue billion Forecast, by Country 2020 & 2033
- Table 60: Global Catheter Securement Device Industry Volume K Units Forecast, by Country 2020 & 2033
- Table 61: GCC Catheter Securement Device Industry Revenue (billion) Forecast, by Application 2020 & 2033
- Table 62: GCC Catheter Securement Device Industry Volume (K Units) Forecast, by Application 2020 & 2033
- Table 63: South Africa Catheter Securement Device Industry Revenue (billion) Forecast, by Application 2020 & 2033
- Table 64: South Africa Catheter Securement Device Industry Volume (K Units) Forecast, by Application 2020 & 2033
- Table 65: Rest of Middle East and Africa Catheter Securement Device Industry Revenue (billion) Forecast, by Application 2020 & 2033
- Table 66: Rest of Middle East and Africa Catheter Securement Device Industry Volume (K Units) Forecast, by Application 2020 & 2033
- Table 67: Global Catheter Securement Device Industry Revenue billion Forecast, by Product 2020 & 2033
- Table 68: Global Catheter Securement Device Industry Volume K Units Forecast, by Product 2020 & 2033
- Table 69: Global Catheter Securement Device Industry Revenue billion Forecast, by End-User 2020 & 2033
- Table 70: Global Catheter Securement Device Industry Volume K Units Forecast, by End-User 2020 & 2033
- Table 71: Global Catheter Securement Device Industry Revenue billion Forecast, by Country 2020 & 2033
- Table 72: Global Catheter Securement Device Industry Volume K Units Forecast, by Country 2020 & 2033
- Table 73: Brazil Catheter Securement Device Industry Revenue (billion) Forecast, by Application 2020 & 2033
- Table 74: Brazil Catheter Securement Device Industry Volume (K Units) Forecast, by Application 2020 & 2033
- Table 75: Argentina Catheter Securement Device Industry Revenue (billion) Forecast, by Application 2020 & 2033
- Table 76: Argentina Catheter Securement Device Industry Volume (K Units) Forecast, by Application 2020 & 2033
- Table 77: Rest of South America Catheter Securement Device Industry Revenue (billion) Forecast, by Application 2020 & 2033
- Table 78: Rest of South America Catheter Securement Device Industry Volume (K Units) Forecast, by Application 2020 & 2033
Frequently Asked Questions
1. What is the projected Compound Annual Growth Rate (CAGR) of the Catheter Securement Device Industry?
The projected CAGR is approximately 6.4%.
2. Which companies are prominent players in the Catheter Securement Device Industry?
Key companies in the market include Argon Medical Devices, Merit Medical Systems, Tractus Vascular, Levity Products Incorporated, BD (C R Bard), 3M, Baxter, ConvaTec Group PLC, Medtronic PLC, TIDI Products LLC, B Braun SE, Medline Industries Inc, CATHETRIX.
3. What are the main segments of the Catheter Securement Device Industry?
The market segments include Product, End-User.
4. Can you provide details about the market size?
The market size is estimated to be USD 1.29 billion as of 2022.
5. What are some drivers contributing to market growth?
The increasing number of surgical procedures worldwide contributes to the demand for catheter securement devices. Catheters are commonly used in various surgeries. necessitating effective securement solutions.
6. What are the notable trends driving market growth?
There is a growing trend toward the use of disposable catheter securement devices due to concerns about infection control and convenience. Single-use products help minimize the risk of contamination and ensure patient safety.
7. Are there any restraints impacting market growth?
Budget constraints in healthcare facilities can limit the adoption of advanced catheter securement devices. Hospitals may prioritize cost-effective solutions. potentially impacting the market for premium products.
8. Can you provide examples of recent developments in the market?
April 2022: Bedal and Gulf Medical Co. Ltd. (GMC) signed a distribution agreement for Saudi Arabia and Bahrain. The Bedal products will be distributed by GMC in the Saudi Arabian and Bahraini markets, representing a major part of the market for the usage of catheter stabilization devices in the Gulf region.
9. What pricing options are available for accessing the report?
Pricing options include single-user, multi-user, and enterprise licenses priced at USD 4750, USD 5250, and USD 8750 respectively.
10. Is the market size provided in terms of value or volume?
The market size is provided in terms of value, measured in billion and volume, measured in K Units.
11. Are there any specific market keywords associated with the report?
Yes, the market keyword associated with the report is "Catheter Securement Device Industry," which aids in identifying and referencing the specific market segment covered.
12. How do I determine which pricing option suits my needs best?
The pricing options vary based on user requirements and access needs. Individual users may opt for single-user licenses, while businesses requiring broader access may choose multi-user or enterprise licenses for cost-effective access to the report.
13. Are there any additional resources or data provided in the Catheter Securement Device Industry report?
While the report offers comprehensive insights, it's advisable to review the specific contents or supplementary materials provided to ascertain if additional resources or data are available.
14. How can I stay updated on further developments or reports in the Catheter Securement Device Industry?
To stay informed about further developments, trends, and reports in the Catheter Securement Device Industry, consider subscribing to industry newsletters, following relevant companies and organizations, or regularly checking reputable industry news sources and publications.
Methodology
Step 1 - Identification of Relevant Samples Size from Population Database



Step 2 - Approaches for Defining Global Market Size (Value, Volume* & Price*)

Note*: In applicable scenarios
Step 3 - Data Sources
Primary Research
- Web Analytics
- Survey Reports
- Research Institute
- Latest Research Reports
- Opinion Leaders
Secondary Research
- Annual Reports
- White Paper
- Latest Press Release
- Industry Association
- Paid Database
- Investor Presentations

Step 4 - Data Triangulation
Involves using different sources of information in order to increase the validity of a study
These sources are likely to be stakeholders in a program - participants, other researchers, program staff, other community members, and so on.
Then we put all data in single framework & apply various statistical tools to find out the dynamic on the market.
During the analysis stage, feedback from the stakeholder groups would be compared to determine areas of agreement as well as areas of divergence


