Key Insights
The global Gene Engineered Subunit Vaccine market is poised for significant expansion, projected to reach $12.47 billion by 2025, with a robust Compound Annual Growth Rate (CAGR) of 11.4% anticipated from 2025 to 2033. This impressive growth is underpinned by several powerful market drivers. The increasing prevalence of infectious diseases, coupled with a growing global emphasis on preventative healthcare, is a primary catalyst. Furthermore, advancements in genetic engineering and biotechnology are enabling the development of more targeted, effective, and safer vaccines, thereby accelerating market adoption. The demand for both human and veterinary applications, across therapeutic and preventative vaccine types, underscores the broad utility and expanding reach of this vaccine technology. Key companies such as Merck, GSK, Pfizer, and Sanofi Pasteur SA are at the forefront of this innovation, investing heavily in research and development to bring novel gene-engineered subunit vaccines to market.
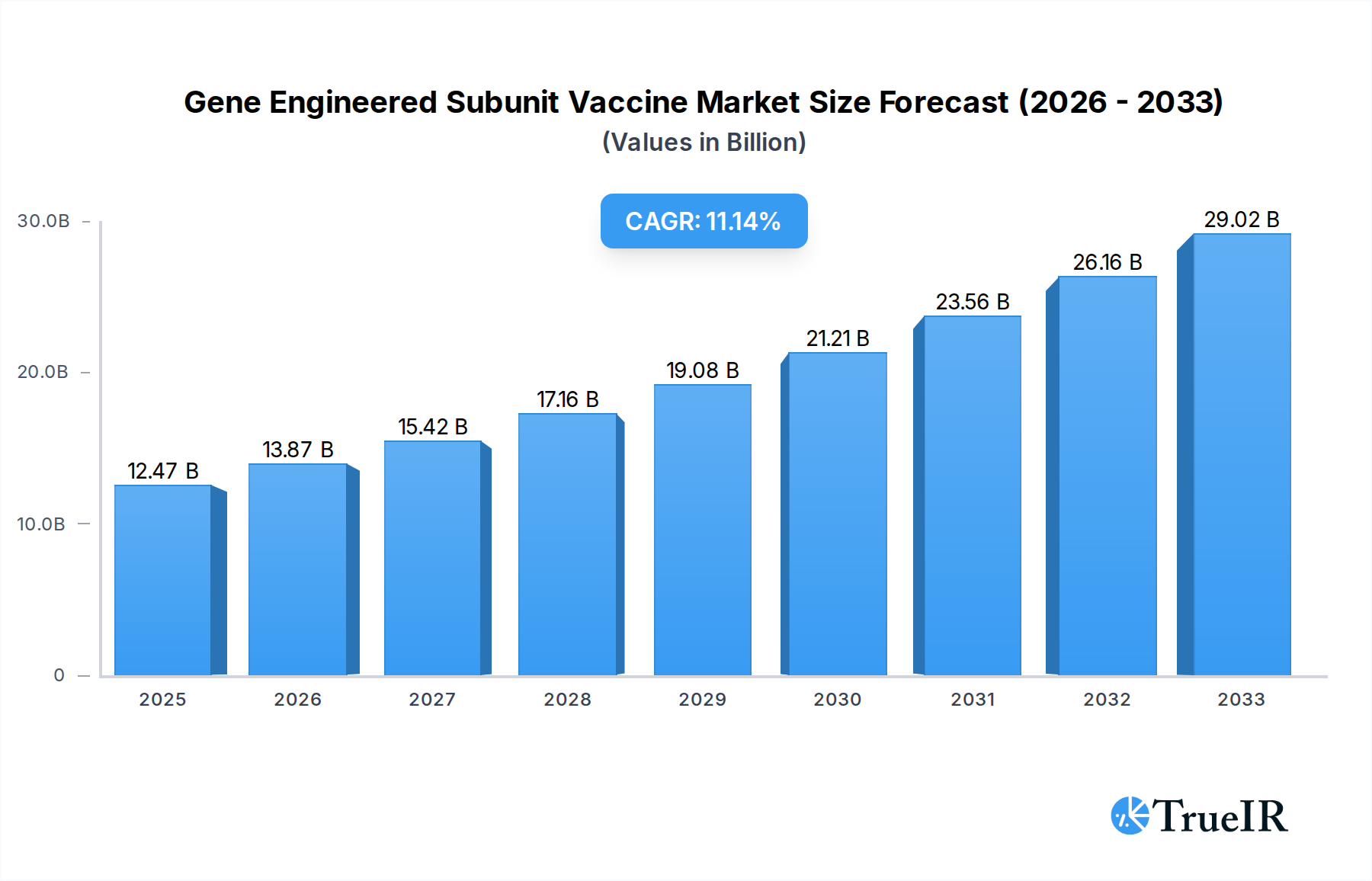
Gene Engineered Subunit Vaccine Market Size (In Billion)

The market is characterized by a dynamic landscape of trends, including a heightened focus on personalized medicine and the development of vaccines against emerging pathogens. The growing acceptance and trust in scientifically advanced vaccine technologies, especially post-pandemic, are further bolstering market sentiment. While the market presents immense opportunities, certain restraints, such as high research and development costs and stringent regulatory hurdles for new vaccine approvals, could temper the pace of growth. However, the inherent advantages of gene-engineered subunit vaccines, including their enhanced safety profiles and ability to elicit strong immune responses, are expected to outweigh these challenges. Geographically, North America and Europe are expected to lead the market due to advanced healthcare infrastructure and high R&D expenditure. The Asia Pacific region, driven by China and India, is also emerging as a significant growth hub owing to a large patient pool and increasing investments in biotechnology.
Gene Engineered Subunit Vaccine Company Market Share

Here's a dynamic, SEO-optimized report description for Gene Engineered Subunit Vaccines, crafted to engage industry audiences and enhance search rankings.
This in-depth report provides a definitive analysis of the global Gene Engineered Subunit Vaccine market, exploring its current landscape, emerging trends, and future trajectory. Leveraging high-volume keywords like "gene engineered vaccines," "subunit vaccines," "biotechnology," "vaccine development," and "biologics," this study is essential for stakeholders seeking to understand market dynamics, competitive strategies, and investment opportunities. The report covers the Study Period of 2019–2033, with a Base Year and Estimated Year of 2025, and a detailed Forecast Period from 2025–2033, building upon a thorough Historical Period of 2019–2024.
Gene Engineered Subunit Vaccine Market Structure & Competitive Landscape
The global Gene Engineered Subunit Vaccine market exhibits a dynamic and evolving structure, characterized by significant innovation and strategic partnerships. Market concentration is moderate, with key players investing heavily in research and development to drive technological advancements. Innovation drivers include the pursuit of vaccines with enhanced efficacy, improved safety profiles, and broader target pathogen coverage, particularly in the face of emerging infectious diseases and the growing demand for personalized medicine. Regulatory impacts are substantial, with stringent approval processes and evolving guidelines influencing market entry and product development timelines. Product substitutes, while present in the broader vaccine market, are less direct for highly specific gene-engineered subunit vaccines, though traditional vaccine types continue to represent a competitive benchmark. End-user segmentation spans both human and veterinary applications, with a notable shift towards more targeted and sophisticated therapeutic and preventative solutions. Mergers and acquisitions (M&A) play a crucial role in consolidating market share and acquiring novel technologies, with an estimated volume of over fifty billion dollars in M&A activities observed throughout the historical period.
- Key Market Concentration Factors:
- High R&D expenditure by leading biopharmaceutical companies.
- Intellectual property protection and patent exclusivity.
- Establishment of robust manufacturing capabilities.
- Innovation Drivers:
- Development of novel antigen delivery systems.
- Platform technologies enabling rapid vaccine development.
- Targeting of complex diseases with high unmet needs.
- Regulatory Impacts:
- Streamlined approval pathways for pandemic-related vaccines.
- Increased scrutiny on manufacturing quality and biosafety.
- Global harmonization of regulatory standards.
- M&A Trends:
- Acquisition of early-stage biotech firms with promising vaccine platforms.
- Strategic alliances for co-development and commercialization.
- Consolidation to achieve economies of scale in production.
Gene Engineered Subunit Vaccine Market Trends & Opportunities
The global Gene Engineered Subunit Vaccine market is poised for remarkable expansion, driven by a confluence of factors including increasing global health expenditure, a heightened awareness of disease prevention, and significant advancements in biotechnology. The market size is projected to grow at a Compound Annual Growth Rate (CAGR) of over 15% from 2025 to 2033, potentially reaching a valuation exceeding one trillion dollars by the end of the forecast period. Technological shifts are central to this growth, with mRNA and viral vector platforms revolutionizing vaccine design and accelerating development cycles. Consumer preferences are increasingly leaning towards vaccines that offer superior immunogenicity and reduced side effects compared to traditional formulations. The competitive dynamics are intensifying, with both established pharmaceutical giants and agile biotechnology startups vying for market leadership through innovation and strategic collaborations. Opportunities abound in addressing unmet medical needs, particularly for chronic diseases, neglected tropical diseases, and emerging zoonotic threats. The increasing adoption of gene-editing technologies in vaccine research further enhances the potential for highly precise and effective preventative and therapeutic agents. Market penetration rates for advanced subunit vaccines are expected to rise significantly as manufacturing scales up and regulatory approvals broaden across diverse therapeutic areas.
The growing prevalence of chronic diseases, such as cancer and autoimmune disorders, presents a substantial opportunity for therapeutic gene engineered subunit vaccines. These advanced biologicals offer a more targeted approach to stimulating the immune system, paving the way for personalized cancer immunotherapies and treatments for debilitating autoimmune conditions. Furthermore, the persistent threat of global pandemics and the rise of antimicrobial resistance are driving unprecedented demand for novel vaccine solutions. Gene engineered subunit vaccines, with their inherent flexibility and rapid development potential, are at the forefront of this response. The investment landscape is also showing robust growth, with venture capital and government funding flowing into companies at the cutting edge of vaccine innovation. This capital infusion is critical for scaling up manufacturing capabilities, conducting extensive clinical trials, and navigating the complex regulatory pathways. The increasing global demand for animal health solutions also represents a significant untapped market for veterinary gene engineered subunit vaccines, offering protection against a wide range of diseases in livestock and companion animals.
Dominant Markets & Segments in Gene Engineered Subunit Vaccine
The Human Use segment is overwhelmingly dominant within the Gene Engineered Subunit Vaccine market, driven by extensive public health initiatives, significant healthcare spending, and a robust pipeline of innovative products targeting a wide array of human diseases. Within this, the Preventative Gene Engineered Subunit Vaccine category commands the largest share, fueled by ongoing vaccination programs against infectious diseases and the development of next-generation vaccines offering broader protection and improved safety profiles.
Leading Region: North America stands out as the dominant market, primarily due to its advanced healthcare infrastructure, substantial government and private sector investment in R&D, and a proactive regulatory environment that fosters innovation. The United States, in particular, is a hub for vaccine development and commercialization, with numerous leading companies headquartered and operating within its borders.
Key Growth Drivers in Dominant Segments:
- Human Use - Preventative Vaccines:
- Infrastructure: Extensive public health networks and established vaccination infrastructures facilitating widespread delivery.
- Policies: Government mandates for childhood vaccinations and national immunization programs.
- R&D Investment: Significant funding allocated to infectious disease prevention research.
- Disease Burden: Persistent threats from influenza, respiratory syncytial virus (RSV), and emerging infectious diseases.
- Therapeutic Gene Engineered Subunit Vaccine (Human Use):
- Technological Advancements: Breakthroughs in immunotherapy and personalized medicine.
- Unmet Medical Needs: Growing demand for effective treatments for cancers, autoimmune diseases, and chronic viral infections.
- Clinical Trial Success: Positive outcomes in late-stage clinical trials demonstrating efficacy.
- Regulatory Support: Evolving pathways for the approval of novel therapeutic biologics.
The Veterinary Use segment, while smaller, is exhibiting strong growth, driven by the increasing recognition of animal welfare, the economic importance of livestock health, and the demand for safe and effective vaccines for companion animals. The Preventative Gene Engineered Subunit Vaccine type also leads within veterinary applications, addressing diseases that impact food security and pet health.
Growth Drivers in Veterinary Segment:
- Infrastructure: Expanding veterinary healthcare systems and animal production facilities.
- Policies: Government initiatives to control animal diseases and ensure food safety.
- Economic Impact: Significant losses averted through effective animal disease prevention.
- Companion Animal Market: Growing pet ownership and increased spending on animal health.
Gene Engineered Subunit Vaccine Product Analysis
Gene engineered subunit vaccines represent a paradigm shift in vaccine technology, offering precise antigen presentation and enhanced immunogenicity. Innovations are centered on novel delivery platforms, such as nanoparticles and recombinant protein expression systems, enabling the development of vaccines with superior efficacy and reduced side effects. Their applications span the prevention and treatment of infectious diseases, cancer immunotherapies, and autoimmune disorders. Competitive advantages lie in their targeted immune response, manufacturing scalability, and potential for combination therapies. Technological advancements are continuously refining antigen design and adjuvant formulations, leading to more potent and durable immune protection.
Key Drivers, Barriers & Challenges in Gene Engineered Subunit Vaccine
Key Drivers:
- Technological Advancements: Rapid progress in genetic engineering, protein expression, and adjuvant technologies.
- Economic Factors: Increasing healthcare expenditure and government investment in public health.
- Policy-Driven Factors: Supportive regulatory environments and global health initiatives promoting vaccine development and access.
- Pandemic Preparedness: Heightened global focus on developing rapid-response vaccines against emerging infectious diseases.
Barriers & Challenges:
- Regulatory Hurdles: Stringent and often lengthy approval processes for novel biologics.
- Supply Chain Issues: Complex manufacturing requirements and global distribution challenges, impacting the availability of raw materials and finished products.
- Competitive Pressures: Intense competition from established vaccine manufacturers and the need for continuous innovation.
- Cost of Development: High R&D expenses and significant investment required for clinical trials and manufacturing scale-up, estimated to be in the tens of billions of dollars for blockbuster vaccines.
- Public Perception and Acceptance: Addressing concerns regarding vaccine safety and efficacy requires sustained public education efforts.
Growth Drivers in the Gene Engineered Subunit Vaccine Market
The Gene Engineered Subunit Vaccine market is propelled by a potent combination of technological innovation and escalating global health demands. Technological drivers include the maturation of platforms like mRNA, viral vectors, and protein-based subunit technologies, which enable faster development and improved vaccine profiles. Economic drivers are evident in the substantial increase in global healthcare spending and government mandates for infectious disease prevention, projected to reach over two trillion dollars annually in the forecast period. Regulatory factors play a dual role; while a barrier, they also foster growth through incentivized pathways for pandemic preparedness and support for novel therapeutic agents, creating a dynamic and opportunity-rich environment.
Challenges Impacting Gene Engineered Subunit Vaccine Growth
Despite immense potential, the Gene Engineered Subunit Vaccine market faces significant headwinds. Regulatory complexities remain a primary challenge, with evolving guidelines and lengthy approval processes potentially delaying market entry. Supply chain disruptions, exacerbated by geopolitical factors and the need for specialized manufacturing facilities, can hinder production scalability and global distribution, with estimated supply chain costs potentially reaching hundreds of billions of dollars annually. Competitive pressures are intense, requiring continuous innovation and substantial investment to differentiate products and secure market share. Furthermore, the high cost of research and development, estimated in the billions of dollars for each successful vaccine candidate, presents a financial barrier for smaller players.
Key Players Shaping the Gene Engineered Subunit Vaccine Market
- Merck
- GSK
- Tiantan
- Pulike
- Greffex
- CureVac
- Yebio
- Sanofi Pasteur SA
- Virbac
- Pfizer Inc.
- Walvax Biotechnology
- Kontec
- Novavax
- Sanofi Pasteur
- Bharat Biotech
- GlaxoSmithKline
- Pfizer
- Moderna
Significant Gene Engineered Subunit Vaccine Industry Milestones
- 2019: Emergence of early-stage research highlighting the potential of mRNA technology for therapeutic applications beyond infectious diseases.
- 2020 January: Initial reports of a novel coronavirus, spurring unprecedented global R&D efforts.
- 2020 March: Initiation of the first clinical trials for mRNA-based COVID-19 vaccines, demonstrating accelerated development timelines.
- 2020 December: Emergency Use Authorization granted for the first mRNA COVID-19 vaccines, marking a pivotal moment in vaccine history.
- 2021 Q1: Advancements in protein-subunit vaccine development for respiratory syncytial virus (RSV), showing promising efficacy.
- 2021 Q3: Successful large-scale manufacturing and global distribution of COVID-19 vaccines, overcoming significant logistical hurdles.
- 2022 Q1: Progress in therapeutic cancer vaccine trials, utilizing gene-engineered subunit approaches for solid tumors.
- 2022 Q4: Increased investment in platform technologies capable of rapid adaptation to new viral variants.
- 2023 Q2: Regulatory submissions for novel gene-engineered vaccines targeting other high-burden infectious diseases, such as malaria and dengue.
- 2023 Q4: Strategic partnerships announced for the co-development of combination vaccines, integrating multiple antigen targets.
- 2024 Q1: Emerging data from veterinary vaccine trials demonstrating enhanced protective immunity against livestock diseases.
Future Outlook for Gene Engineered Subunit Vaccine Market
The future of the Gene Engineered Subunit Vaccine market is exceptionally bright, characterized by sustained innovation and expanding applications. Growth catalysts include the ongoing research into personalized cancer vaccines, the development of broad-spectrum antivirals, and the increasing use of these technologies for treating autoimmune and chronic diseases. Strategic opportunities lie in leveraging established platforms for rapid response to future pandemics, expanding into underserved veterinary markets, and forging collaborative ventures to accelerate commercialization. The market is projected to witness significant growth, driven by a commitment to tackling global health challenges and advancing precision medicine, with a potential market size exceeding two trillion dollars in the long term.
Gene Engineered Subunit Vaccine Segmentation
-
1. Application
- 1.1. Human Use
- 1.2. Veterinary Use
-
2. Type
- 2.1. Therapeutic Gene Engineered Subunit Vaccine
- 2.2. Preventative Gene Engineered Subunit Vaccine
Gene Engineered Subunit Vaccine Segmentation By Geography
-
1. North America
- 1.1. United States
- 1.2. Canada
- 1.3. Mexico
-
2. South America
- 2.1. Brazil
- 2.2. Argentina
- 2.3. Rest of South America
-
3. Europe
- 3.1. United Kingdom
- 3.2. Germany
- 3.3. France
- 3.4. Italy
- 3.5. Spain
- 3.6. Russia
- 3.7. Benelux
- 3.8. Nordics
- 3.9. Rest of Europe
-
4. Middle East & Africa
- 4.1. Turkey
- 4.2. Israel
- 4.3. GCC
- 4.4. North Africa
- 4.5. South Africa
- 4.6. Rest of Middle East & Africa
-
5. Asia Pacific
- 5.1. China
- 5.2. India
- 5.3. Japan
- 5.4. South Korea
- 5.5. ASEAN
- 5.6. Oceania
- 5.7. Rest of Asia Pacific
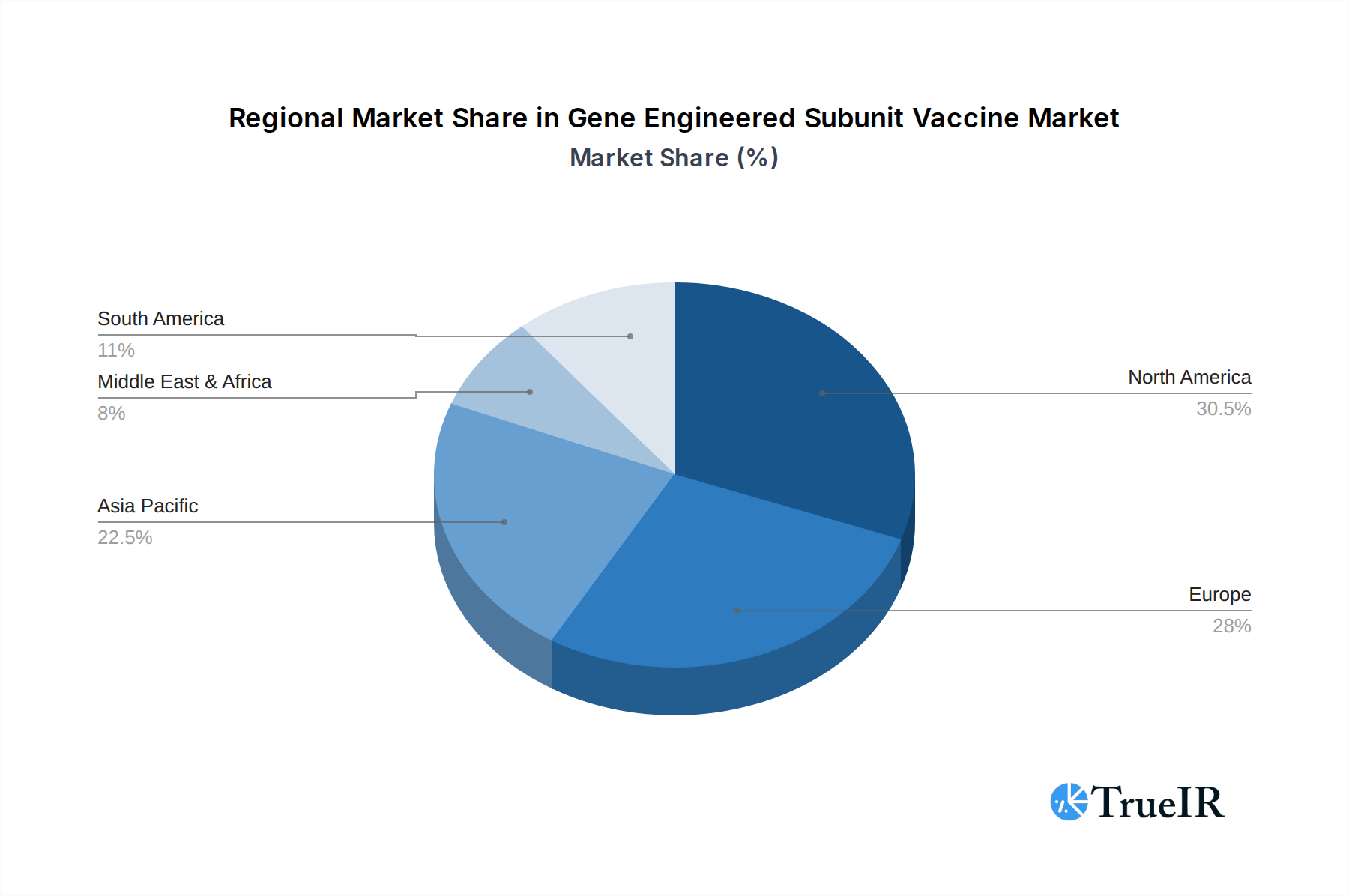
Gene Engineered Subunit Vaccine Regional Market Share

Geographic Coverage of Gene Engineered Subunit Vaccine
Gene Engineered Subunit Vaccine REPORT HIGHLIGHTS
| Aspects | Details |
|---|---|
| Study Period | 2020-2034 |
| Base Year | 2025 |
| Estimated Year | 2026 |
| Forecast Period | 2026-2034 |
| Historical Period | 2020-2025 |
| Growth Rate | CAGR of 11.4% from 2020-2034 |
| Segmentation |
|
Table of Contents
- 1. Introduction
- 1.1. Research Scope
- 1.2. Market Segmentation
- 1.3. Research Methodology
- 1.4. Definitions and Assumptions
- 2. Executive Summary
- 2.1. Introduction
- 3. Market Dynamics
- 3.1. Introduction
- 3.2. Market Drivers
- 3.3. Market Restrains
- 3.4. Market Trends
- 4. Market Factor Analysis
- 4.1. Porters Five Forces
- 4.2. Supply/Value Chain
- 4.3. PESTEL analysis
- 4.4. Market Entropy
- 4.5. Patent/Trademark Analysis
- 5. Global Gene Engineered Subunit Vaccine Analysis, Insights and Forecast, 2020-2032
- 5.1. Market Analysis, Insights and Forecast - by Application
- 5.1.1. Human Use
- 5.1.2. Veterinary Use
- 5.2. Market Analysis, Insights and Forecast - by Type
- 5.2.1. Therapeutic Gene Engineered Subunit Vaccine
- 5.2.2. Preventative Gene Engineered Subunit Vaccine
- 5.3. Market Analysis, Insights and Forecast - by Region
- 5.3.1. North America
- 5.3.2. South America
- 5.3.3. Europe
- 5.3.4. Middle East & Africa
- 5.3.5. Asia Pacific
- 5.1. Market Analysis, Insights and Forecast - by Application
- 6. North America Gene Engineered Subunit Vaccine Analysis, Insights and Forecast, 2020-2032
- 6.1. Market Analysis, Insights and Forecast - by Application
- 6.1.1. Human Use
- 6.1.2. Veterinary Use
- 6.2. Market Analysis, Insights and Forecast - by Type
- 6.2.1. Therapeutic Gene Engineered Subunit Vaccine
- 6.2.2. Preventative Gene Engineered Subunit Vaccine
- 6.1. Market Analysis, Insights and Forecast - by Application
- 7. South America Gene Engineered Subunit Vaccine Analysis, Insights and Forecast, 2020-2032
- 7.1. Market Analysis, Insights and Forecast - by Application
- 7.1.1. Human Use
- 7.1.2. Veterinary Use
- 7.2. Market Analysis, Insights and Forecast - by Type
- 7.2.1. Therapeutic Gene Engineered Subunit Vaccine
- 7.2.2. Preventative Gene Engineered Subunit Vaccine
- 7.1. Market Analysis, Insights and Forecast - by Application
- 8. Europe Gene Engineered Subunit Vaccine Analysis, Insights and Forecast, 2020-2032
- 8.1. Market Analysis, Insights and Forecast - by Application
- 8.1.1. Human Use
- 8.1.2. Veterinary Use
- 8.2. Market Analysis, Insights and Forecast - by Type
- 8.2.1. Therapeutic Gene Engineered Subunit Vaccine
- 8.2.2. Preventative Gene Engineered Subunit Vaccine
- 8.1. Market Analysis, Insights and Forecast - by Application
- 9. Middle East & Africa Gene Engineered Subunit Vaccine Analysis, Insights and Forecast, 2020-2032
- 9.1. Market Analysis, Insights and Forecast - by Application
- 9.1.1. Human Use
- 9.1.2. Veterinary Use
- 9.2. Market Analysis, Insights and Forecast - by Type
- 9.2.1. Therapeutic Gene Engineered Subunit Vaccine
- 9.2.2. Preventative Gene Engineered Subunit Vaccine
- 9.1. Market Analysis, Insights and Forecast - by Application
- 10. Asia Pacific Gene Engineered Subunit Vaccine Analysis, Insights and Forecast, 2020-2032
- 10.1. Market Analysis, Insights and Forecast - by Application
- 10.1.1. Human Use
- 10.1.2. Veterinary Use
- 10.2. Market Analysis, Insights and Forecast - by Type
- 10.2.1. Therapeutic Gene Engineered Subunit Vaccine
- 10.2.2. Preventative Gene Engineered Subunit Vaccine
- 10.1. Market Analysis, Insights and Forecast - by Application
- 11. Competitive Analysis
- 11.1. Global Market Share Analysis 2025
- 11.2. Company Profiles
- 11.2.1 Merck
- 11.2.1.1. Overview
- 11.2.1.2. Products
- 11.2.1.3. SWOT Analysis
- 11.2.1.4. Recent Developments
- 11.2.1.5. Financials (Based on Availability)
- 11.2.2 GSK
- 11.2.2.1. Overview
- 11.2.2.2. Products
- 11.2.2.3. SWOT Analysis
- 11.2.2.4. Recent Developments
- 11.2.2.5. Financials (Based on Availability)
- 11.2.3 Tiantan
- 11.2.3.1. Overview
- 11.2.3.2. Products
- 11.2.3.3. SWOT Analysis
- 11.2.3.4. Recent Developments
- 11.2.3.5. Financials (Based on Availability)
- 11.2.4 Pulike
- 11.2.4.1. Overview
- 11.2.4.2. Products
- 11.2.4.3. SWOT Analysis
- 11.2.4.4. Recent Developments
- 11.2.4.5. Financials (Based on Availability)
- 11.2.5 Greffex
- 11.2.5.1. Overview
- 11.2.5.2. Products
- 11.2.5.3. SWOT Analysis
- 11.2.5.4. Recent Developments
- 11.2.5.5. Financials (Based on Availability)
- 11.2.6 CureVac
- 11.2.6.1. Overview
- 11.2.6.2. Products
- 11.2.6.3. SWOT Analysis
- 11.2.6.4. Recent Developments
- 11.2.6.5. Financials (Based on Availability)
- 11.2.7 Yebio
- 11.2.7.1. Overview
- 11.2.7.2. Products
- 11.2.7.3. SWOT Analysis
- 11.2.7.4. Recent Developments
- 11.2.7.5. Financials (Based on Availability)
- 11.2.8 Sanofi Pasteur SA
- 11.2.8.1. Overview
- 11.2.8.2. Products
- 11.2.8.3. SWOT Analysis
- 11.2.8.4. Recent Developments
- 11.2.8.5. Financials (Based on Availability)
- 11.2.9 Virbac
- 11.2.9.1. Overview
- 11.2.9.2. Products
- 11.2.9.3. SWOT Analysis
- 11.2.9.4. Recent Developments
- 11.2.9.5. Financials (Based on Availability)
- 11.2.10 Pfizer Inc.
- 11.2.10.1. Overview
- 11.2.10.2. Products
- 11.2.10.3. SWOT Analysis
- 11.2.10.4. Recent Developments
- 11.2.10.5. Financials (Based on Availability)
- 11.2.11 Walvax Biotechnology
- 11.2.11.1. Overview
- 11.2.11.2. Products
- 11.2.11.3. SWOT Analysis
- 11.2.11.4. Recent Developments
- 11.2.11.5. Financials (Based on Availability)
- 11.2.12 Kontec
- 11.2.12.1. Overview
- 11.2.12.2. Products
- 11.2.12.3. SWOT Analysis
- 11.2.12.4. Recent Developments
- 11.2.12.5. Financials (Based on Availability)
- 11.2.13 Novavax
- 11.2.13.1. Overview
- 11.2.13.2. Products
- 11.2.13.3. SWOT Analysis
- 11.2.13.4. Recent Developments
- 11.2.13.5. Financials (Based on Availability)
- 11.2.14 Sanofi Pasteur
- 11.2.14.1. Overview
- 11.2.14.2. Products
- 11.2.14.3. SWOT Analysis
- 11.2.14.4. Recent Developments
- 11.2.14.5. Financials (Based on Availability)
- 11.2.15 Bharat Biotech
- 11.2.15.1. Overview
- 11.2.15.2. Products
- 11.2.15.3. SWOT Analysis
- 11.2.15.4. Recent Developments
- 11.2.15.5. Financials (Based on Availability)
- 11.2.16 GlaxoSmithKline
- 11.2.16.1. Overview
- 11.2.16.2. Products
- 11.2.16.3. SWOT Analysis
- 11.2.16.4. Recent Developments
- 11.2.16.5. Financials (Based on Availability)
- 11.2.17 Pfizer
- 11.2.17.1. Overview
- 11.2.17.2. Products
- 11.2.17.3. SWOT Analysis
- 11.2.17.4. Recent Developments
- 11.2.17.5. Financials (Based on Availability)
- 11.2.18 Moderna
- 11.2.18.1. Overview
- 11.2.18.2. Products
- 11.2.18.3. SWOT Analysis
- 11.2.18.4. Recent Developments
- 11.2.18.5. Financials (Based on Availability)
- 11.2.1 Merck
List of Figures
- Figure 1: Global Gene Engineered Subunit Vaccine Revenue Breakdown (undefined, %) by Region 2025 & 2033
- Figure 2: Global Gene Engineered Subunit Vaccine Volume Breakdown (K, %) by Region 2025 & 2033
- Figure 3: North America Gene Engineered Subunit Vaccine Revenue (undefined), by Application 2025 & 2033
- Figure 4: North America Gene Engineered Subunit Vaccine Volume (K), by Application 2025 & 2033
- Figure 5: North America Gene Engineered Subunit Vaccine Revenue Share (%), by Application 2025 & 2033
- Figure 6: North America Gene Engineered Subunit Vaccine Volume Share (%), by Application 2025 & 2033
- Figure 7: North America Gene Engineered Subunit Vaccine Revenue (undefined), by Type 2025 & 2033
- Figure 8: North America Gene Engineered Subunit Vaccine Volume (K), by Type 2025 & 2033
- Figure 9: North America Gene Engineered Subunit Vaccine Revenue Share (%), by Type 2025 & 2033
- Figure 10: North America Gene Engineered Subunit Vaccine Volume Share (%), by Type 2025 & 2033
- Figure 11: North America Gene Engineered Subunit Vaccine Revenue (undefined), by Country 2025 & 2033
- Figure 12: North America Gene Engineered Subunit Vaccine Volume (K), by Country 2025 & 2033
- Figure 13: North America Gene Engineered Subunit Vaccine Revenue Share (%), by Country 2025 & 2033
- Figure 14: North America Gene Engineered Subunit Vaccine Volume Share (%), by Country 2025 & 2033
- Figure 15: South America Gene Engineered Subunit Vaccine Revenue (undefined), by Application 2025 & 2033
- Figure 16: South America Gene Engineered Subunit Vaccine Volume (K), by Application 2025 & 2033
- Figure 17: South America Gene Engineered Subunit Vaccine Revenue Share (%), by Application 2025 & 2033
- Figure 18: South America Gene Engineered Subunit Vaccine Volume Share (%), by Application 2025 & 2033
- Figure 19: South America Gene Engineered Subunit Vaccine Revenue (undefined), by Type 2025 & 2033
- Figure 20: South America Gene Engineered Subunit Vaccine Volume (K), by Type 2025 & 2033
- Figure 21: South America Gene Engineered Subunit Vaccine Revenue Share (%), by Type 2025 & 2033
- Figure 22: South America Gene Engineered Subunit Vaccine Volume Share (%), by Type 2025 & 2033
- Figure 23: South America Gene Engineered Subunit Vaccine Revenue (undefined), by Country 2025 & 2033
- Figure 24: South America Gene Engineered Subunit Vaccine Volume (K), by Country 2025 & 2033
- Figure 25: South America Gene Engineered Subunit Vaccine Revenue Share (%), by Country 2025 & 2033
- Figure 26: South America Gene Engineered Subunit Vaccine Volume Share (%), by Country 2025 & 2033
- Figure 27: Europe Gene Engineered Subunit Vaccine Revenue (undefined), by Application 2025 & 2033
- Figure 28: Europe Gene Engineered Subunit Vaccine Volume (K), by Application 2025 & 2033
- Figure 29: Europe Gene Engineered Subunit Vaccine Revenue Share (%), by Application 2025 & 2033
- Figure 30: Europe Gene Engineered Subunit Vaccine Volume Share (%), by Application 2025 & 2033
- Figure 31: Europe Gene Engineered Subunit Vaccine Revenue (undefined), by Type 2025 & 2033
- Figure 32: Europe Gene Engineered Subunit Vaccine Volume (K), by Type 2025 & 2033
- Figure 33: Europe Gene Engineered Subunit Vaccine Revenue Share (%), by Type 2025 & 2033
- Figure 34: Europe Gene Engineered Subunit Vaccine Volume Share (%), by Type 2025 & 2033
- Figure 35: Europe Gene Engineered Subunit Vaccine Revenue (undefined), by Country 2025 & 2033
- Figure 36: Europe Gene Engineered Subunit Vaccine Volume (K), by Country 2025 & 2033
- Figure 37: Europe Gene Engineered Subunit Vaccine Revenue Share (%), by Country 2025 & 2033
- Figure 38: Europe Gene Engineered Subunit Vaccine Volume Share (%), by Country 2025 & 2033
- Figure 39: Middle East & Africa Gene Engineered Subunit Vaccine Revenue (undefined), by Application 2025 & 2033
- Figure 40: Middle East & Africa Gene Engineered Subunit Vaccine Volume (K), by Application 2025 & 2033
- Figure 41: Middle East & Africa Gene Engineered Subunit Vaccine Revenue Share (%), by Application 2025 & 2033
- Figure 42: Middle East & Africa Gene Engineered Subunit Vaccine Volume Share (%), by Application 2025 & 2033
- Figure 43: Middle East & Africa Gene Engineered Subunit Vaccine Revenue (undefined), by Type 2025 & 2033
- Figure 44: Middle East & Africa Gene Engineered Subunit Vaccine Volume (K), by Type 2025 & 2033
- Figure 45: Middle East & Africa Gene Engineered Subunit Vaccine Revenue Share (%), by Type 2025 & 2033
- Figure 46: Middle East & Africa Gene Engineered Subunit Vaccine Volume Share (%), by Type 2025 & 2033
- Figure 47: Middle East & Africa Gene Engineered Subunit Vaccine Revenue (undefined), by Country 2025 & 2033
- Figure 48: Middle East & Africa Gene Engineered Subunit Vaccine Volume (K), by Country 2025 & 2033
- Figure 49: Middle East & Africa Gene Engineered Subunit Vaccine Revenue Share (%), by Country 2025 & 2033
- Figure 50: Middle East & Africa Gene Engineered Subunit Vaccine Volume Share (%), by Country 2025 & 2033
- Figure 51: Asia Pacific Gene Engineered Subunit Vaccine Revenue (undefined), by Application 2025 & 2033
- Figure 52: Asia Pacific Gene Engineered Subunit Vaccine Volume (K), by Application 2025 & 2033
- Figure 53: Asia Pacific Gene Engineered Subunit Vaccine Revenue Share (%), by Application 2025 & 2033
- Figure 54: Asia Pacific Gene Engineered Subunit Vaccine Volume Share (%), by Application 2025 & 2033
- Figure 55: Asia Pacific Gene Engineered Subunit Vaccine Revenue (undefined), by Type 2025 & 2033
- Figure 56: Asia Pacific Gene Engineered Subunit Vaccine Volume (K), by Type 2025 & 2033
- Figure 57: Asia Pacific Gene Engineered Subunit Vaccine Revenue Share (%), by Type 2025 & 2033
- Figure 58: Asia Pacific Gene Engineered Subunit Vaccine Volume Share (%), by Type 2025 & 2033
- Figure 59: Asia Pacific Gene Engineered Subunit Vaccine Revenue (undefined), by Country 2025 & 2033
- Figure 60: Asia Pacific Gene Engineered Subunit Vaccine Volume (K), by Country 2025 & 2033
- Figure 61: Asia Pacific Gene Engineered Subunit Vaccine Revenue Share (%), by Country 2025 & 2033
- Figure 62: Asia Pacific Gene Engineered Subunit Vaccine Volume Share (%), by Country 2025 & 2033
List of Tables
- Table 1: Global Gene Engineered Subunit Vaccine Revenue undefined Forecast, by Application 2020 & 2033
- Table 2: Global Gene Engineered Subunit Vaccine Volume K Forecast, by Application 2020 & 2033
- Table 3: Global Gene Engineered Subunit Vaccine Revenue undefined Forecast, by Type 2020 & 2033
- Table 4: Global Gene Engineered Subunit Vaccine Volume K Forecast, by Type 2020 & 2033
- Table 5: Global Gene Engineered Subunit Vaccine Revenue undefined Forecast, by Region 2020 & 2033
- Table 6: Global Gene Engineered Subunit Vaccine Volume K Forecast, by Region 2020 & 2033
- Table 7: Global Gene Engineered Subunit Vaccine Revenue undefined Forecast, by Application 2020 & 2033
- Table 8: Global Gene Engineered Subunit Vaccine Volume K Forecast, by Application 2020 & 2033
- Table 9: Global Gene Engineered Subunit Vaccine Revenue undefined Forecast, by Type 2020 & 2033
- Table 10: Global Gene Engineered Subunit Vaccine Volume K Forecast, by Type 2020 & 2033
- Table 11: Global Gene Engineered Subunit Vaccine Revenue undefined Forecast, by Country 2020 & 2033
- Table 12: Global Gene Engineered Subunit Vaccine Volume K Forecast, by Country 2020 & 2033
- Table 13: United States Gene Engineered Subunit Vaccine Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 14: United States Gene Engineered Subunit Vaccine Volume (K) Forecast, by Application 2020 & 2033
- Table 15: Canada Gene Engineered Subunit Vaccine Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 16: Canada Gene Engineered Subunit Vaccine Volume (K) Forecast, by Application 2020 & 2033
- Table 17: Mexico Gene Engineered Subunit Vaccine Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 18: Mexico Gene Engineered Subunit Vaccine Volume (K) Forecast, by Application 2020 & 2033
- Table 19: Global Gene Engineered Subunit Vaccine Revenue undefined Forecast, by Application 2020 & 2033
- Table 20: Global Gene Engineered Subunit Vaccine Volume K Forecast, by Application 2020 & 2033
- Table 21: Global Gene Engineered Subunit Vaccine Revenue undefined Forecast, by Type 2020 & 2033
- Table 22: Global Gene Engineered Subunit Vaccine Volume K Forecast, by Type 2020 & 2033
- Table 23: Global Gene Engineered Subunit Vaccine Revenue undefined Forecast, by Country 2020 & 2033
- Table 24: Global Gene Engineered Subunit Vaccine Volume K Forecast, by Country 2020 & 2033
- Table 25: Brazil Gene Engineered Subunit Vaccine Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 26: Brazil Gene Engineered Subunit Vaccine Volume (K) Forecast, by Application 2020 & 2033
- Table 27: Argentina Gene Engineered Subunit Vaccine Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 28: Argentina Gene Engineered Subunit Vaccine Volume (K) Forecast, by Application 2020 & 2033
- Table 29: Rest of South America Gene Engineered Subunit Vaccine Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 30: Rest of South America Gene Engineered Subunit Vaccine Volume (K) Forecast, by Application 2020 & 2033
- Table 31: Global Gene Engineered Subunit Vaccine Revenue undefined Forecast, by Application 2020 & 2033
- Table 32: Global Gene Engineered Subunit Vaccine Volume K Forecast, by Application 2020 & 2033
- Table 33: Global Gene Engineered Subunit Vaccine Revenue undefined Forecast, by Type 2020 & 2033
- Table 34: Global Gene Engineered Subunit Vaccine Volume K Forecast, by Type 2020 & 2033
- Table 35: Global Gene Engineered Subunit Vaccine Revenue undefined Forecast, by Country 2020 & 2033
- Table 36: Global Gene Engineered Subunit Vaccine Volume K Forecast, by Country 2020 & 2033
- Table 37: United Kingdom Gene Engineered Subunit Vaccine Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 38: United Kingdom Gene Engineered Subunit Vaccine Volume (K) Forecast, by Application 2020 & 2033
- Table 39: Germany Gene Engineered Subunit Vaccine Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 40: Germany Gene Engineered Subunit Vaccine Volume (K) Forecast, by Application 2020 & 2033
- Table 41: France Gene Engineered Subunit Vaccine Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 42: France Gene Engineered Subunit Vaccine Volume (K) Forecast, by Application 2020 & 2033
- Table 43: Italy Gene Engineered Subunit Vaccine Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 44: Italy Gene Engineered Subunit Vaccine Volume (K) Forecast, by Application 2020 & 2033
- Table 45: Spain Gene Engineered Subunit Vaccine Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 46: Spain Gene Engineered Subunit Vaccine Volume (K) Forecast, by Application 2020 & 2033
- Table 47: Russia Gene Engineered Subunit Vaccine Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 48: Russia Gene Engineered Subunit Vaccine Volume (K) Forecast, by Application 2020 & 2033
- Table 49: Benelux Gene Engineered Subunit Vaccine Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 50: Benelux Gene Engineered Subunit Vaccine Volume (K) Forecast, by Application 2020 & 2033
- Table 51: Nordics Gene Engineered Subunit Vaccine Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 52: Nordics Gene Engineered Subunit Vaccine Volume (K) Forecast, by Application 2020 & 2033
- Table 53: Rest of Europe Gene Engineered Subunit Vaccine Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 54: Rest of Europe Gene Engineered Subunit Vaccine Volume (K) Forecast, by Application 2020 & 2033
- Table 55: Global Gene Engineered Subunit Vaccine Revenue undefined Forecast, by Application 2020 & 2033
- Table 56: Global Gene Engineered Subunit Vaccine Volume K Forecast, by Application 2020 & 2033
- Table 57: Global Gene Engineered Subunit Vaccine Revenue undefined Forecast, by Type 2020 & 2033
- Table 58: Global Gene Engineered Subunit Vaccine Volume K Forecast, by Type 2020 & 2033
- Table 59: Global Gene Engineered Subunit Vaccine Revenue undefined Forecast, by Country 2020 & 2033
- Table 60: Global Gene Engineered Subunit Vaccine Volume K Forecast, by Country 2020 & 2033
- Table 61: Turkey Gene Engineered Subunit Vaccine Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 62: Turkey Gene Engineered Subunit Vaccine Volume (K) Forecast, by Application 2020 & 2033
- Table 63: Israel Gene Engineered Subunit Vaccine Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 64: Israel Gene Engineered Subunit Vaccine Volume (K) Forecast, by Application 2020 & 2033
- Table 65: GCC Gene Engineered Subunit Vaccine Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 66: GCC Gene Engineered Subunit Vaccine Volume (K) Forecast, by Application 2020 & 2033
- Table 67: North Africa Gene Engineered Subunit Vaccine Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 68: North Africa Gene Engineered Subunit Vaccine Volume (K) Forecast, by Application 2020 & 2033
- Table 69: South Africa Gene Engineered Subunit Vaccine Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 70: South Africa Gene Engineered Subunit Vaccine Volume (K) Forecast, by Application 2020 & 2033
- Table 71: Rest of Middle East & Africa Gene Engineered Subunit Vaccine Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 72: Rest of Middle East & Africa Gene Engineered Subunit Vaccine Volume (K) Forecast, by Application 2020 & 2033
- Table 73: Global Gene Engineered Subunit Vaccine Revenue undefined Forecast, by Application 2020 & 2033
- Table 74: Global Gene Engineered Subunit Vaccine Volume K Forecast, by Application 2020 & 2033
- Table 75: Global Gene Engineered Subunit Vaccine Revenue undefined Forecast, by Type 2020 & 2033
- Table 76: Global Gene Engineered Subunit Vaccine Volume K Forecast, by Type 2020 & 2033
- Table 77: Global Gene Engineered Subunit Vaccine Revenue undefined Forecast, by Country 2020 & 2033
- Table 78: Global Gene Engineered Subunit Vaccine Volume K Forecast, by Country 2020 & 2033
- Table 79: China Gene Engineered Subunit Vaccine Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 80: China Gene Engineered Subunit Vaccine Volume (K) Forecast, by Application 2020 & 2033
- Table 81: India Gene Engineered Subunit Vaccine Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 82: India Gene Engineered Subunit Vaccine Volume (K) Forecast, by Application 2020 & 2033
- Table 83: Japan Gene Engineered Subunit Vaccine Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 84: Japan Gene Engineered Subunit Vaccine Volume (K) Forecast, by Application 2020 & 2033
- Table 85: South Korea Gene Engineered Subunit Vaccine Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 86: South Korea Gene Engineered Subunit Vaccine Volume (K) Forecast, by Application 2020 & 2033
- Table 87: ASEAN Gene Engineered Subunit Vaccine Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 88: ASEAN Gene Engineered Subunit Vaccine Volume (K) Forecast, by Application 2020 & 2033
- Table 89: Oceania Gene Engineered Subunit Vaccine Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 90: Oceania Gene Engineered Subunit Vaccine Volume (K) Forecast, by Application 2020 & 2033
- Table 91: Rest of Asia Pacific Gene Engineered Subunit Vaccine Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 92: Rest of Asia Pacific Gene Engineered Subunit Vaccine Volume (K) Forecast, by Application 2020 & 2033
Frequently Asked Questions
1. What is the projected Compound Annual Growth Rate (CAGR) of the Gene Engineered Subunit Vaccine?
The projected CAGR is approximately 11.4%.
2. Which companies are prominent players in the Gene Engineered Subunit Vaccine?
Key companies in the market include Merck, GSK, Tiantan, Pulike, Greffex, CureVac, Yebio, Sanofi Pasteur SA, Virbac, Pfizer Inc., Walvax Biotechnology, Kontec, Novavax, Sanofi Pasteur, Bharat Biotech, GlaxoSmithKline, Pfizer, Moderna.
3. What are the main segments of the Gene Engineered Subunit Vaccine?
The market segments include Application, Type.
4. Can you provide details about the market size?
The market size is estimated to be USD XXX N/A as of 2022.
5. What are some drivers contributing to market growth?
N/A
6. What are the notable trends driving market growth?
N/A
7. Are there any restraints impacting market growth?
N/A
8. Can you provide examples of recent developments in the market?
N/A
9. What pricing options are available for accessing the report?
Pricing options include single-user, multi-user, and enterprise licenses priced at USD 3950.00, USD 5925.00, and USD 7900.00 respectively.
10. Is the market size provided in terms of value or volume?
The market size is provided in terms of value, measured in N/A and volume, measured in K.
11. Are there any specific market keywords associated with the report?
Yes, the market keyword associated with the report is "Gene Engineered Subunit Vaccine," which aids in identifying and referencing the specific market segment covered.
12. How do I determine which pricing option suits my needs best?
The pricing options vary based on user requirements and access needs. Individual users may opt for single-user licenses, while businesses requiring broader access may choose multi-user or enterprise licenses for cost-effective access to the report.
13. Are there any additional resources or data provided in the Gene Engineered Subunit Vaccine report?
While the report offers comprehensive insights, it's advisable to review the specific contents or supplementary materials provided to ascertain if additional resources or data are available.
14. How can I stay updated on further developments or reports in the Gene Engineered Subunit Vaccine?
To stay informed about further developments, trends, and reports in the Gene Engineered Subunit Vaccine, consider subscribing to industry newsletters, following relevant companies and organizations, or regularly checking reputable industry news sources and publications.
Methodology
Step 1 - Identification of Relevant Samples Size from Population Database



Step 2 - Approaches for Defining Global Market Size (Value, Volume* & Price*)

Note*: In applicable scenarios
Step 3 - Data Sources
Primary Research
- Web Analytics
- Survey Reports
- Research Institute
- Latest Research Reports
- Opinion Leaders
Secondary Research
- Annual Reports
- White Paper
- Latest Press Release
- Industry Association
- Paid Database
- Investor Presentations

Step 4 - Data Triangulation
Involves using different sources of information in order to increase the validity of a study
These sources are likely to be stakeholders in a program - participants, other researchers, program staff, other community members, and so on.
Then we put all data in single framework & apply various statistical tools to find out the dynamic on the market.
During the analysis stage, feedback from the stakeholder groups would be compared to determine areas of agreement as well as areas of divergence


