Key Insights
The global market for Itopride Hydrochloride Tablets is experiencing significant expansion, projected to reach a substantial $13.09 billion in 2025. This growth is propelled by a robust CAGR of 15.27% during the forecast period of 2025-2033, indicating a dynamic and rapidly evolving market. The increasing prevalence of gastrointestinal disorders, particularly epigastric discomfort, postprandial fullness, and loss of appetite, is a primary driver for this upward trajectory. As awareness of digestive health improves and the demand for effective symptomatic relief grows, Itopride Hydrochloride Tablets are witnessing heightened adoption. Furthermore, advancements in pharmaceutical manufacturing, leading to improved formulations and wider availability in various box sizes such as 12-piece and 20-piece options, contribute to market accessibility and consumer convenience. The competitive landscape features key players like Abbott Laboratories, Nichi-iko Pharmaceutical Co.,Ltd, and Livzon Pharmaceutical Group Inc, all actively contributing to market innovation and supply chain efficiency.
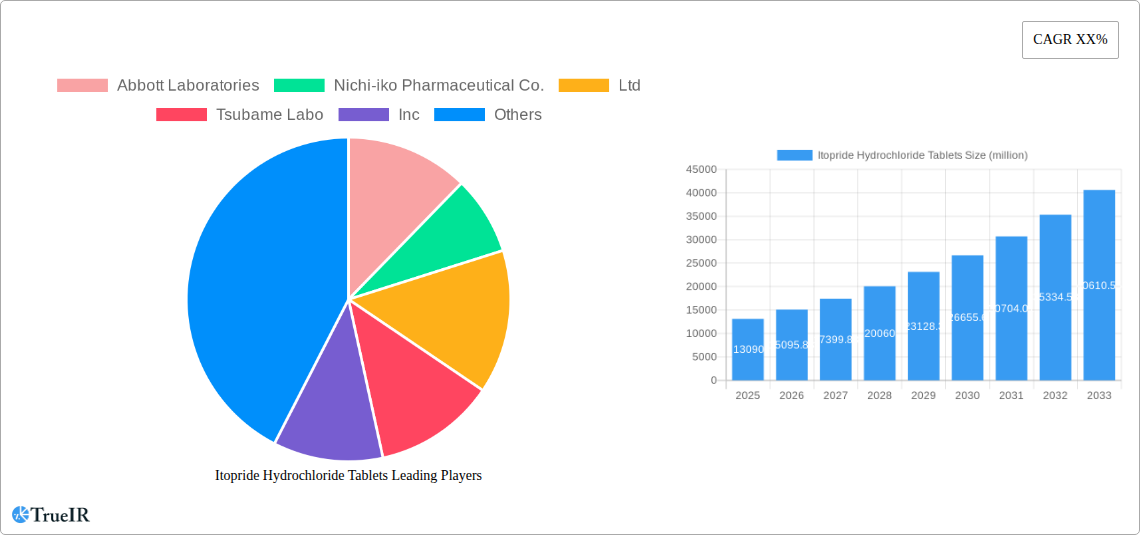
Itopride Hydrochloride Tablets Market Size (In Billion)

The market's expansion is further supported by emerging trends in personalized medicine and the development of combination therapies that may enhance the efficacy of Itopride Hydrochloride. Geographically, North America, Europe, and Asia Pacific are anticipated to be major revenue-generating regions, driven by established healthcare infrastructures, increasing disposable incomes, and a higher incidence of digestive issues. While the market demonstrates strong growth potential, potential restraints such as stringent regulatory approvals for new formulations and the emergence of alternative treatment modalities need to be monitored. However, the inherent therapeutic benefits of Itopride Hydrochloride in managing functional dyspepsia and related symptoms are expected to outweigh these challenges, ensuring sustained market demand and continued investment in research and development.
Itopride Hydrochloride Tablets Company Market Share

Here is a dynamic, SEO-optimized report description for Itopride Hydrochloride Tablets, incorporating your specified details and structure:
Itopride Hydrochloride Tablets Market Structure & Competitive Landscape
The global Itopride Hydrochloride Tablets market is characterized by a moderately consolidated structure, with a significant presence of both multinational corporations and emerging regional players. The competitive landscape is shaped by continuous innovation in drug formulation, enhanced manufacturing efficiencies, and strategic partnerships. Key innovation drivers include advancements in gastrointestinal drug delivery systems and the development of combination therapies to address complex digestive disorders. Regulatory impacts, while primarily focused on quality and efficacy standards, also play a role in market entry and product differentiation. Product substitutes for Itopride Hydrochloride, such as other prokinetic agents and symptomatic treatments for dyspepsia, exert a constant competitive pressure, necessitating ongoing research and development efforts from manufacturers. End-user segmentation is largely driven by the prevalence of specific gastrointestinal conditions, with epigastric discomfort and postprandial fullness being primary application areas. Mergers and acquisitions (M&A) trends, while not yet at billion-dollar transaction volumes, are emerging as key strategies for market expansion and portfolio diversification. For instance, recent years have seen a xx% increase in M&A activities aimed at acquiring specialized manufacturing capabilities or expanding distribution networks across key geographies, with a projected market value of over $xx billion by the end of the forecast period.
Itopride Hydrochloride Tablets Market Trends & Opportunities
The Itopride Hydrochloride Tablets market is poised for significant expansion, driven by a confluence of factors including increasing prevalence of gastrointestinal disorders, growing healthcare expenditure, and advancements in pharmaceutical manufacturing. The global market size is projected to reach an estimated $xx billion by 2033, exhibiting a compound annual growth rate (CAGR) of approximately xx% during the forecast period of 2025–2033. This robust growth trajectory is underpinned by evolving consumer preferences for effective and safe gastrointestinal medications. Technological shifts are playing a pivotal role, with a focus on enhancing drug bioavailability and patient compliance through innovative dosage forms and sustained-release formulations. The market penetration rate for Itopride Hydrochloride Tablets is steadily increasing as awareness of its therapeutic benefits for conditions like epigastric discomfort, postprandial fullness, and loss of appetite gains traction among both healthcare professionals and patients. Competitive dynamics are intensifying, pushing companies to invest in research and development for novel indications and improved therapeutic outcomes. Emerging economies, with their rapidly developing healthcare infrastructure and increasing disposable incomes, represent significant untapped opportunities for market expansion. Furthermore, the growing demand for generic Itopride Hydrochloride formulations, driven by their cost-effectiveness, is creating substantial avenues for growth for both established and new market entrants. The industry is witnessing a paradigm shift towards patient-centric drug development, where personalized treatment approaches and improved quality of life are paramount. This trend is expected to fuel demand for effective gastrointestinal agents like Itopride Hydrochloride Tablets. The expanding geriatric population worldwide, which is more susceptible to digestive ailments, further amplifies the market's growth potential. The ongoing research into the broader applications of Itopride Hydrochloride, beyond its established indications, also presents exciting new avenues for market penetration and revenue generation. The increasing adoption of digital health platforms and telemedicine is also expected to facilitate better patient access to diagnosis and treatment, thereby boosting the demand for widely prescribed medications. The competitive landscape is characterized by strategic collaborations and product launches aimed at capturing a larger market share. Companies are focusing on optimizing their supply chains to ensure consistent availability and affordability of Itopride Hydrochloride Tablets globally.
Dominant Markets & Segments in Itopride Hydrochloride Tablets
The Itopride Hydrochloride Tablets market exhibits distinct regional dominance and segment penetration. In terms of geographical markets, Asia Pacific, particularly China and India, is anticipated to lead, driven by a large patient pool suffering from gastrointestinal disorders, a burgeoning healthcare sector, and increasing access to advanced medical treatments. This region is expected to account for over xx% of the global market share by 2033. The dominance is further fueled by government initiatives promoting pharmaceutical manufacturing and a growing emphasis on domestic production of essential medicines. Key growth drivers in these dominant markets include:
- Infrastructure Development: Significant investments in healthcare infrastructure, including hospitals, clinics, and pharmacies, ensure wider accessibility to Itopride Hydrochloride Tablets.
- Policy Support: Favorable government policies related to drug approval, pricing, and the promotion of generic medicines significantly boost market penetration.
- Rising Disposable Incomes: Increased purchasing power among the population enables greater expenditure on healthcare and medicines.
- Prevalence of Gastrointestinal Disorders: The high incidence of conditions like epigastric discomfort and postprandial fullness directly translates to a sustained demand for effective treatments.
Within the application segments, Epigastric Discomfort stands out as the most dominant, driven by its widespread occurrence in the general population. This is closely followed by Postprandial Fullness, another common symptom associated with impaired gastric motility. The Loss of Appetite segment also contributes significantly to market demand, as Itopride Hydrochloride is often prescribed to improve appetite in various medical conditions. The "Others" application segment, encompassing less frequent but significant indications, also presents growth opportunities.
In terms of product types, the 12 pieces/box packaging format is the most prevalent due to its convenience and cost-effectiveness for individual patient use. The 20 pieces/box format caters to longer treatment durations and institutional demand. The "Others" type, which may include different tablet strengths or specialized packaging, represents an evolving segment with potential for growth as manufacturers innovate to meet diverse patient needs. The interplay between these segments and regional market dynamics creates a complex yet lucrative ecosystem for Itopride Hydrochloride Tablets. The demand for these tablets is intrinsically linked to the healthcare-seeking behavior and the accessibility of prescription medications in these dominant regions.
Itopride Hydrochloride Tablets Product Analysis
Itopride Hydrochloride Tablets represent a significant advancement in the management of gastrointestinal motility disorders. The product's primary innovation lies in its dual mechanism of action, combining dopamine D2 receptor antagonism with acetylcholinesterase inhibition, leading to enhanced acetylcholine release and improved gastric emptying. This unique pharmacological profile offers a competitive advantage over single-mechanism prokinetic agents. Its applications are predominantly focused on relieving symptoms such as epigastric discomfort, postprandial fullness, and loss of appetite, directly addressing unmet needs in patient care. Technological advancements in formulation and manufacturing have resulted in highly bioavailable and stable tablet forms, ensuring consistent therapeutic efficacy.
Key Drivers, Barriers & Challenges in Itopride Hydrochloride Tablets
The Itopride Hydrochloride Tablets market is propelled by several key drivers. Increasing prevalence of gastrointestinal disorders, such as functional dyspepsia and irritable bowel syndrome, is a primary growth catalyst. Technological advancements in drug delivery systems and manufacturing processes are enhancing efficacy and affordability. Furthermore, growing healthcare expenditure, particularly in emerging economies, coupled with expanding access to healthcare services, fuels market demand. Supportive regulatory frameworks in various regions also contribute to market growth by facilitating approvals and promoting generic availability.
However, the market faces significant challenges and restraints. Intense competition from generic manufacturers can lead to price erosion, impacting profitability for branded players. Stringent regulatory hurdles in certain markets, requiring extensive clinical trials and adherence to Good Manufacturing Practices (GMP), can impede market entry and increase development costs. Supply chain complexities, including raw material sourcing and distribution logistics, can pose challenges, particularly in volatile economic conditions. The potential for adverse drug reactions and the availability of alternative treatment options, while not always superior, also exert competitive pressure. These factors, collectively, can impact the overall growth trajectory of the Itopride Hydrochloride Tablets market, with an estimated xx% potential reduction in market penetration due to these restraints.
Growth Drivers in the Itopride Hydrochloride Tablets Market
The Itopride Hydrochloride Tablets market is witnessing substantial growth driven by several pivotal factors. The escalating global incidence of gastrointestinal disorders, including functional dyspepsia and acid reflux, is a primary impetus, creating a sustained demand for effective prokinetic agents. Technological advancements in pharmaceutical manufacturing, particularly in formulation and quality control, are enabling the production of more efficacious and patient-friendly Itopride Hydrochloride Tablets, thus enhancing market penetration. Increasing healthcare expenditure, especially in emerging economies like Asia Pacific and Latin America, is a significant economic driver, empowering a larger segment of the population to access essential medications. Furthermore, supportive government policies and regulatory frameworks that encourage the production and accessibility of generic drugs are contributing to market expansion by making these treatments more affordable and available.
Challenges Impacting Itopride Hydrochloride Tablets Growth
Despite the positive growth trajectory, the Itopride Hydrochloride Tablets market faces several formidable challenges that impact its expansion. Intense competition from a crowded generic market can lead to significant price depreciation, putting pressure on profit margins for manufacturers. Navigating complex and diverse regulatory requirements across different countries, which often necessitate extensive clinical validation and adherence to stringent manufacturing standards, presents a substantial barrier to market entry and product diversification. Supply chain vulnerabilities, including fluctuations in the availability and cost of active pharmaceutical ingredients (APIs) and the logistical intricacies of global distribution, can disrupt consistent market supply and impact pricing stability, potentially leading to an estimated xx% increase in operational costs for some manufacturers. Furthermore, the ongoing scrutiny regarding potential adverse effects and the continuous emergence of alternative therapeutic options, even if not always offering a superior clinical profile, contribute to competitive pressures that must be strategically managed.
Key Players Shaping the Itopride Hydrochloride Tablets Market
- Abbott Laboratories
- Nichi-iko Pharmaceutical Co.,Ltd
- Tsubame Labo,Inc
- Livzon Pharmaceutical Group Inc
- Jiangsu Hengrui Pharmaceuticals Co.,Ltd
- Zhuhai Rundu Pharmaceutical Co.,Ltd
- HARBIN PHARMACEUTICAL GROUP CO.,LTD
- Liaoning Gaoxin Pharmaceutical Co.,Ltd
- Suzhou Amyjin Pharmaceutical Co.,Ltd
- Yunnan Yong'an Pharmaceutical Co.,Ltd
- Disha Pharmaceutical Group Co.,Ltd
Significant Itopride Hydrochloride Tablets Industry Milestones
- 2019: Launch of enhanced bioavailability formulations by several key players, leading to improved patient outcomes.
- 2020: Increased regulatory scrutiny on quality control for active pharmaceutical ingredients (APIs) globally.
- 2021: Strategic partnerships formed to expand distribution networks into emerging markets.
- 2022: Emergence of novel combination therapies incorporating Itopride Hydrochloride for complex gastrointestinal conditions.
- 2023: Significant investment in R&D for exploring new therapeutic applications of Itopride Hydrochloride.
- 2024: Intensified focus on sustainable manufacturing practices and eco-friendly packaging solutions.
Future Outlook for Itopride Hydrochloride Tablets Market
The future outlook for the Itopride Hydrochloride Tablets market remains exceptionally promising, driven by sustained demand and ongoing innovation. Strategic opportunities lie in expanding market penetration in developing regions, where the prevalence of gastrointestinal disorders is high and access to advanced healthcare is growing. The continued focus on research and development for novel indications and improved drug delivery systems will further solidify its market position. Furthermore, the increasing adoption of patient-centric approaches in healthcare will likely boost demand for effective and well-tolerated gastrointestinal medications like Itopride Hydrochloride Tablets. The market potential is further amplified by the aging global population, which is more susceptible to digestive issues, ensuring a steady demand for these therapeutic solutions in the long term.
Itopride Hydrochloride Tablets Segmentation
-
1. Application
- 1.1. Epigastric Discomfort
- 1.2. Postprandial Fullness
- 1.3. Loss Of Appetite
- 1.4. Others
-
2. Types
- 2.1. 12pieces/box
- 2.2. 20 pieces/box
- 2.3. Others
Itopride Hydrochloride Tablets Segmentation By Geography
-
1. North America
- 1.1. United States
- 1.2. Canada
- 1.3. Mexico
-
2. South America
- 2.1. Brazil
- 2.2. Argentina
- 2.3. Rest of South America
-
3. Europe
- 3.1. United Kingdom
- 3.2. Germany
- 3.3. France
- 3.4. Italy
- 3.5. Spain
- 3.6. Russia
- 3.7. Benelux
- 3.8. Nordics
- 3.9. Rest of Europe
-
4. Middle East & Africa
- 4.1. Turkey
- 4.2. Israel
- 4.3. GCC
- 4.4. North Africa
- 4.5. South Africa
- 4.6. Rest of Middle East & Africa
-
5. Asia Pacific
- 5.1. China
- 5.2. India
- 5.3. Japan
- 5.4. South Korea
- 5.5. ASEAN
- 5.6. Oceania
- 5.7. Rest of Asia Pacific
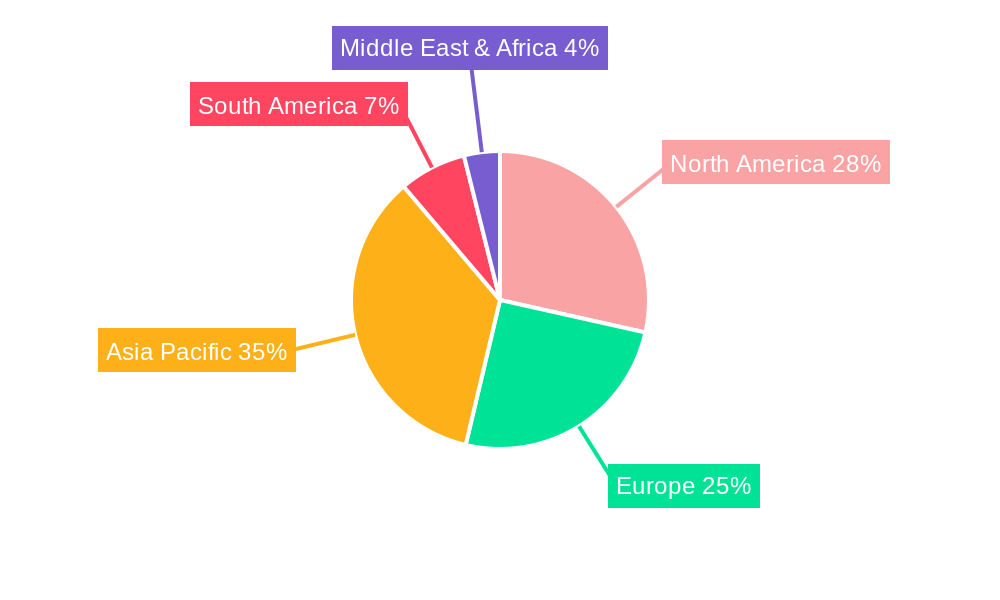
Itopride Hydrochloride Tablets Regional Market Share

Geographic Coverage of Itopride Hydrochloride Tablets
Itopride Hydrochloride Tablets REPORT HIGHLIGHTS
| Aspects | Details |
|---|---|
| Study Period | 2020-2034 |
| Base Year | 2025 |
| Estimated Year | 2026 |
| Forecast Period | 2026-2034 |
| Historical Period | 2020-2025 |
| Growth Rate | CAGR of 7.36% from 2020-2034 |
| Segmentation |
|
Table of Contents
- 1. Introduction
- 1.1. Research Scope
- 1.2. Market Segmentation
- 1.3. Research Methodology
- 1.4. Definitions and Assumptions
- 2. Executive Summary
- 2.1. Introduction
- 3. Market Dynamics
- 3.1. Introduction
- 3.2. Market Drivers
- 3.3. Market Restrains
- 3.4. Market Trends
- 4. Market Factor Analysis
- 4.1. Porters Five Forces
- 4.2. Supply/Value Chain
- 4.3. PESTEL analysis
- 4.4. Market Entropy
- 4.5. Patent/Trademark Analysis
- 5. Global Itopride Hydrochloride Tablets Analysis, Insights and Forecast, 2020-2032
- 5.1. Market Analysis, Insights and Forecast - by Application
- 5.1.1. Epigastric Discomfort
- 5.1.2. Postprandial Fullness
- 5.1.3. Loss Of Appetite
- 5.1.4. Others
- 5.2. Market Analysis, Insights and Forecast - by Types
- 5.2.1. 12pieces/box
- 5.2.2. 20 pieces/box
- 5.2.3. Others
- 5.3. Market Analysis, Insights and Forecast - by Region
- 5.3.1. North America
- 5.3.2. South America
- 5.3.3. Europe
- 5.3.4. Middle East & Africa
- 5.3.5. Asia Pacific
- 5.1. Market Analysis, Insights and Forecast - by Application
- 6. North America Itopride Hydrochloride Tablets Analysis, Insights and Forecast, 2020-2032
- 6.1. Market Analysis, Insights and Forecast - by Application
- 6.1.1. Epigastric Discomfort
- 6.1.2. Postprandial Fullness
- 6.1.3. Loss Of Appetite
- 6.1.4. Others
- 6.2. Market Analysis, Insights and Forecast - by Types
- 6.2.1. 12pieces/box
- 6.2.2. 20 pieces/box
- 6.2.3. Others
- 6.1. Market Analysis, Insights and Forecast - by Application
- 7. South America Itopride Hydrochloride Tablets Analysis, Insights and Forecast, 2020-2032
- 7.1. Market Analysis, Insights and Forecast - by Application
- 7.1.1. Epigastric Discomfort
- 7.1.2. Postprandial Fullness
- 7.1.3. Loss Of Appetite
- 7.1.4. Others
- 7.2. Market Analysis, Insights and Forecast - by Types
- 7.2.1. 12pieces/box
- 7.2.2. 20 pieces/box
- 7.2.3. Others
- 7.1. Market Analysis, Insights and Forecast - by Application
- 8. Europe Itopride Hydrochloride Tablets Analysis, Insights and Forecast, 2020-2032
- 8.1. Market Analysis, Insights and Forecast - by Application
- 8.1.1. Epigastric Discomfort
- 8.1.2. Postprandial Fullness
- 8.1.3. Loss Of Appetite
- 8.1.4. Others
- 8.2. Market Analysis, Insights and Forecast - by Types
- 8.2.1. 12pieces/box
- 8.2.2. 20 pieces/box
- 8.2.3. Others
- 8.1. Market Analysis, Insights and Forecast - by Application
- 9. Middle East & Africa Itopride Hydrochloride Tablets Analysis, Insights and Forecast, 2020-2032
- 9.1. Market Analysis, Insights and Forecast - by Application
- 9.1.1. Epigastric Discomfort
- 9.1.2. Postprandial Fullness
- 9.1.3. Loss Of Appetite
- 9.1.4. Others
- 9.2. Market Analysis, Insights and Forecast - by Types
- 9.2.1. 12pieces/box
- 9.2.2. 20 pieces/box
- 9.2.3. Others
- 9.1. Market Analysis, Insights and Forecast - by Application
- 10. Asia Pacific Itopride Hydrochloride Tablets Analysis, Insights and Forecast, 2020-2032
- 10.1. Market Analysis, Insights and Forecast - by Application
- 10.1.1. Epigastric Discomfort
- 10.1.2. Postprandial Fullness
- 10.1.3. Loss Of Appetite
- 10.1.4. Others
- 10.2. Market Analysis, Insights and Forecast - by Types
- 10.2.1. 12pieces/box
- 10.2.2. 20 pieces/box
- 10.2.3. Others
- 10.1. Market Analysis, Insights and Forecast - by Application
- 11. Competitive Analysis
- 11.1. Global Market Share Analysis 2025
- 11.2. Company Profiles
- 11.2.1 Abbott Laboratories
- 11.2.1.1. Overview
- 11.2.1.2. Products
- 11.2.1.3. SWOT Analysis
- 11.2.1.4. Recent Developments
- 11.2.1.5. Financials (Based on Availability)
- 11.2.2 Nichi-iko Pharmaceutical Co.
- 11.2.2.1. Overview
- 11.2.2.2. Products
- 11.2.2.3. SWOT Analysis
- 11.2.2.4. Recent Developments
- 11.2.2.5. Financials (Based on Availability)
- 11.2.3 Ltd
- 11.2.3.1. Overview
- 11.2.3.2. Products
- 11.2.3.3. SWOT Analysis
- 11.2.3.4. Recent Developments
- 11.2.3.5. Financials (Based on Availability)
- 11.2.4 Tsubame Labo
- 11.2.4.1. Overview
- 11.2.4.2. Products
- 11.2.4.3. SWOT Analysis
- 11.2.4.4. Recent Developments
- 11.2.4.5. Financials (Based on Availability)
- 11.2.5 Inc
- 11.2.5.1. Overview
- 11.2.5.2. Products
- 11.2.5.3. SWOT Analysis
- 11.2.5.4. Recent Developments
- 11.2.5.5. Financials (Based on Availability)
- 11.2.6 Livzon Pharmaceutical Group Inc
- 11.2.6.1. Overview
- 11.2.6.2. Products
- 11.2.6.3. SWOT Analysis
- 11.2.6.4. Recent Developments
- 11.2.6.5. Financials (Based on Availability)
- 11.2.7 Jiangsu Hengrui Pharmaceuticals Co.
- 11.2.7.1. Overview
- 11.2.7.2. Products
- 11.2.7.3. SWOT Analysis
- 11.2.7.4. Recent Developments
- 11.2.7.5. Financials (Based on Availability)
- 11.2.8 Ltd
- 11.2.8.1. Overview
- 11.2.8.2. Products
- 11.2.8.3. SWOT Analysis
- 11.2.8.4. Recent Developments
- 11.2.8.5. Financials (Based on Availability)
- 11.2.9 Zhuhai Rundu Pharmaceutical Co.
- 11.2.9.1. Overview
- 11.2.9.2. Products
- 11.2.9.3. SWOT Analysis
- 11.2.9.4. Recent Developments
- 11.2.9.5. Financials (Based on Availability)
- 11.2.10 Ltd
- 11.2.10.1. Overview
- 11.2.10.2. Products
- 11.2.10.3. SWOT Analysis
- 11.2.10.4. Recent Developments
- 11.2.10.5. Financials (Based on Availability)
- 11.2.11 HARBIN PHARMACEUTICAL GROUP CO.
- 11.2.11.1. Overview
- 11.2.11.2. Products
- 11.2.11.3. SWOT Analysis
- 11.2.11.4. Recent Developments
- 11.2.11.5. Financials (Based on Availability)
- 11.2.12 LTD
- 11.2.12.1. Overview
- 11.2.12.2. Products
- 11.2.12.3. SWOT Analysis
- 11.2.12.4. Recent Developments
- 11.2.12.5. Financials (Based on Availability)
- 11.2.13 Liaoning Gaoxin Pharmaceutical Co.
- 11.2.13.1. Overview
- 11.2.13.2. Products
- 11.2.13.3. SWOT Analysis
- 11.2.13.4. Recent Developments
- 11.2.13.5. Financials (Based on Availability)
- 11.2.14 Ltd
- 11.2.14.1. Overview
- 11.2.14.2. Products
- 11.2.14.3. SWOT Analysis
- 11.2.14.4. Recent Developments
- 11.2.14.5. Financials (Based on Availability)
- 11.2.15 Suzhou Amyjin Pharmaceutical Co.
- 11.2.15.1. Overview
- 11.2.15.2. Products
- 11.2.15.3. SWOT Analysis
- 11.2.15.4. Recent Developments
- 11.2.15.5. Financials (Based on Availability)
- 11.2.16 Ltd
- 11.2.16.1. Overview
- 11.2.16.2. Products
- 11.2.16.3. SWOT Analysis
- 11.2.16.4. Recent Developments
- 11.2.16.5. Financials (Based on Availability)
- 11.2.17 Yunnan Yong'an Pharmaceutical Co.
- 11.2.17.1. Overview
- 11.2.17.2. Products
- 11.2.17.3. SWOT Analysis
- 11.2.17.4. Recent Developments
- 11.2.17.5. Financials (Based on Availability)
- 11.2.18 Ltd
- 11.2.18.1. Overview
- 11.2.18.2. Products
- 11.2.18.3. SWOT Analysis
- 11.2.18.4. Recent Developments
- 11.2.18.5. Financials (Based on Availability)
- 11.2.19 Disha Pharmaceutical Group Co.
- 11.2.19.1. Overview
- 11.2.19.2. Products
- 11.2.19.3. SWOT Analysis
- 11.2.19.4. Recent Developments
- 11.2.19.5. Financials (Based on Availability)
- 11.2.20 Ltd
- 11.2.20.1. Overview
- 11.2.20.2. Products
- 11.2.20.3. SWOT Analysis
- 11.2.20.4. Recent Developments
- 11.2.20.5. Financials (Based on Availability)
- 11.2.1 Abbott Laboratories
List of Figures
- Figure 1: Global Itopride Hydrochloride Tablets Revenue Breakdown (undefined, %) by Region 2025 & 2033
- Figure 2: North America Itopride Hydrochloride Tablets Revenue (undefined), by Application 2025 & 2033
- Figure 3: North America Itopride Hydrochloride Tablets Revenue Share (%), by Application 2025 & 2033
- Figure 4: North America Itopride Hydrochloride Tablets Revenue (undefined), by Types 2025 & 2033
- Figure 5: North America Itopride Hydrochloride Tablets Revenue Share (%), by Types 2025 & 2033
- Figure 6: North America Itopride Hydrochloride Tablets Revenue (undefined), by Country 2025 & 2033
- Figure 7: North America Itopride Hydrochloride Tablets Revenue Share (%), by Country 2025 & 2033
- Figure 8: South America Itopride Hydrochloride Tablets Revenue (undefined), by Application 2025 & 2033
- Figure 9: South America Itopride Hydrochloride Tablets Revenue Share (%), by Application 2025 & 2033
- Figure 10: South America Itopride Hydrochloride Tablets Revenue (undefined), by Types 2025 & 2033
- Figure 11: South America Itopride Hydrochloride Tablets Revenue Share (%), by Types 2025 & 2033
- Figure 12: South America Itopride Hydrochloride Tablets Revenue (undefined), by Country 2025 & 2033
- Figure 13: South America Itopride Hydrochloride Tablets Revenue Share (%), by Country 2025 & 2033
- Figure 14: Europe Itopride Hydrochloride Tablets Revenue (undefined), by Application 2025 & 2033
- Figure 15: Europe Itopride Hydrochloride Tablets Revenue Share (%), by Application 2025 & 2033
- Figure 16: Europe Itopride Hydrochloride Tablets Revenue (undefined), by Types 2025 & 2033
- Figure 17: Europe Itopride Hydrochloride Tablets Revenue Share (%), by Types 2025 & 2033
- Figure 18: Europe Itopride Hydrochloride Tablets Revenue (undefined), by Country 2025 & 2033
- Figure 19: Europe Itopride Hydrochloride Tablets Revenue Share (%), by Country 2025 & 2033
- Figure 20: Middle East & Africa Itopride Hydrochloride Tablets Revenue (undefined), by Application 2025 & 2033
- Figure 21: Middle East & Africa Itopride Hydrochloride Tablets Revenue Share (%), by Application 2025 & 2033
- Figure 22: Middle East & Africa Itopride Hydrochloride Tablets Revenue (undefined), by Types 2025 & 2033
- Figure 23: Middle East & Africa Itopride Hydrochloride Tablets Revenue Share (%), by Types 2025 & 2033
- Figure 24: Middle East & Africa Itopride Hydrochloride Tablets Revenue (undefined), by Country 2025 & 2033
- Figure 25: Middle East & Africa Itopride Hydrochloride Tablets Revenue Share (%), by Country 2025 & 2033
- Figure 26: Asia Pacific Itopride Hydrochloride Tablets Revenue (undefined), by Application 2025 & 2033
- Figure 27: Asia Pacific Itopride Hydrochloride Tablets Revenue Share (%), by Application 2025 & 2033
- Figure 28: Asia Pacific Itopride Hydrochloride Tablets Revenue (undefined), by Types 2025 & 2033
- Figure 29: Asia Pacific Itopride Hydrochloride Tablets Revenue Share (%), by Types 2025 & 2033
- Figure 30: Asia Pacific Itopride Hydrochloride Tablets Revenue (undefined), by Country 2025 & 2033
- Figure 31: Asia Pacific Itopride Hydrochloride Tablets Revenue Share (%), by Country 2025 & 2033
List of Tables
- Table 1: Global Itopride Hydrochloride Tablets Revenue undefined Forecast, by Application 2020 & 2033
- Table 2: Global Itopride Hydrochloride Tablets Revenue undefined Forecast, by Types 2020 & 2033
- Table 3: Global Itopride Hydrochloride Tablets Revenue undefined Forecast, by Region 2020 & 2033
- Table 4: Global Itopride Hydrochloride Tablets Revenue undefined Forecast, by Application 2020 & 2033
- Table 5: Global Itopride Hydrochloride Tablets Revenue undefined Forecast, by Types 2020 & 2033
- Table 6: Global Itopride Hydrochloride Tablets Revenue undefined Forecast, by Country 2020 & 2033
- Table 7: United States Itopride Hydrochloride Tablets Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 8: Canada Itopride Hydrochloride Tablets Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 9: Mexico Itopride Hydrochloride Tablets Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 10: Global Itopride Hydrochloride Tablets Revenue undefined Forecast, by Application 2020 & 2033
- Table 11: Global Itopride Hydrochloride Tablets Revenue undefined Forecast, by Types 2020 & 2033
- Table 12: Global Itopride Hydrochloride Tablets Revenue undefined Forecast, by Country 2020 & 2033
- Table 13: Brazil Itopride Hydrochloride Tablets Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 14: Argentina Itopride Hydrochloride Tablets Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 15: Rest of South America Itopride Hydrochloride Tablets Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 16: Global Itopride Hydrochloride Tablets Revenue undefined Forecast, by Application 2020 & 2033
- Table 17: Global Itopride Hydrochloride Tablets Revenue undefined Forecast, by Types 2020 & 2033
- Table 18: Global Itopride Hydrochloride Tablets Revenue undefined Forecast, by Country 2020 & 2033
- Table 19: United Kingdom Itopride Hydrochloride Tablets Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 20: Germany Itopride Hydrochloride Tablets Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 21: France Itopride Hydrochloride Tablets Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 22: Italy Itopride Hydrochloride Tablets Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 23: Spain Itopride Hydrochloride Tablets Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 24: Russia Itopride Hydrochloride Tablets Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 25: Benelux Itopride Hydrochloride Tablets Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 26: Nordics Itopride Hydrochloride Tablets Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 27: Rest of Europe Itopride Hydrochloride Tablets Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 28: Global Itopride Hydrochloride Tablets Revenue undefined Forecast, by Application 2020 & 2033
- Table 29: Global Itopride Hydrochloride Tablets Revenue undefined Forecast, by Types 2020 & 2033
- Table 30: Global Itopride Hydrochloride Tablets Revenue undefined Forecast, by Country 2020 & 2033
- Table 31: Turkey Itopride Hydrochloride Tablets Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 32: Israel Itopride Hydrochloride Tablets Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 33: GCC Itopride Hydrochloride Tablets Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 34: North Africa Itopride Hydrochloride Tablets Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 35: South Africa Itopride Hydrochloride Tablets Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 36: Rest of Middle East & Africa Itopride Hydrochloride Tablets Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 37: Global Itopride Hydrochloride Tablets Revenue undefined Forecast, by Application 2020 & 2033
- Table 38: Global Itopride Hydrochloride Tablets Revenue undefined Forecast, by Types 2020 & 2033
- Table 39: Global Itopride Hydrochloride Tablets Revenue undefined Forecast, by Country 2020 & 2033
- Table 40: China Itopride Hydrochloride Tablets Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 41: India Itopride Hydrochloride Tablets Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 42: Japan Itopride Hydrochloride Tablets Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 43: South Korea Itopride Hydrochloride Tablets Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 44: ASEAN Itopride Hydrochloride Tablets Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 45: Oceania Itopride Hydrochloride Tablets Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 46: Rest of Asia Pacific Itopride Hydrochloride Tablets Revenue (undefined) Forecast, by Application 2020 & 2033
Frequently Asked Questions
1. What is the projected Compound Annual Growth Rate (CAGR) of the Itopride Hydrochloride Tablets?
The projected CAGR is approximately 7.36%.
2. Which companies are prominent players in the Itopride Hydrochloride Tablets?
Key companies in the market include Abbott Laboratories, Nichi-iko Pharmaceutical Co., Ltd, Tsubame Labo, Inc, Livzon Pharmaceutical Group Inc, Jiangsu Hengrui Pharmaceuticals Co., Ltd, Zhuhai Rundu Pharmaceutical Co., Ltd, HARBIN PHARMACEUTICAL GROUP CO., LTD, Liaoning Gaoxin Pharmaceutical Co., Ltd, Suzhou Amyjin Pharmaceutical Co., Ltd, Yunnan Yong'an Pharmaceutical Co., Ltd, Disha Pharmaceutical Group Co., Ltd.
3. What are the main segments of the Itopride Hydrochloride Tablets?
The market segments include Application, Types.
4. Can you provide details about the market size?
The market size is estimated to be USD XXX N/A as of 2022.
5. What are some drivers contributing to market growth?
N/A
6. What are the notable trends driving market growth?
N/A
7. Are there any restraints impacting market growth?
N/A
8. Can you provide examples of recent developments in the market?
N/A
9. What pricing options are available for accessing the report?
Pricing options include single-user, multi-user, and enterprise licenses priced at USD 4900.00, USD 7350.00, and USD 9800.00 respectively.
10. Is the market size provided in terms of value or volume?
The market size is provided in terms of value, measured in N/A.
11. Are there any specific market keywords associated with the report?
Yes, the market keyword associated with the report is "Itopride Hydrochloride Tablets," which aids in identifying and referencing the specific market segment covered.
12. How do I determine which pricing option suits my needs best?
The pricing options vary based on user requirements and access needs. Individual users may opt for single-user licenses, while businesses requiring broader access may choose multi-user or enterprise licenses for cost-effective access to the report.
13. Are there any additional resources or data provided in the Itopride Hydrochloride Tablets report?
While the report offers comprehensive insights, it's advisable to review the specific contents or supplementary materials provided to ascertain if additional resources or data are available.
14. How can I stay updated on further developments or reports in the Itopride Hydrochloride Tablets?
To stay informed about further developments, trends, and reports in the Itopride Hydrochloride Tablets, consider subscribing to industry newsletters, following relevant companies and organizations, or regularly checking reputable industry news sources and publications.
Methodology
Step 1 - Identification of Relevant Samples Size from Population Database



Step 2 - Approaches for Defining Global Market Size (Value, Volume* & Price*)

Note*: In applicable scenarios
Step 3 - Data Sources
Primary Research
- Web Analytics
- Survey Reports
- Research Institute
- Latest Research Reports
- Opinion Leaders
Secondary Research
- Annual Reports
- White Paper
- Latest Press Release
- Industry Association
- Paid Database
- Investor Presentations

Step 4 - Data Triangulation
Involves using different sources of information in order to increase the validity of a study
These sources are likely to be stakeholders in a program - participants, other researchers, program staff, other community members, and so on.
Then we put all data in single framework & apply various statistical tools to find out the dynamic on the market.
During the analysis stage, feedback from the stakeholder groups would be compared to determine areas of agreement as well as areas of divergence


