Key Insights
The global Recombinant Human Erythropoietin (RHE) Injection market is poised for significant expansion, projected to reach $8,889 million in 2025. This robust growth is fueled by a CAGR of 6.3% over the forecast period, indicating a dynamic and expanding sector. A primary driver for this upward trajectory is the increasing prevalence of renal failure anemia, a condition directly addressed by RHE injections. As the global population ages and the incidence of chronic kidney disease (CKD) rises, the demand for effective anemia treatments in these patients is expected to surge. Furthermore, advancements in biotechnology and manufacturing processes are leading to more accessible and cost-effective RHE formulations, expanding their reach to a wider patient base, including those suffering from non-renal anemia where erythropoietin deficiency may also be a factor. The ongoing research into new applications and improved delivery mechanisms for erythropoietin also contributes to market vitality.
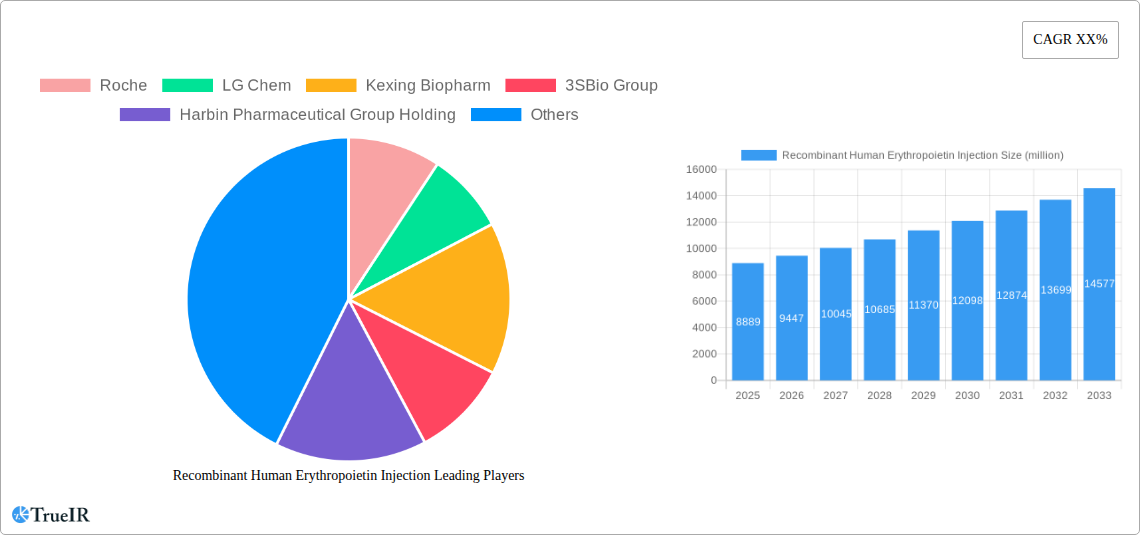
Recombinant Human Erythropoietin Injection Market Size (In Billion)

The market is segmented by application into Renal Failure Anemia and Non-Renal Anemia, with Renal Failure Anemia being the dominant segment due to the direct correlation between kidney dysfunction and erythropoietin production. Within types, the market sees offerings in various volumes such as 0.3ml, 0.6ml, and 1ml branches, catering to diverse treatment protocols and patient needs. Key players like Roche, LG Chem, and Kexing Biopharm are actively investing in research and development, expanding their product portfolios, and strengthening their geographical presence. The market's growth is particularly strong in regions with a high prevalence of CKD and robust healthcare infrastructures, such as North America and Europe, with Asia Pacific emerging as a rapid growth area due to increasing healthcare expenditure and awareness. Challenges such as the high cost of treatment and the availability of biosimilars are being addressed through technological innovation and strategic market penetration.
Recombinant Human Erythropoietin Injection Company Market Share

This comprehensive report provides an in-depth analysis of the global Recombinant Human Erythropoietin (rhEPO) Injection market. Spanning the historical period of 2019–2024, with a base year of 2025 and a forecast period extending to 2033, this study offers critical insights into market structure, trends, opportunities, dominant segments, product advancements, key drivers, challenges, and the competitive landscape. Leveraging high-volume keywords and detailed market intelligence, this report is designed for industry stakeholders seeking to understand and capitalize on the burgeoning rhEPO injection market.
Recombinant Human Erythropoietin Injection Market Structure & Competitive Landscape
The Recombinant Human Erythropoietin Injection market exhibits a moderately concentrated structure, with a handful of dominant global players and a growing number of regional manufacturers. Innovation drivers are primarily focused on enhancing product efficacy, reducing immunogenicity, and developing more convenient delivery mechanisms. Regulatory impacts, particularly stringent approvals from bodies like the FDA and EMA, significantly influence market entry and product development timelines. Product substitutes, such as iron supplements and newer anemia treatments, pose a competitive threat but rhEPO remains the gold standard for many indications. End-user segmentation includes hospitals, clinics, and home healthcare settings, each with distinct purchasing patterns and service needs. Merger and acquisition (M&A) trends are observed as companies seek to expand their product portfolios, geographic reach, and manufacturing capabilities. For instance, the past five years have seen approximately 5-10 significant M&A activities, with deal values often exceeding $100 million for key acquisitions. Concentration ratios (CR4) are estimated to be around 45-55%, indicating a significant market share held by the top four players.
Recombinant Human Erythropoietin Injection Market Trends & Opportunities
The global Recombinant Human Erythropoietin Injection market is poised for robust growth, projected to witness a Compound Annual Growth Rate (CAGR) of approximately 7-9% during the forecast period of 2025–2033. This expansion is fueled by the increasing prevalence of chronic kidney disease (CKD) and associated anemia, driving demand for rhEPO injections as a primary therapeutic intervention. Technological shifts are evident in the development of biosimil rhEPO, which are gaining traction due to their cost-effectiveness and wider accessibility, particularly in emerging markets. Consumer preferences are leaning towards treatments that offer improved patient compliance and reduced administration frequency. The market penetration rate for rhEPO in treating renal failure anemia is already substantial, estimated at over 60% in developed economies, with significant growth potential in underserved regions. Competitive dynamics are intensifying as companies invest heavily in research and development to create next-generation erythropoiesis-stimulating agents (ESAs) with enhanced safety profiles and therapeutic benefits. The market size is expected to grow from an estimated $5,000 million in the base year 2025 to over $10,000 million by 2033. Opportunities lie in developing novel formulations, expanding indications beyond traditional renal failure anemia, and leveraging digital health platforms for patient monitoring and support. The increasing adoption of value-based healthcare models also presents an opportunity for rhEPO manufacturers to demonstrate the economic benefits of their therapies in improving patient outcomes and reducing healthcare costs. The growing awareness of anemia management in non-renal conditions, such as cancer chemotherapy-induced anemia and anemia of chronic disease, is further broadening the market landscape.
Dominant Markets & Segments in Recombinant Human Erythropoietin Injection
The Renal Failure Anemia segment dominates the Recombinant Human Erythropoietin Injection market, driven by the high global burden of chronic kidney disease. This segment is projected to account for over 70% of the market revenue during the forecast period. Key growth drivers in this segment include the aging global population, increasing incidence of diabetes and hypertension – major precursors to CKD, and advancements in dialysis technologies that extend patient lifespans, thereby increasing the need for anemia management. Geographically, North America and Europe are currently the largest markets, owing to established healthcare infrastructures, higher healthcare expenditure, and widespread adoption of rhEPO therapies. However, the Asia-Pacific region is exhibiting the fastest growth, propelled by increasing healthcare access, a growing middle class, and rising awareness about anemia treatment.
Application Dominance:
- Renal Failure Anemia: The cornerstone of the rhEPO market, driven by widespread CKD prevalence and established treatment protocols.
- Non Renal Anemia: Emerging segment with growing applications in chemotherapy-induced anemia, anemia of chronic disease, and pre-operative anemia.
Type Dominance:
- 0.6ml Branch: This size is widely adopted due to its convenience and suitability for typical rhEPO dosing requirements in renal failure patients.
- 0.3ml Branch & 1ml Branch: Cater to specific patient needs and physician preferences, offering flexibility in dosage administration.
- Others: Includes larger volume presentations or specialized formulations.
The market dominance in terms of value is expected to remain with North America and Europe through 2028, with the Asia-Pacific region projected to witness a CAGR of approximately 9-11%, surpassing other regions in growth velocity by 2030. Government policies supporting chronic disease management and healthcare access in countries like China and India are significant accelerators for this regional growth.
Recombinant Human Erythropoietin Injection Product Analysis
Product innovation in Recombinant Human Erythropoietin Injection is centered on developing biosimil versions, enhancing formulation stability, and exploring novel drug delivery systems. These advancements aim to improve patient compliance, reduce treatment costs, and broaden therapeutic applications. Competitive advantages stem from robust clinical data, strong regulatory approvals, and established manufacturing expertise. The market fit for rhEPO injections remains strong due to their proven efficacy in stimulating red blood cell production and managing anemia across various chronic conditions. Technological advancements are leading to the development of longer-acting formulations, reducing injection frequency and improving patient quality of life.
Key Drivers, Barriers & Challenges in Recombinant Human Erythropoietin Injection
Key Drivers:
- Rising prevalence of Chronic Kidney Disease (CKD): A primary driver, increasing the demand for rhEPO to treat renal anemia.
- Aging Global Population: Older demographics are more susceptible to chronic diseases, including anemia.
- Technological Advancements: Development of biosimil rhEPO and novel formulations enhancing efficacy and patient compliance.
- Expanding Therapeutic Indications: Growing use in non-renal anemia, such as chemotherapy-induced anemia.
Barriers & Challenges:
- High Manufacturing Costs: Complex production processes contribute to significant product costs.
- Stringent Regulatory Landscape: Extensive clinical trials and regulatory hurdles for new product approvals.
- Competition from Biosimil Products: Increasing availability of biosimil rhEPO exerts downward pressure on pricing.
- Potential for Adverse Events: Risks associated with iron overload and thromboembolic events necessitate careful patient monitoring.
- Reimbursement Policies: Variations in healthcare reimbursement policies across different countries can impact market access and affordability. The global market faces an estimated 10-15% impact from fluctuating reimbursement rates.
Growth Drivers in the Recombinant Human Erythropoietin Injection Market
The growth of the Recombinant Human Erythropoietin Injection market is propelled by several key factors. The increasing global incidence of chronic kidney disease, a direct consequence of rising rates of diabetes and hypertension, is a monumental driver, creating a consistent and growing demand for rhEPO to combat anemia. Technological advancements in biopharmaceutical manufacturing have enabled the development of more cost-effective biosimilar rhEPO products, thereby increasing accessibility for a wider patient population, particularly in emerging economies. Furthermore, the expanding understanding of anemia in non-renal conditions, such as cancer therapy-induced anemia and anemia of chronic disease, is opening up new avenues for rhEPO application. Economic factors, including increasing healthcare expenditure in developing nations and government initiatives to improve access to essential medicines, also significantly contribute to market expansion.
Challenges Impacting Recombinant Human Erythropoietin Injection Growth
Despite its growth potential, the Recombinant Human Erythropoietin Injection market faces several formidable challenges. The complex and highly regulated nature of biopharmaceutical production leads to significant manufacturing costs, which translate into high product prices. Regulatory hurdles, including stringent clinical trial requirements and lengthy approval processes by health authorities, can delay market entry for new products and biosimil versions. The growing presence of biosimilar rhEPO products, while beneficial for affordability, intensifies price competition among manufacturers, potentially impacting profit margins. Supply chain complexities and the need for cold chain logistics add another layer of challenge. Moreover, the potential for adverse events associated with rhEPO therapy, such as polycythemia and thrombotic events, necessitates careful patient monitoring and can lead to increased scrutiny from regulatory bodies and healthcare providers.
Key Players Shaping the Recombinant Human Erythropoietin Injection Market
- Roche
- LG Chem
- Kexing Biopharm
- 3SBio Group
- Harbin Pharmaceutical Group Holding
- Dong-E-E-Jiao
- Humanwell Healthcare
- Ncpc Genetech Biotechnology
- Shandong E-hua Biotech Pharmaceutical
- Intas Pharmaceuticals
- PeproTech
- WELLONA PHARMA
- SI HUAN SHENG WU
- Shanxi Weiqidaguangming Pharmaceutical
Significant Recombinant Human Erythropoietin Injection Industry Milestones
- 2019 December: Launch of a new biosimilar rhEPO in a key European market, impacting pricing dynamics.
- 2020 April: FDA approval for expanded use of rhEPO in a non-renal anemia indication, opening new market segments.
- 2021 June: Major acquisition of a regional rhEPO manufacturer by a global biopharmaceutical company, consolidating market share.
- 2022 October: Introduction of a novel long-acting rhEPO formulation in Asian markets, enhancing patient convenience.
- 2023 March: Significant investment in R&D for next-generation ESAs by leading industry players, signaling future innovation.
- 2024 January: Harmonization of regulatory guidelines for biosimilar rhEPO in several major economies, facilitating market entry.
Future Outlook for Recombinant Human Erythropoietin Injection Market
The future outlook for the Recombinant Human Erythropoietin Injection market is highly promising, driven by sustained demand from an expanding patient pool suffering from anemia associated with chronic diseases. Strategic opportunities lie in the ongoing development of novel, high-potency, and long-acting rhEPO formulations that offer enhanced patient convenience and adherence, potentially commanding premium pricing. The increasing penetration of biosimilar rhEPO in emerging markets will drive volume growth and market accessibility. Furthermore, the exploration of rhEPO's therapeutic benefits in under-addressed anemia types and its integration into comprehensive treatment protocols for chronic diseases will unlock further market potential. The market is expected to see continued innovation and strategic collaborations, reinforcing its trajectory of significant growth and value creation, with an estimated market expansion of over 100% from the base year 2025 to 2033.
Recombinant Human Erythropoietin Injection Segmentation
-
1. Application
- 1.1. Renal Failure Anemia
- 1.2. Non Renal Anemia
-
2. Types
- 2.1. 0.3ml Branch
- 2.2. 0.6ml Branch
- 2.3. 1ml Branch
- 2.4. Others
Recombinant Human Erythropoietin Injection Segmentation By Geography
-
1. North America
- 1.1. United States
- 1.2. Canada
- 1.3. Mexico
-
2. South America
- 2.1. Brazil
- 2.2. Argentina
- 2.3. Rest of South America
-
3. Europe
- 3.1. United Kingdom
- 3.2. Germany
- 3.3. France
- 3.4. Italy
- 3.5. Spain
- 3.6. Russia
- 3.7. Benelux
- 3.8. Nordics
- 3.9. Rest of Europe
-
4. Middle East & Africa
- 4.1. Turkey
- 4.2. Israel
- 4.3. GCC
- 4.4. North Africa
- 4.5. South Africa
- 4.6. Rest of Middle East & Africa
-
5. Asia Pacific
- 5.1. China
- 5.2. India
- 5.3. Japan
- 5.4. South Korea
- 5.5. ASEAN
- 5.6. Oceania
- 5.7. Rest of Asia Pacific
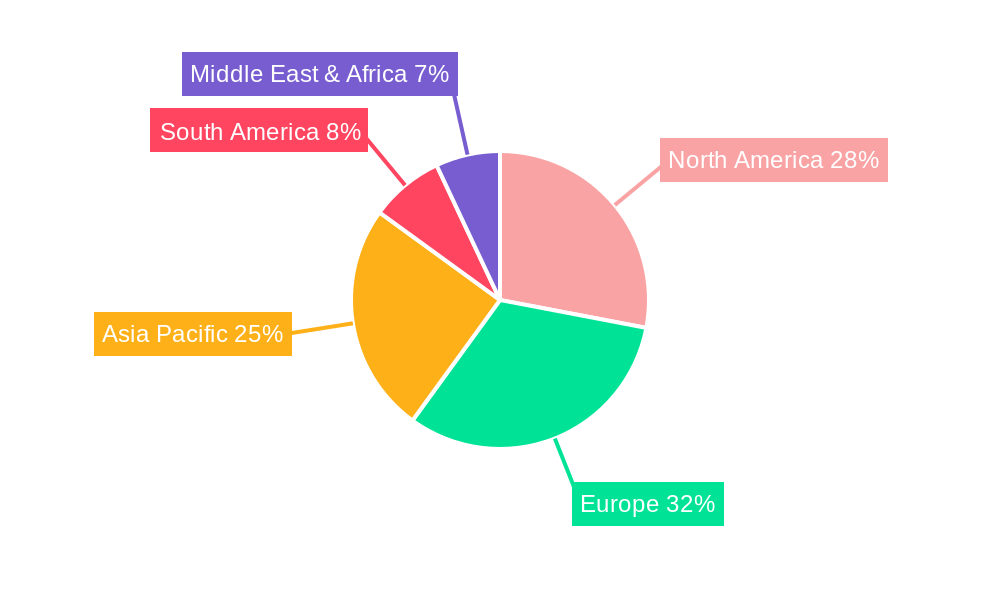
Recombinant Human Erythropoietin Injection Regional Market Share

Geographic Coverage of Recombinant Human Erythropoietin Injection
Recombinant Human Erythropoietin Injection REPORT HIGHLIGHTS
| Aspects | Details |
|---|---|
| Study Period | 2020-2034 |
| Base Year | 2025 |
| Estimated Year | 2026 |
| Forecast Period | 2026-2034 |
| Historical Period | 2020-2025 |
| Growth Rate | CAGR of 6.3% from 2020-2034 |
| Segmentation |
|
Table of Contents
- 1. Introduction
- 1.1. Research Scope
- 1.2. Market Segmentation
- 1.3. Research Methodology
- 1.4. Definitions and Assumptions
- 2. Executive Summary
- 2.1. Introduction
- 3. Market Dynamics
- 3.1. Introduction
- 3.2. Market Drivers
- 3.3. Market Restrains
- 3.4. Market Trends
- 4. Market Factor Analysis
- 4.1. Porters Five Forces
- 4.2. Supply/Value Chain
- 4.3. PESTEL analysis
- 4.4. Market Entropy
- 4.5. Patent/Trademark Analysis
- 5. Global Recombinant Human Erythropoietin Injection Analysis, Insights and Forecast, 2020-2032
- 5.1. Market Analysis, Insights and Forecast - by Application
- 5.1.1. Renal Failure Anemia
- 5.1.2. Non Renal Anemia
- 5.2. Market Analysis, Insights and Forecast - by Types
- 5.2.1. 0.3ml Branch
- 5.2.2. 0.6ml Branch
- 5.2.3. 1ml Branch
- 5.2.4. Others
- 5.3. Market Analysis, Insights and Forecast - by Region
- 5.3.1. North America
- 5.3.2. South America
- 5.3.3. Europe
- 5.3.4. Middle East & Africa
- 5.3.5. Asia Pacific
- 5.1. Market Analysis, Insights and Forecast - by Application
- 6. North America Recombinant Human Erythropoietin Injection Analysis, Insights and Forecast, 2020-2032
- 6.1. Market Analysis, Insights and Forecast - by Application
- 6.1.1. Renal Failure Anemia
- 6.1.2. Non Renal Anemia
- 6.2. Market Analysis, Insights and Forecast - by Types
- 6.2.1. 0.3ml Branch
- 6.2.2. 0.6ml Branch
- 6.2.3. 1ml Branch
- 6.2.4. Others
- 6.1. Market Analysis, Insights and Forecast - by Application
- 7. South America Recombinant Human Erythropoietin Injection Analysis, Insights and Forecast, 2020-2032
- 7.1. Market Analysis, Insights and Forecast - by Application
- 7.1.1. Renal Failure Anemia
- 7.1.2. Non Renal Anemia
- 7.2. Market Analysis, Insights and Forecast - by Types
- 7.2.1. 0.3ml Branch
- 7.2.2. 0.6ml Branch
- 7.2.3. 1ml Branch
- 7.2.4. Others
- 7.1. Market Analysis, Insights and Forecast - by Application
- 8. Europe Recombinant Human Erythropoietin Injection Analysis, Insights and Forecast, 2020-2032
- 8.1. Market Analysis, Insights and Forecast - by Application
- 8.1.1. Renal Failure Anemia
- 8.1.2. Non Renal Anemia
- 8.2. Market Analysis, Insights and Forecast - by Types
- 8.2.1. 0.3ml Branch
- 8.2.2. 0.6ml Branch
- 8.2.3. 1ml Branch
- 8.2.4. Others
- 8.1. Market Analysis, Insights and Forecast - by Application
- 9. Middle East & Africa Recombinant Human Erythropoietin Injection Analysis, Insights and Forecast, 2020-2032
- 9.1. Market Analysis, Insights and Forecast - by Application
- 9.1.1. Renal Failure Anemia
- 9.1.2. Non Renal Anemia
- 9.2. Market Analysis, Insights and Forecast - by Types
- 9.2.1. 0.3ml Branch
- 9.2.2. 0.6ml Branch
- 9.2.3. 1ml Branch
- 9.2.4. Others
- 9.1. Market Analysis, Insights and Forecast - by Application
- 10. Asia Pacific Recombinant Human Erythropoietin Injection Analysis, Insights and Forecast, 2020-2032
- 10.1. Market Analysis, Insights and Forecast - by Application
- 10.1.1. Renal Failure Anemia
- 10.1.2. Non Renal Anemia
- 10.2. Market Analysis, Insights and Forecast - by Types
- 10.2.1. 0.3ml Branch
- 10.2.2. 0.6ml Branch
- 10.2.3. 1ml Branch
- 10.2.4. Others
- 10.1. Market Analysis, Insights and Forecast - by Application
- 11. Competitive Analysis
- 11.1. Global Market Share Analysis 2025
- 11.2. Company Profiles
- 11.2.1 Roche
- 11.2.1.1. Overview
- 11.2.1.2. Products
- 11.2.1.3. SWOT Analysis
- 11.2.1.4. Recent Developments
- 11.2.1.5. Financials (Based on Availability)
- 11.2.2 LG Chem
- 11.2.2.1. Overview
- 11.2.2.2. Products
- 11.2.2.3. SWOT Analysis
- 11.2.2.4. Recent Developments
- 11.2.2.5. Financials (Based on Availability)
- 11.2.3 Kexing Biopharm
- 11.2.3.1. Overview
- 11.2.3.2. Products
- 11.2.3.3. SWOT Analysis
- 11.2.3.4. Recent Developments
- 11.2.3.5. Financials (Based on Availability)
- 11.2.4 3SBio Group
- 11.2.4.1. Overview
- 11.2.4.2. Products
- 11.2.4.3. SWOT Analysis
- 11.2.4.4. Recent Developments
- 11.2.4.5. Financials (Based on Availability)
- 11.2.5 Harbin Pharmaceutical Group Holding
- 11.2.5.1. Overview
- 11.2.5.2. Products
- 11.2.5.3. SWOT Analysis
- 11.2.5.4. Recent Developments
- 11.2.5.5. Financials (Based on Availability)
- 11.2.6 Dong-E-E-Jiao
- 11.2.6.1. Overview
- 11.2.6.2. Products
- 11.2.6.3. SWOT Analysis
- 11.2.6.4. Recent Developments
- 11.2.6.5. Financials (Based on Availability)
- 11.2.7 Humanwell Healthcare
- 11.2.7.1. Overview
- 11.2.7.2. Products
- 11.2.7.3. SWOT Analysis
- 11.2.7.4. Recent Developments
- 11.2.7.5. Financials (Based on Availability)
- 11.2.8 Ncpc Genetech Biotechnology
- 11.2.8.1. Overview
- 11.2.8.2. Products
- 11.2.8.3. SWOT Analysis
- 11.2.8.4. Recent Developments
- 11.2.8.5. Financials (Based on Availability)
- 11.2.9 Shandong E-hua Biotech Pharmaceutical
- 11.2.9.1. Overview
- 11.2.9.2. Products
- 11.2.9.3. SWOT Analysis
- 11.2.9.4. Recent Developments
- 11.2.9.5. Financials (Based on Availability)
- 11.2.10 Intas Pharmaceuticals
- 11.2.10.1. Overview
- 11.2.10.2. Products
- 11.2.10.3. SWOT Analysis
- 11.2.10.4. Recent Developments
- 11.2.10.5. Financials (Based on Availability)
- 11.2.11 PeproTech
- 11.2.11.1. Overview
- 11.2.11.2. Products
- 11.2.11.3. SWOT Analysis
- 11.2.11.4. Recent Developments
- 11.2.11.5. Financials (Based on Availability)
- 11.2.12 WELLONA PHARMA
- 11.2.12.1. Overview
- 11.2.12.2. Products
- 11.2.12.3. SWOT Analysis
- 11.2.12.4. Recent Developments
- 11.2.12.5. Financials (Based on Availability)
- 11.2.13 SI HUAN SHENG WU
- 11.2.13.1. Overview
- 11.2.13.2. Products
- 11.2.13.3. SWOT Analysis
- 11.2.13.4. Recent Developments
- 11.2.13.5. Financials (Based on Availability)
- 11.2.14 Shanxi Weiqidaguangming Pharmaceutical
- 11.2.14.1. Overview
- 11.2.14.2. Products
- 11.2.14.3. SWOT Analysis
- 11.2.14.4. Recent Developments
- 11.2.14.5. Financials (Based on Availability)
- 11.2.1 Roche
List of Figures
- Figure 1: Global Recombinant Human Erythropoietin Injection Revenue Breakdown (undefined, %) by Region 2025 & 2033
- Figure 2: North America Recombinant Human Erythropoietin Injection Revenue (undefined), by Application 2025 & 2033
- Figure 3: North America Recombinant Human Erythropoietin Injection Revenue Share (%), by Application 2025 & 2033
- Figure 4: North America Recombinant Human Erythropoietin Injection Revenue (undefined), by Types 2025 & 2033
- Figure 5: North America Recombinant Human Erythropoietin Injection Revenue Share (%), by Types 2025 & 2033
- Figure 6: North America Recombinant Human Erythropoietin Injection Revenue (undefined), by Country 2025 & 2033
- Figure 7: North America Recombinant Human Erythropoietin Injection Revenue Share (%), by Country 2025 & 2033
- Figure 8: South America Recombinant Human Erythropoietin Injection Revenue (undefined), by Application 2025 & 2033
- Figure 9: South America Recombinant Human Erythropoietin Injection Revenue Share (%), by Application 2025 & 2033
- Figure 10: South America Recombinant Human Erythropoietin Injection Revenue (undefined), by Types 2025 & 2033
- Figure 11: South America Recombinant Human Erythropoietin Injection Revenue Share (%), by Types 2025 & 2033
- Figure 12: South America Recombinant Human Erythropoietin Injection Revenue (undefined), by Country 2025 & 2033
- Figure 13: South America Recombinant Human Erythropoietin Injection Revenue Share (%), by Country 2025 & 2033
- Figure 14: Europe Recombinant Human Erythropoietin Injection Revenue (undefined), by Application 2025 & 2033
- Figure 15: Europe Recombinant Human Erythropoietin Injection Revenue Share (%), by Application 2025 & 2033
- Figure 16: Europe Recombinant Human Erythropoietin Injection Revenue (undefined), by Types 2025 & 2033
- Figure 17: Europe Recombinant Human Erythropoietin Injection Revenue Share (%), by Types 2025 & 2033
- Figure 18: Europe Recombinant Human Erythropoietin Injection Revenue (undefined), by Country 2025 & 2033
- Figure 19: Europe Recombinant Human Erythropoietin Injection Revenue Share (%), by Country 2025 & 2033
- Figure 20: Middle East & Africa Recombinant Human Erythropoietin Injection Revenue (undefined), by Application 2025 & 2033
- Figure 21: Middle East & Africa Recombinant Human Erythropoietin Injection Revenue Share (%), by Application 2025 & 2033
- Figure 22: Middle East & Africa Recombinant Human Erythropoietin Injection Revenue (undefined), by Types 2025 & 2033
- Figure 23: Middle East & Africa Recombinant Human Erythropoietin Injection Revenue Share (%), by Types 2025 & 2033
- Figure 24: Middle East & Africa Recombinant Human Erythropoietin Injection Revenue (undefined), by Country 2025 & 2033
- Figure 25: Middle East & Africa Recombinant Human Erythropoietin Injection Revenue Share (%), by Country 2025 & 2033
- Figure 26: Asia Pacific Recombinant Human Erythropoietin Injection Revenue (undefined), by Application 2025 & 2033
- Figure 27: Asia Pacific Recombinant Human Erythropoietin Injection Revenue Share (%), by Application 2025 & 2033
- Figure 28: Asia Pacific Recombinant Human Erythropoietin Injection Revenue (undefined), by Types 2025 & 2033
- Figure 29: Asia Pacific Recombinant Human Erythropoietin Injection Revenue Share (%), by Types 2025 & 2033
- Figure 30: Asia Pacific Recombinant Human Erythropoietin Injection Revenue (undefined), by Country 2025 & 2033
- Figure 31: Asia Pacific Recombinant Human Erythropoietin Injection Revenue Share (%), by Country 2025 & 2033
List of Tables
- Table 1: Global Recombinant Human Erythropoietin Injection Revenue undefined Forecast, by Application 2020 & 2033
- Table 2: Global Recombinant Human Erythropoietin Injection Revenue undefined Forecast, by Types 2020 & 2033
- Table 3: Global Recombinant Human Erythropoietin Injection Revenue undefined Forecast, by Region 2020 & 2033
- Table 4: Global Recombinant Human Erythropoietin Injection Revenue undefined Forecast, by Application 2020 & 2033
- Table 5: Global Recombinant Human Erythropoietin Injection Revenue undefined Forecast, by Types 2020 & 2033
- Table 6: Global Recombinant Human Erythropoietin Injection Revenue undefined Forecast, by Country 2020 & 2033
- Table 7: United States Recombinant Human Erythropoietin Injection Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 8: Canada Recombinant Human Erythropoietin Injection Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 9: Mexico Recombinant Human Erythropoietin Injection Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 10: Global Recombinant Human Erythropoietin Injection Revenue undefined Forecast, by Application 2020 & 2033
- Table 11: Global Recombinant Human Erythropoietin Injection Revenue undefined Forecast, by Types 2020 & 2033
- Table 12: Global Recombinant Human Erythropoietin Injection Revenue undefined Forecast, by Country 2020 & 2033
- Table 13: Brazil Recombinant Human Erythropoietin Injection Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 14: Argentina Recombinant Human Erythropoietin Injection Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 15: Rest of South America Recombinant Human Erythropoietin Injection Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 16: Global Recombinant Human Erythropoietin Injection Revenue undefined Forecast, by Application 2020 & 2033
- Table 17: Global Recombinant Human Erythropoietin Injection Revenue undefined Forecast, by Types 2020 & 2033
- Table 18: Global Recombinant Human Erythropoietin Injection Revenue undefined Forecast, by Country 2020 & 2033
- Table 19: United Kingdom Recombinant Human Erythropoietin Injection Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 20: Germany Recombinant Human Erythropoietin Injection Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 21: France Recombinant Human Erythropoietin Injection Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 22: Italy Recombinant Human Erythropoietin Injection Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 23: Spain Recombinant Human Erythropoietin Injection Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 24: Russia Recombinant Human Erythropoietin Injection Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 25: Benelux Recombinant Human Erythropoietin Injection Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 26: Nordics Recombinant Human Erythropoietin Injection Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 27: Rest of Europe Recombinant Human Erythropoietin Injection Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 28: Global Recombinant Human Erythropoietin Injection Revenue undefined Forecast, by Application 2020 & 2033
- Table 29: Global Recombinant Human Erythropoietin Injection Revenue undefined Forecast, by Types 2020 & 2033
- Table 30: Global Recombinant Human Erythropoietin Injection Revenue undefined Forecast, by Country 2020 & 2033
- Table 31: Turkey Recombinant Human Erythropoietin Injection Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 32: Israel Recombinant Human Erythropoietin Injection Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 33: GCC Recombinant Human Erythropoietin Injection Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 34: North Africa Recombinant Human Erythropoietin Injection Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 35: South Africa Recombinant Human Erythropoietin Injection Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 36: Rest of Middle East & Africa Recombinant Human Erythropoietin Injection Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 37: Global Recombinant Human Erythropoietin Injection Revenue undefined Forecast, by Application 2020 & 2033
- Table 38: Global Recombinant Human Erythropoietin Injection Revenue undefined Forecast, by Types 2020 & 2033
- Table 39: Global Recombinant Human Erythropoietin Injection Revenue undefined Forecast, by Country 2020 & 2033
- Table 40: China Recombinant Human Erythropoietin Injection Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 41: India Recombinant Human Erythropoietin Injection Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 42: Japan Recombinant Human Erythropoietin Injection Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 43: South Korea Recombinant Human Erythropoietin Injection Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 44: ASEAN Recombinant Human Erythropoietin Injection Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 45: Oceania Recombinant Human Erythropoietin Injection Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 46: Rest of Asia Pacific Recombinant Human Erythropoietin Injection Revenue (undefined) Forecast, by Application 2020 & 2033
Frequently Asked Questions
1. What is the projected Compound Annual Growth Rate (CAGR) of the Recombinant Human Erythropoietin Injection?
The projected CAGR is approximately 6.3%.
2. Which companies are prominent players in the Recombinant Human Erythropoietin Injection?
Key companies in the market include Roche, LG Chem, Kexing Biopharm, 3SBio Group, Harbin Pharmaceutical Group Holding, Dong-E-E-Jiao, Humanwell Healthcare, Ncpc Genetech Biotechnology, Shandong E-hua Biotech Pharmaceutical, Intas Pharmaceuticals, PeproTech, WELLONA PHARMA, SI HUAN SHENG WU, Shanxi Weiqidaguangming Pharmaceutical.
3. What are the main segments of the Recombinant Human Erythropoietin Injection?
The market segments include Application, Types.
4. Can you provide details about the market size?
The market size is estimated to be USD XXX N/A as of 2022.
5. What are some drivers contributing to market growth?
N/A
6. What are the notable trends driving market growth?
N/A
7. Are there any restraints impacting market growth?
N/A
8. Can you provide examples of recent developments in the market?
N/A
9. What pricing options are available for accessing the report?
Pricing options include single-user, multi-user, and enterprise licenses priced at USD 2900.00, USD 4350.00, and USD 5800.00 respectively.
10. Is the market size provided in terms of value or volume?
The market size is provided in terms of value, measured in N/A.
11. Are there any specific market keywords associated with the report?
Yes, the market keyword associated with the report is "Recombinant Human Erythropoietin Injection," which aids in identifying and referencing the specific market segment covered.
12. How do I determine which pricing option suits my needs best?
The pricing options vary based on user requirements and access needs. Individual users may opt for single-user licenses, while businesses requiring broader access may choose multi-user or enterprise licenses for cost-effective access to the report.
13. Are there any additional resources or data provided in the Recombinant Human Erythropoietin Injection report?
While the report offers comprehensive insights, it's advisable to review the specific contents or supplementary materials provided to ascertain if additional resources or data are available.
14. How can I stay updated on further developments or reports in the Recombinant Human Erythropoietin Injection?
To stay informed about further developments, trends, and reports in the Recombinant Human Erythropoietin Injection, consider subscribing to industry newsletters, following relevant companies and organizations, or regularly checking reputable industry news sources and publications.
Methodology
Step 1 - Identification of Relevant Samples Size from Population Database



Step 2 - Approaches for Defining Global Market Size (Value, Volume* & Price*)

Note*: In applicable scenarios
Step 3 - Data Sources
Primary Research
- Web Analytics
- Survey Reports
- Research Institute
- Latest Research Reports
- Opinion Leaders
Secondary Research
- Annual Reports
- White Paper
- Latest Press Release
- Industry Association
- Paid Database
- Investor Presentations

Step 4 - Data Triangulation
Involves using different sources of information in order to increase the validity of a study
These sources are likely to be stakeholders in a program - participants, other researchers, program staff, other community members, and so on.
Then we put all data in single framework & apply various statistical tools to find out the dynamic on the market.
During the analysis stage, feedback from the stakeholder groups would be compared to determine areas of agreement as well as areas of divergence


