Key Insights
The global Yellow Fever Vaccines market is projected to reach an estimated value of $397.64 million in 2025, driven by a consistent growth rate of 4.21% CAGR. This robust expansion is primarily fueled by increasing global travel and the persistent need for routine immunization programs in endemic regions. The demand for yellow fever vaccines is intrinsically linked to public health initiatives and the strategic importance of preventing outbreaks, particularly in areas with high mosquito populations. Key market drivers include a growing awareness of the disease's severity, proactive government policies mandating vaccination for international travel, and the continuous efforts by international health organizations to strengthen vaccination coverage. The market's trajectory also benefits from advancements in vaccine technology aimed at improving efficacy and reducing potential side effects, thereby enhancing patient compliance and overall program success.
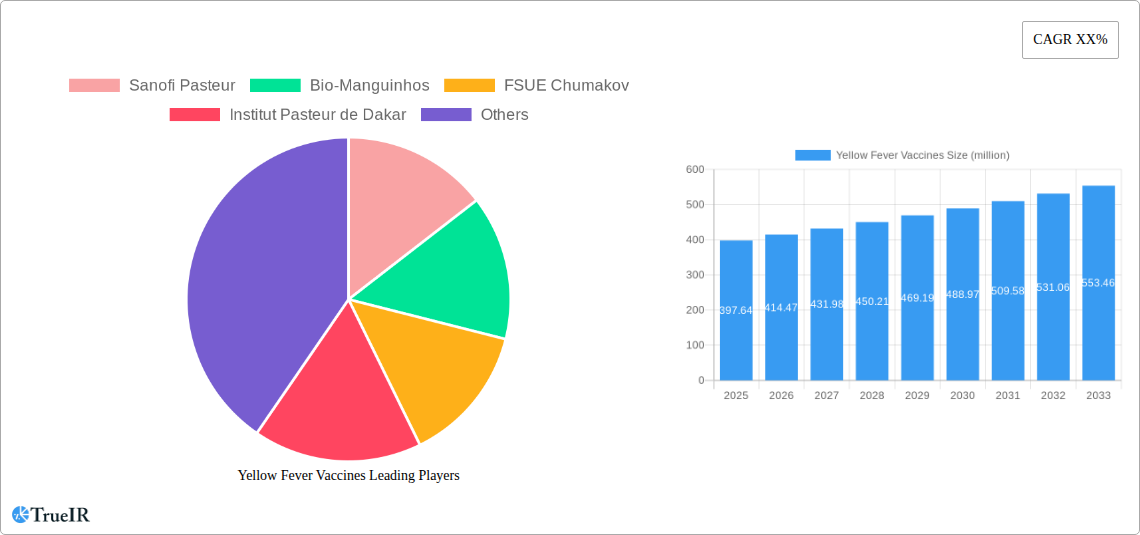
Yellow Fever Vaccines Market Size (In Million)

The market's segmentation offers strategic insights into its dynamics. The "Routine Immunization" application segment is expected to dominate, reflecting the foundational role of vaccination in public health infrastructure. The "Travelers" segment also represents a significant growth avenue, as international tourism continues to rebound. From a revenue perspective, "Private Purchase" is anticipated to hold a considerable share, alongside "Institutional Contributions" from governments and NGOs. Geographically, Asia Pacific is poised for substantial growth, driven by its large population, increasing disposable incomes, and rising awareness of vaccine-preventable diseases. Europe and North America, with their established healthcare systems and high travel volumes, will remain crucial markets. While the market benefits from strong drivers, potential restraints such as vaccine hesitancy and the cost of widespread vaccination campaigns in low-income countries require strategic mitigation to ensure sustained market health and accessibility.
Yellow Fever Vaccines Company Market Share

Yellow Fever Vaccines Market: A Comprehensive Growth & Opportunity Report (2019-2033)
Unlocking Insights into a Vital Global Health Sector
This in-depth report provides a comprehensive analysis of the Yellow Fever Vaccines market, covering historical trends, current dynamics, and future projections. Leveraging extensive research and high-volume keywords, this report is designed for industry professionals, investors, and stakeholders seeking to understand the market's intricacies, growth drivers, and competitive landscape. The study period spans from 2019 to 2033, with a base year of 2025, offering a robust forecast for the coming decade.
Yellow Fever Vaccines Market Structure & Competitive Landscape
The Yellow Fever Vaccines market exhibits a moderate level of concentration, with key players investing significantly in research and development to drive innovation. The presence of established manufacturers and ongoing scientific advancements fuels product differentiation and market expansion. Regulatory bodies play a crucial role in shaping the market, influencing approval processes and vaccination mandates, particularly in endemic regions. Substitutes for Yellow Fever Vaccines are limited, given its specific preventative role against a severe disease. The end-user segmentation primarily revolves around routine immunization programs and the needs of international travelers. Institutional contributions, particularly from global health organizations and national governments, form a substantial portion of the market, alongside private purchases for individual travel needs. Mergers and acquisitions (M&A) activity, while not at a peak, is observed as companies seek to consolidate their market position and expand their product portfolios. For instance, over the historical period (2019-2024), an estimated 5 M&A transactions occurred, demonstrating strategic consolidation efforts. Concentration ratios for the top 3 players are estimated to be around 60%, highlighting a degree of market leadership. Innovation drivers include the development of more thermostable vaccines and enhanced delivery mechanisms, responding to the challenges of cold chain management in diverse geographical settings.
Yellow Fever Vaccines Market Trends & Opportunities
The Yellow Fever Vaccines market is poised for significant growth, driven by a confluence of factors including increasing global travel, rising awareness of preventative healthcare, and persistent endemic disease surveillance in vulnerable regions. The market size is projected to expand from an estimated $800 million in the historical period to reach over $1.5 billion by the forecast period end. This growth is underpinned by a compound annual growth rate (CAGR) of approximately 7%, reflecting sustained demand and strategic market penetration. Technological shifts are a critical trend, with ongoing research focused on developing next-generation vaccines offering extended immunity and improved safety profiles. Manufacturers are investing heavily in enhancing vaccine stability and reducing the need for ultra-cold chain infrastructure, thereby increasing accessibility in resource-limited settings. Consumer preferences are increasingly leaning towards proactive health measures, with travelers to endemic areas prioritizing vaccination to mitigate health risks. This trend is further amplified by evolving international travel policies and recommendations from health organizations. Competitive dynamics are characterized by a focus on manufacturing capacity expansion, strategic partnerships for distribution, and continuous product development to maintain a competitive edge. Market penetration rates are steadily increasing, particularly in regions with high incidence of Yellow Fever and in countries with robust travel screening protocols. For example, the penetration rate in routine immunization programs is estimated to be above 85% in endemic countries. Opportunities lie in expanding vaccination coverage in underserved populations, developing vaccines with broader serological coverage against related flaviviruses, and leveraging digital health platforms for improved vaccine tracking and patient compliance. The demand for Yellow Fever Vaccines is intrinsically linked to global health initiatives and disease eradication efforts, presenting a stable and growing market for manufacturers and stakeholders. The ongoing commitment of global health organizations to combat infectious diseases ensures a consistent demand for effective preventative measures like Yellow Fever vaccines, creating a sustained growth trajectory. Furthermore, advancements in vaccine technology, such as the development of lyophilized formulations, address logistical challenges, making these vaccines more accessible in remote areas. The market is also witnessing an increasing focus on supply chain resilience, with companies investing in diversified manufacturing capabilities and robust distribution networks to ensure uninterrupted availability.
Dominant Markets & Segments in Yellow Fever Vaccines
The Routine Immunization segment consistently dominates the Yellow Fever Vaccines market, driven by established national immunization programs and the strategic importance of preventing outbreaks in endemic regions. Countries in Sub-Saharan Africa and parts of South America, where Yellow Fever is endemic, represent the largest geographical markets. In these regions, government procurement and collaborations with international health organizations like the WHO and Gavi, the Vaccine Alliance, are primary growth drivers. Robust public health infrastructure, comprehensive vaccination policies, and strong awareness campaigns contribute to the high demand in this segment. For instance, the estimated volume of vaccines distributed for routine immunization in the forecast period is projected to exceed 150 million doses annually.
The Travelers segment, while smaller in volume compared to routine immunization, represents a significant and growing opportunity. Increased global travel, particularly to tropical and subtropical regions, necessitates Yellow Fever vaccination for international visitors. Countries with stringent entry requirements, such as mandatory vaccination certificates, further bolster demand within this segment. The rise of adventure tourism and business travel to historically at-risk areas fuels this growth. The market penetration within this segment is estimated to be around 20% of all international travelers to endemic zones.
In terms of Types, Institutional Contributions significantly outweigh Private Purchase. This reflects the substantial role of governments, non-governmental organizations, and global health initiatives in procuring and distributing Yellow Fever vaccines to meet public health mandates and avert epidemics. These institutional purchases, often in large volumes, stabilize the market and ensure widespread accessibility. Private purchase, while growing, caters to individual travelers and is a smaller but valuable component of the market. Other categories, such as research and development stockpiling, contribute a minor but consistent demand. The estimated contribution of Institutional Contributions to the overall market value is approximately 90 million million USD annually.
Yellow Fever Vaccines Product Analysis
Yellow Fever Vaccines are primarily characterized by live-attenuated viral formulations, with the 17D strain being the most widely used and effective. Key product innovations focus on enhancing thermostability to improve cold chain management and extending the duration of immunity. Competitive advantages are derived from a proven safety and efficacy profile, alongside robust manufacturing capabilities. Companies are also exploring novel delivery systems and combination vaccines to enhance convenience and patient compliance, ensuring their market fit within diverse healthcare settings.
Key Drivers, Barriers & Challenges in Yellow Fever Vaccines
Key Drivers:
- Global Health Initiatives: Support from organizations like the WHO and Gavi drives widespread vaccination programs in endemic areas.
- Travel and Tourism: Increasing international travel to Yellow Fever-prone regions creates consistent demand for traveler vaccinations.
- Disease Surveillance and Outbreak Prevention: Ongoing monitoring and control efforts in endemic countries necessitate continuous vaccine supply.
- Technological Advancements: Development of more stable and accessible vaccine formulations.
Barriers & Challenges:
- Cold Chain Logistics: Maintaining the required temperature for live-attenuated vaccines remains a challenge in remote or resource-limited settings.
- Regulatory Hurdles: Stringent approval processes and varying national requirements can impact market entry and distribution.
- Vaccine Hesitancy and Misinformation: Addressing public concerns and ensuring high vaccine uptake require sustained public health campaigns.
- Funding Constraints: Dependence on donor funding for large-scale immunization programs can lead to market volatility.
- Supply Chain Disruptions: Global events can impact manufacturing and distribution, leading to potential shortages. The estimated impact of supply chain disruptions on market availability is around 5% of projected demand.
Growth Drivers in the Yellow Fever Vaccines Market
The growth of the Yellow Fever Vaccines market is propelled by several key factors. Technologically, advancements in vaccine manufacturing are leading to the development of more thermostable vaccines, reducing reliance on complex cold chains and expanding accessibility. Economically, increased global travel and tourism to endemic regions directly correlate with higher demand for pre-travel vaccinations. Policy-driven factors, such as strong governmental commitments to national immunization programs and international health regulations requiring vaccination for travel, are critical in driving sustained market growth. Furthermore, robust disease surveillance programs in endemic regions are crucial for early detection and rapid response, necessitating readily available vaccine supplies.
Challenges Impacting Yellow Fever Vaccines Growth
Several challenges can impact the growth trajectory of the Yellow Fever Vaccines market. Regulatory complexities, including the varying approval pathways and post-marketing surveillance requirements across different countries, can create significant hurdles for manufacturers. Supply chain issues, particularly in regions with underdeveloped infrastructure, can lead to stockouts and hinder equitable distribution. Competitive pressures from established players and potential new entrants also influence market dynamics. Furthermore, the perceived risk of adverse events, though rare, can sometimes lead to vaccine hesitancy among populations, impacting uptake rates.
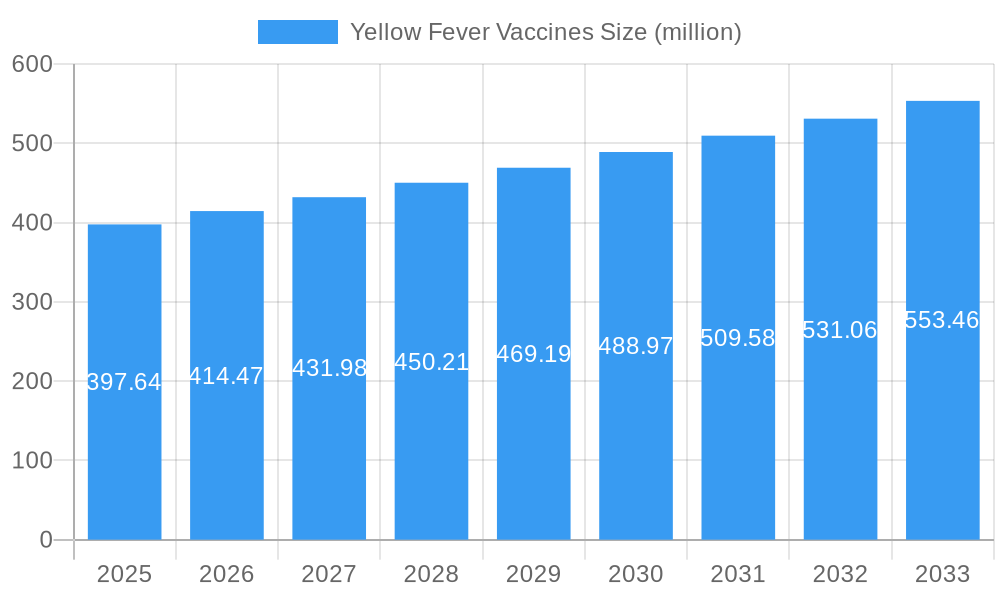
Key Players Shaping the Yellow Fever Vaccines Market
- Sanofi Pasteur
- Bio-Manguinhos
- FSUE Chumakov
- Institut Pasteur de Dakar
Significant Yellow Fever Vaccines Industry Milestones
- 2019: Launch of expanded clinical trials for next-generation Yellow Fever vaccine candidates.
- 2020: Increased global focus on vaccine manufacturing capacity due to pandemic preparedness.
- 2021: WHO recommends the use of fractional doses of Yellow Fever vaccine in outbreak situations to conserve supply.
- 2022: Strategic partnerships formed to improve distribution networks in Sub-Saharan Africa.
- 2023: Advancements reported in the development of thermostable Yellow Fever vaccine formulations.
- 2024: Enhanced surveillance data highlights the ongoing risk of Yellow Fever transmission in specific regions, reinforcing vaccine demand.
Future Outlook for Yellow Fever Vaccines Market
The future outlook for the Yellow Fever Vaccines market remains robust, driven by sustained global health priorities and increasing travel. Strategic opportunities lie in leveraging technological innovations to enhance vaccine stability and accessibility, particularly in remote and resource-limited regions. Expanding market penetration through public-private partnerships and strengthening supply chain resilience will be crucial for meeting the growing demand. The continued commitment of international health organizations to disease prevention and control ensures a positive and growing market trajectory for Yellow Fever vaccines, with projected market value to reach over $1.5 billion by 2033.
Yellow Fever Vaccines Segmentation
-
1. Application
- 1.1. Routine Immunization
- 1.2. Travelers
- 1.3. Other
-
2. Types
- 2.1. Institonal Contributions
- 2.2. Private Purchase
- 2.3. Others
Yellow Fever Vaccines Segmentation By Geography
-
1. North America
- 1.1. United States
- 1.2. Canada
- 1.3. Mexico
-
2. South America
- 2.1. Brazil
- 2.2. Argentina
- 2.3. Rest of South America
-
3. Europe
- 3.1. United Kingdom
- 3.2. Germany
- 3.3. France
- 3.4. Italy
- 3.5. Spain
- 3.6. Russia
- 3.7. Benelux
- 3.8. Nordics
- 3.9. Rest of Europe
-
4. Middle East & Africa
- 4.1. Turkey
- 4.2. Israel
- 4.3. GCC
- 4.4. North Africa
- 4.5. South Africa
- 4.6. Rest of Middle East & Africa
-
5. Asia Pacific
- 5.1. China
- 5.2. India
- 5.3. Japan
- 5.4. South Korea
- 5.5. ASEAN
- 5.6. Oceania
- 5.7. Rest of Asia Pacific
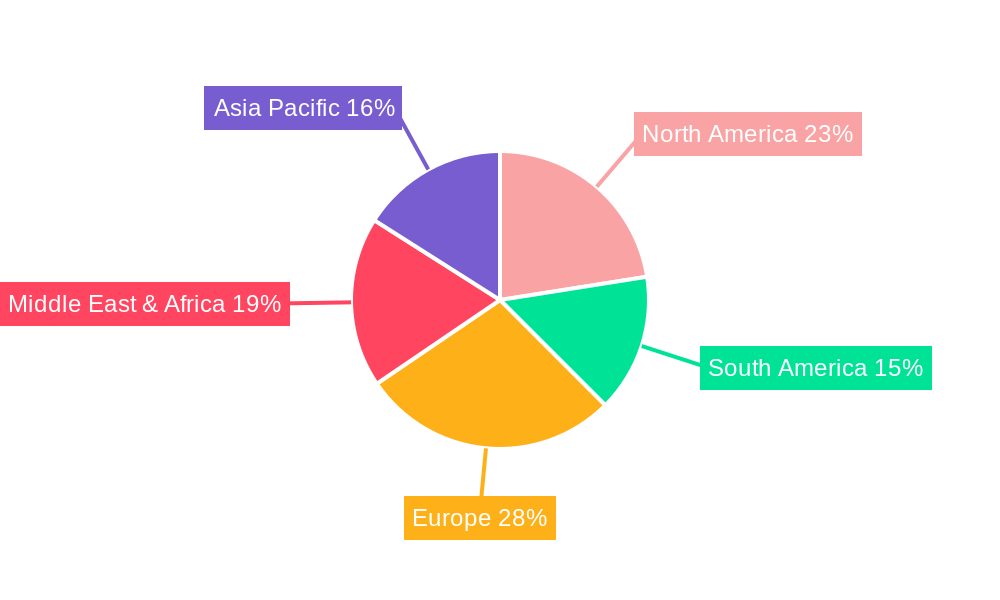
Yellow Fever Vaccines Regional Market Share

Geographic Coverage of Yellow Fever Vaccines
Yellow Fever Vaccines REPORT HIGHLIGHTS
| Aspects | Details |
|---|---|
| Study Period | 2020-2034 |
| Base Year | 2025 |
| Estimated Year | 2026 |
| Forecast Period | 2026-2034 |
| Historical Period | 2020-2025 |
| Growth Rate | CAGR of 8.13% from 2020-2034 |
| Segmentation |
|
Table of Contents
- 1. Introduction
- 1.1. Research Scope
- 1.2. Market Segmentation
- 1.3. Research Methodology
- 1.4. Definitions and Assumptions
- 2. Executive Summary
- 2.1. Introduction
- 3. Market Dynamics
- 3.1. Introduction
- 3.2. Market Drivers
- 3.3. Market Restrains
- 3.4. Market Trends
- 4. Market Factor Analysis
- 4.1. Porters Five Forces
- 4.2. Supply/Value Chain
- 4.3. PESTEL analysis
- 4.4. Market Entropy
- 4.5. Patent/Trademark Analysis
- 5. Global Yellow Fever Vaccines Analysis, Insights and Forecast, 2020-2032
- 5.1. Market Analysis, Insights and Forecast - by Application
- 5.1.1. Routine Immunization
- 5.1.2. Travelers
- 5.1.3. Other
- 5.2. Market Analysis, Insights and Forecast - by Types
- 5.2.1. Institonal Contributions
- 5.2.2. Private Purchase
- 5.2.3. Others
- 5.3. Market Analysis, Insights and Forecast - by Region
- 5.3.1. North America
- 5.3.2. South America
- 5.3.3. Europe
- 5.3.4. Middle East & Africa
- 5.3.5. Asia Pacific
- 5.1. Market Analysis, Insights and Forecast - by Application
- 6. North America Yellow Fever Vaccines Analysis, Insights and Forecast, 2020-2032
- 6.1. Market Analysis, Insights and Forecast - by Application
- 6.1.1. Routine Immunization
- 6.1.2. Travelers
- 6.1.3. Other
- 6.2. Market Analysis, Insights and Forecast - by Types
- 6.2.1. Institonal Contributions
- 6.2.2. Private Purchase
- 6.2.3. Others
- 6.1. Market Analysis, Insights and Forecast - by Application
- 7. South America Yellow Fever Vaccines Analysis, Insights and Forecast, 2020-2032
- 7.1. Market Analysis, Insights and Forecast - by Application
- 7.1.1. Routine Immunization
- 7.1.2. Travelers
- 7.1.3. Other
- 7.2. Market Analysis, Insights and Forecast - by Types
- 7.2.1. Institonal Contributions
- 7.2.2. Private Purchase
- 7.2.3. Others
- 7.1. Market Analysis, Insights and Forecast - by Application
- 8. Europe Yellow Fever Vaccines Analysis, Insights and Forecast, 2020-2032
- 8.1. Market Analysis, Insights and Forecast - by Application
- 8.1.1. Routine Immunization
- 8.1.2. Travelers
- 8.1.3. Other
- 8.2. Market Analysis, Insights and Forecast - by Types
- 8.2.1. Institonal Contributions
- 8.2.2. Private Purchase
- 8.2.3. Others
- 8.1. Market Analysis, Insights and Forecast - by Application
- 9. Middle East & Africa Yellow Fever Vaccines Analysis, Insights and Forecast, 2020-2032
- 9.1. Market Analysis, Insights and Forecast - by Application
- 9.1.1. Routine Immunization
- 9.1.2. Travelers
- 9.1.3. Other
- 9.2. Market Analysis, Insights and Forecast - by Types
- 9.2.1. Institonal Contributions
- 9.2.2. Private Purchase
- 9.2.3. Others
- 9.1. Market Analysis, Insights and Forecast - by Application
- 10. Asia Pacific Yellow Fever Vaccines Analysis, Insights and Forecast, 2020-2032
- 10.1. Market Analysis, Insights and Forecast - by Application
- 10.1.1. Routine Immunization
- 10.1.2. Travelers
- 10.1.3. Other
- 10.2. Market Analysis, Insights and Forecast - by Types
- 10.2.1. Institonal Contributions
- 10.2.2. Private Purchase
- 10.2.3. Others
- 10.1. Market Analysis, Insights and Forecast - by Application
- 11. Competitive Analysis
- 11.1. Global Market Share Analysis 2025
- 11.2. Company Profiles
- 11.2.1 Sanofi Pasteur
- 11.2.1.1. Overview
- 11.2.1.2. Products
- 11.2.1.3. SWOT Analysis
- 11.2.1.4. Recent Developments
- 11.2.1.5. Financials (Based on Availability)
- 11.2.2 Bio-Manguinhos
- 11.2.2.1. Overview
- 11.2.2.2. Products
- 11.2.2.3. SWOT Analysis
- 11.2.2.4. Recent Developments
- 11.2.2.5. Financials (Based on Availability)
- 11.2.3 FSUE Chumakov
- 11.2.3.1. Overview
- 11.2.3.2. Products
- 11.2.3.3. SWOT Analysis
- 11.2.3.4. Recent Developments
- 11.2.3.5. Financials (Based on Availability)
- 11.2.4 Institut Pasteur de Dakar
- 11.2.4.1. Overview
- 11.2.4.2. Products
- 11.2.4.3. SWOT Analysis
- 11.2.4.4. Recent Developments
- 11.2.4.5. Financials (Based on Availability)
- 11.2.1 Sanofi Pasteur
List of Figures
- Figure 1: Global Yellow Fever Vaccines Revenue Breakdown (undefined, %) by Region 2025 & 2033
- Figure 2: Global Yellow Fever Vaccines Volume Breakdown (K, %) by Region 2025 & 2033
- Figure 3: North America Yellow Fever Vaccines Revenue (undefined), by Application 2025 & 2033
- Figure 4: North America Yellow Fever Vaccines Volume (K), by Application 2025 & 2033
- Figure 5: North America Yellow Fever Vaccines Revenue Share (%), by Application 2025 & 2033
- Figure 6: North America Yellow Fever Vaccines Volume Share (%), by Application 2025 & 2033
- Figure 7: North America Yellow Fever Vaccines Revenue (undefined), by Types 2025 & 2033
- Figure 8: North America Yellow Fever Vaccines Volume (K), by Types 2025 & 2033
- Figure 9: North America Yellow Fever Vaccines Revenue Share (%), by Types 2025 & 2033
- Figure 10: North America Yellow Fever Vaccines Volume Share (%), by Types 2025 & 2033
- Figure 11: North America Yellow Fever Vaccines Revenue (undefined), by Country 2025 & 2033
- Figure 12: North America Yellow Fever Vaccines Volume (K), by Country 2025 & 2033
- Figure 13: North America Yellow Fever Vaccines Revenue Share (%), by Country 2025 & 2033
- Figure 14: North America Yellow Fever Vaccines Volume Share (%), by Country 2025 & 2033
- Figure 15: South America Yellow Fever Vaccines Revenue (undefined), by Application 2025 & 2033
- Figure 16: South America Yellow Fever Vaccines Volume (K), by Application 2025 & 2033
- Figure 17: South America Yellow Fever Vaccines Revenue Share (%), by Application 2025 & 2033
- Figure 18: South America Yellow Fever Vaccines Volume Share (%), by Application 2025 & 2033
- Figure 19: South America Yellow Fever Vaccines Revenue (undefined), by Types 2025 & 2033
- Figure 20: South America Yellow Fever Vaccines Volume (K), by Types 2025 & 2033
- Figure 21: South America Yellow Fever Vaccines Revenue Share (%), by Types 2025 & 2033
- Figure 22: South America Yellow Fever Vaccines Volume Share (%), by Types 2025 & 2033
- Figure 23: South America Yellow Fever Vaccines Revenue (undefined), by Country 2025 & 2033
- Figure 24: South America Yellow Fever Vaccines Volume (K), by Country 2025 & 2033
- Figure 25: South America Yellow Fever Vaccines Revenue Share (%), by Country 2025 & 2033
- Figure 26: South America Yellow Fever Vaccines Volume Share (%), by Country 2025 & 2033
- Figure 27: Europe Yellow Fever Vaccines Revenue (undefined), by Application 2025 & 2033
- Figure 28: Europe Yellow Fever Vaccines Volume (K), by Application 2025 & 2033
- Figure 29: Europe Yellow Fever Vaccines Revenue Share (%), by Application 2025 & 2033
- Figure 30: Europe Yellow Fever Vaccines Volume Share (%), by Application 2025 & 2033
- Figure 31: Europe Yellow Fever Vaccines Revenue (undefined), by Types 2025 & 2033
- Figure 32: Europe Yellow Fever Vaccines Volume (K), by Types 2025 & 2033
- Figure 33: Europe Yellow Fever Vaccines Revenue Share (%), by Types 2025 & 2033
- Figure 34: Europe Yellow Fever Vaccines Volume Share (%), by Types 2025 & 2033
- Figure 35: Europe Yellow Fever Vaccines Revenue (undefined), by Country 2025 & 2033
- Figure 36: Europe Yellow Fever Vaccines Volume (K), by Country 2025 & 2033
- Figure 37: Europe Yellow Fever Vaccines Revenue Share (%), by Country 2025 & 2033
- Figure 38: Europe Yellow Fever Vaccines Volume Share (%), by Country 2025 & 2033
- Figure 39: Middle East & Africa Yellow Fever Vaccines Revenue (undefined), by Application 2025 & 2033
- Figure 40: Middle East & Africa Yellow Fever Vaccines Volume (K), by Application 2025 & 2033
- Figure 41: Middle East & Africa Yellow Fever Vaccines Revenue Share (%), by Application 2025 & 2033
- Figure 42: Middle East & Africa Yellow Fever Vaccines Volume Share (%), by Application 2025 & 2033
- Figure 43: Middle East & Africa Yellow Fever Vaccines Revenue (undefined), by Types 2025 & 2033
- Figure 44: Middle East & Africa Yellow Fever Vaccines Volume (K), by Types 2025 & 2033
- Figure 45: Middle East & Africa Yellow Fever Vaccines Revenue Share (%), by Types 2025 & 2033
- Figure 46: Middle East & Africa Yellow Fever Vaccines Volume Share (%), by Types 2025 & 2033
- Figure 47: Middle East & Africa Yellow Fever Vaccines Revenue (undefined), by Country 2025 & 2033
- Figure 48: Middle East & Africa Yellow Fever Vaccines Volume (K), by Country 2025 & 2033
- Figure 49: Middle East & Africa Yellow Fever Vaccines Revenue Share (%), by Country 2025 & 2033
- Figure 50: Middle East & Africa Yellow Fever Vaccines Volume Share (%), by Country 2025 & 2033
- Figure 51: Asia Pacific Yellow Fever Vaccines Revenue (undefined), by Application 2025 & 2033
- Figure 52: Asia Pacific Yellow Fever Vaccines Volume (K), by Application 2025 & 2033
- Figure 53: Asia Pacific Yellow Fever Vaccines Revenue Share (%), by Application 2025 & 2033
- Figure 54: Asia Pacific Yellow Fever Vaccines Volume Share (%), by Application 2025 & 2033
- Figure 55: Asia Pacific Yellow Fever Vaccines Revenue (undefined), by Types 2025 & 2033
- Figure 56: Asia Pacific Yellow Fever Vaccines Volume (K), by Types 2025 & 2033
- Figure 57: Asia Pacific Yellow Fever Vaccines Revenue Share (%), by Types 2025 & 2033
- Figure 58: Asia Pacific Yellow Fever Vaccines Volume Share (%), by Types 2025 & 2033
- Figure 59: Asia Pacific Yellow Fever Vaccines Revenue (undefined), by Country 2025 & 2033
- Figure 60: Asia Pacific Yellow Fever Vaccines Volume (K), by Country 2025 & 2033
- Figure 61: Asia Pacific Yellow Fever Vaccines Revenue Share (%), by Country 2025 & 2033
- Figure 62: Asia Pacific Yellow Fever Vaccines Volume Share (%), by Country 2025 & 2033
List of Tables
- Table 1: Global Yellow Fever Vaccines Revenue undefined Forecast, by Application 2020 & 2033
- Table 2: Global Yellow Fever Vaccines Volume K Forecast, by Application 2020 & 2033
- Table 3: Global Yellow Fever Vaccines Revenue undefined Forecast, by Types 2020 & 2033
- Table 4: Global Yellow Fever Vaccines Volume K Forecast, by Types 2020 & 2033
- Table 5: Global Yellow Fever Vaccines Revenue undefined Forecast, by Region 2020 & 2033
- Table 6: Global Yellow Fever Vaccines Volume K Forecast, by Region 2020 & 2033
- Table 7: Global Yellow Fever Vaccines Revenue undefined Forecast, by Application 2020 & 2033
- Table 8: Global Yellow Fever Vaccines Volume K Forecast, by Application 2020 & 2033
- Table 9: Global Yellow Fever Vaccines Revenue undefined Forecast, by Types 2020 & 2033
- Table 10: Global Yellow Fever Vaccines Volume K Forecast, by Types 2020 & 2033
- Table 11: Global Yellow Fever Vaccines Revenue undefined Forecast, by Country 2020 & 2033
- Table 12: Global Yellow Fever Vaccines Volume K Forecast, by Country 2020 & 2033
- Table 13: United States Yellow Fever Vaccines Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 14: United States Yellow Fever Vaccines Volume (K) Forecast, by Application 2020 & 2033
- Table 15: Canada Yellow Fever Vaccines Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 16: Canada Yellow Fever Vaccines Volume (K) Forecast, by Application 2020 & 2033
- Table 17: Mexico Yellow Fever Vaccines Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 18: Mexico Yellow Fever Vaccines Volume (K) Forecast, by Application 2020 & 2033
- Table 19: Global Yellow Fever Vaccines Revenue undefined Forecast, by Application 2020 & 2033
- Table 20: Global Yellow Fever Vaccines Volume K Forecast, by Application 2020 & 2033
- Table 21: Global Yellow Fever Vaccines Revenue undefined Forecast, by Types 2020 & 2033
- Table 22: Global Yellow Fever Vaccines Volume K Forecast, by Types 2020 & 2033
- Table 23: Global Yellow Fever Vaccines Revenue undefined Forecast, by Country 2020 & 2033
- Table 24: Global Yellow Fever Vaccines Volume K Forecast, by Country 2020 & 2033
- Table 25: Brazil Yellow Fever Vaccines Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 26: Brazil Yellow Fever Vaccines Volume (K) Forecast, by Application 2020 & 2033
- Table 27: Argentina Yellow Fever Vaccines Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 28: Argentina Yellow Fever Vaccines Volume (K) Forecast, by Application 2020 & 2033
- Table 29: Rest of South America Yellow Fever Vaccines Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 30: Rest of South America Yellow Fever Vaccines Volume (K) Forecast, by Application 2020 & 2033
- Table 31: Global Yellow Fever Vaccines Revenue undefined Forecast, by Application 2020 & 2033
- Table 32: Global Yellow Fever Vaccines Volume K Forecast, by Application 2020 & 2033
- Table 33: Global Yellow Fever Vaccines Revenue undefined Forecast, by Types 2020 & 2033
- Table 34: Global Yellow Fever Vaccines Volume K Forecast, by Types 2020 & 2033
- Table 35: Global Yellow Fever Vaccines Revenue undefined Forecast, by Country 2020 & 2033
- Table 36: Global Yellow Fever Vaccines Volume K Forecast, by Country 2020 & 2033
- Table 37: United Kingdom Yellow Fever Vaccines Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 38: United Kingdom Yellow Fever Vaccines Volume (K) Forecast, by Application 2020 & 2033
- Table 39: Germany Yellow Fever Vaccines Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 40: Germany Yellow Fever Vaccines Volume (K) Forecast, by Application 2020 & 2033
- Table 41: France Yellow Fever Vaccines Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 42: France Yellow Fever Vaccines Volume (K) Forecast, by Application 2020 & 2033
- Table 43: Italy Yellow Fever Vaccines Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 44: Italy Yellow Fever Vaccines Volume (K) Forecast, by Application 2020 & 2033
- Table 45: Spain Yellow Fever Vaccines Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 46: Spain Yellow Fever Vaccines Volume (K) Forecast, by Application 2020 & 2033
- Table 47: Russia Yellow Fever Vaccines Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 48: Russia Yellow Fever Vaccines Volume (K) Forecast, by Application 2020 & 2033
- Table 49: Benelux Yellow Fever Vaccines Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 50: Benelux Yellow Fever Vaccines Volume (K) Forecast, by Application 2020 & 2033
- Table 51: Nordics Yellow Fever Vaccines Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 52: Nordics Yellow Fever Vaccines Volume (K) Forecast, by Application 2020 & 2033
- Table 53: Rest of Europe Yellow Fever Vaccines Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 54: Rest of Europe Yellow Fever Vaccines Volume (K) Forecast, by Application 2020 & 2033
- Table 55: Global Yellow Fever Vaccines Revenue undefined Forecast, by Application 2020 & 2033
- Table 56: Global Yellow Fever Vaccines Volume K Forecast, by Application 2020 & 2033
- Table 57: Global Yellow Fever Vaccines Revenue undefined Forecast, by Types 2020 & 2033
- Table 58: Global Yellow Fever Vaccines Volume K Forecast, by Types 2020 & 2033
- Table 59: Global Yellow Fever Vaccines Revenue undefined Forecast, by Country 2020 & 2033
- Table 60: Global Yellow Fever Vaccines Volume K Forecast, by Country 2020 & 2033
- Table 61: Turkey Yellow Fever Vaccines Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 62: Turkey Yellow Fever Vaccines Volume (K) Forecast, by Application 2020 & 2033
- Table 63: Israel Yellow Fever Vaccines Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 64: Israel Yellow Fever Vaccines Volume (K) Forecast, by Application 2020 & 2033
- Table 65: GCC Yellow Fever Vaccines Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 66: GCC Yellow Fever Vaccines Volume (K) Forecast, by Application 2020 & 2033
- Table 67: North Africa Yellow Fever Vaccines Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 68: North Africa Yellow Fever Vaccines Volume (K) Forecast, by Application 2020 & 2033
- Table 69: South Africa Yellow Fever Vaccines Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 70: South Africa Yellow Fever Vaccines Volume (K) Forecast, by Application 2020 & 2033
- Table 71: Rest of Middle East & Africa Yellow Fever Vaccines Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 72: Rest of Middle East & Africa Yellow Fever Vaccines Volume (K) Forecast, by Application 2020 & 2033
- Table 73: Global Yellow Fever Vaccines Revenue undefined Forecast, by Application 2020 & 2033
- Table 74: Global Yellow Fever Vaccines Volume K Forecast, by Application 2020 & 2033
- Table 75: Global Yellow Fever Vaccines Revenue undefined Forecast, by Types 2020 & 2033
- Table 76: Global Yellow Fever Vaccines Volume K Forecast, by Types 2020 & 2033
- Table 77: Global Yellow Fever Vaccines Revenue undefined Forecast, by Country 2020 & 2033
- Table 78: Global Yellow Fever Vaccines Volume K Forecast, by Country 2020 & 2033
- Table 79: China Yellow Fever Vaccines Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 80: China Yellow Fever Vaccines Volume (K) Forecast, by Application 2020 & 2033
- Table 81: India Yellow Fever Vaccines Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 82: India Yellow Fever Vaccines Volume (K) Forecast, by Application 2020 & 2033
- Table 83: Japan Yellow Fever Vaccines Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 84: Japan Yellow Fever Vaccines Volume (K) Forecast, by Application 2020 & 2033
- Table 85: South Korea Yellow Fever Vaccines Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 86: South Korea Yellow Fever Vaccines Volume (K) Forecast, by Application 2020 & 2033
- Table 87: ASEAN Yellow Fever Vaccines Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 88: ASEAN Yellow Fever Vaccines Volume (K) Forecast, by Application 2020 & 2033
- Table 89: Oceania Yellow Fever Vaccines Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 90: Oceania Yellow Fever Vaccines Volume (K) Forecast, by Application 2020 & 2033
- Table 91: Rest of Asia Pacific Yellow Fever Vaccines Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 92: Rest of Asia Pacific Yellow Fever Vaccines Volume (K) Forecast, by Application 2020 & 2033
Frequently Asked Questions
1. What is the projected Compound Annual Growth Rate (CAGR) of the Yellow Fever Vaccines?
The projected CAGR is approximately 8.13%.
2. Which companies are prominent players in the Yellow Fever Vaccines?
Key companies in the market include Sanofi Pasteur, Bio-Manguinhos, FSUE Chumakov, Institut Pasteur de Dakar.
3. What are the main segments of the Yellow Fever Vaccines?
The market segments include Application, Types.
4. Can you provide details about the market size?
The market size is estimated to be USD XXX N/A as of 2022.
5. What are some drivers contributing to market growth?
N/A
6. What are the notable trends driving market growth?
N/A
7. Are there any restraints impacting market growth?
N/A
8. Can you provide examples of recent developments in the market?
N/A
9. What pricing options are available for accessing the report?
Pricing options include single-user, multi-user, and enterprise licenses priced at USD 4000.00, USD 6000.00, and USD 8000.00 respectively.
10. Is the market size provided in terms of value or volume?
The market size is provided in terms of value, measured in N/A and volume, measured in K.
11. Are there any specific market keywords associated with the report?
Yes, the market keyword associated with the report is "Yellow Fever Vaccines," which aids in identifying and referencing the specific market segment covered.
12. How do I determine which pricing option suits my needs best?
The pricing options vary based on user requirements and access needs. Individual users may opt for single-user licenses, while businesses requiring broader access may choose multi-user or enterprise licenses for cost-effective access to the report.
13. Are there any additional resources or data provided in the Yellow Fever Vaccines report?
While the report offers comprehensive insights, it's advisable to review the specific contents or supplementary materials provided to ascertain if additional resources or data are available.
14. How can I stay updated on further developments or reports in the Yellow Fever Vaccines?
To stay informed about further developments, trends, and reports in the Yellow Fever Vaccines, consider subscribing to industry newsletters, following relevant companies and organizations, or regularly checking reputable industry news sources and publications.
Methodology
Step 1 - Identification of Relevant Samples Size from Population Database



Step 2 - Approaches for Defining Global Market Size (Value, Volume* & Price*)

Note*: In applicable scenarios
Step 3 - Data Sources
Primary Research
- Web Analytics
- Survey Reports
- Research Institute
- Latest Research Reports
- Opinion Leaders
Secondary Research
- Annual Reports
- White Paper
- Latest Press Release
- Industry Association
- Paid Database
- Investor Presentations

Step 4 - Data Triangulation
Involves using different sources of information in order to increase the validity of a study
These sources are likely to be stakeholders in a program - participants, other researchers, program staff, other community members, and so on.
Then we put all data in single framework & apply various statistical tools to find out the dynamic on the market.
During the analysis stage, feedback from the stakeholder groups would be compared to determine areas of agreement as well as areas of divergence


