Key Insights
The Autologous Fat Grafting Device market is poised for significant expansion, projected to reach $10.78 billion in 2025. This robust growth is underpinned by a CAGR of 16.04%, indicating a dynamic and rapidly evolving industry. The primary drivers fueling this surge include the increasing demand for minimally invasive aesthetic procedures, the growing awareness and acceptance of autologous fat grafting as a safe and effective cosmetic and reconstructive solution, and advancements in device technology that enhance precision, efficiency, and patient outcomes. As patient preferences shift towards natural-looking results and quicker recovery times, devices that facilitate these aspects are gaining considerable traction. Furthermore, the expanding applications of fat grafting beyond aesthetics, such as in reconstructive surgery and chronic wound management, are contributing to market diversification and overall growth. This trend is amplified by the increasing disposable incomes and a greater emphasis on personal appearance across various demographics.
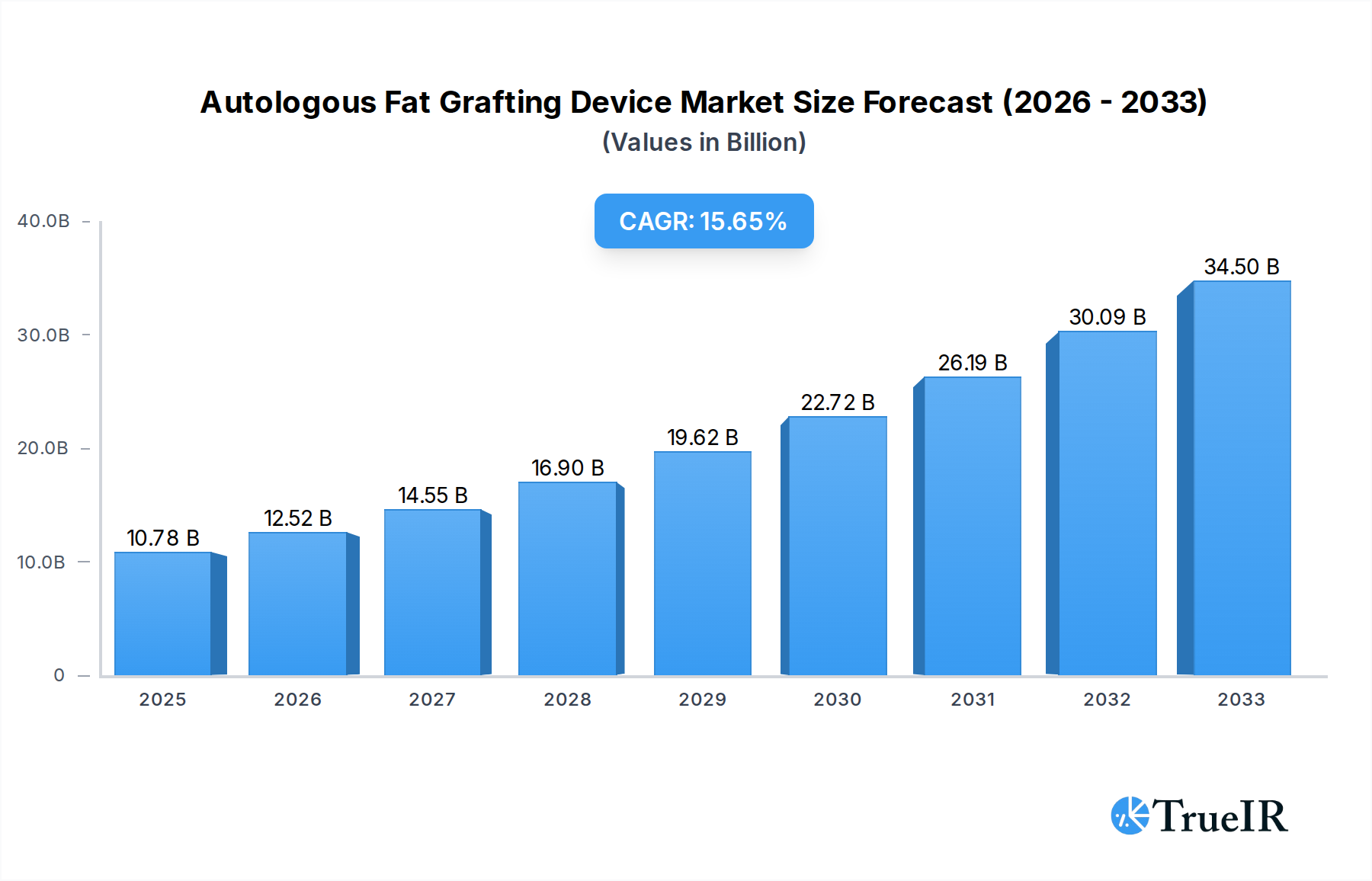
Autologous Fat Grafting Device Market Size (In Billion)

The market's trajectory is further shaped by key trends such as the development of integrated systems combining fat harvesting, processing, and transplantation, offering a streamlined workflow for practitioners. Innovations in laser-assisted and ultrasound-assisted fat grafting devices are improving fat cell viability and graft success rates, thereby enhancing patient satisfaction and clinical efficacy. While the market exhibits strong growth potential, certain restraints need to be acknowledged. These include the high cost of advanced fat grafting devices, the need for specialized training for healthcare professionals, and regulatory hurdles in certain regions that may impact market access and adoption. However, the persistent demand for cosmetic enhancement and reconstructive procedures, coupled with ongoing technological innovations, is expected to outweigh these challenges, paving the way for sustained and substantial market growth in the coming years.
Autologous Fat Grafting Device Company Market Share

Autologous Fat Grafting Device Market Structure & Competitive Landscape
The global Autologous Fat Grafting Device market is characterized by a moderate to high level of concentration, with key players investing heavily in research and development to drive innovation. Several billion dollars in M&A activity have been observed as companies seek to expand their product portfolios and market reach. Innovation drivers include advancements in minimally invasive techniques, improved patient outcomes, and the increasing demand for aesthetic and reconstructive procedures. Regulatory impacts are significant, with stringent approvals required for new devices, influencing market entry and product development timelines. Product substitutes, such as synthetic fillers and advanced liposuction technologies, pose a competitive threat, necessitating continuous product differentiation by autologous fat grafting device manufacturers.
End-user segmentation primarily includes hospitals and clinics, each with distinct purchasing patterns and integration needs for these sophisticated devices. Hospitals, with their comprehensive surgical departments, tend to adopt a wider range of technologies, while specialized clinics often focus on specific therapeutic areas. Mergers and acquisitions (M&A) trends are a significant feature, with larger entities acquiring innovative startups to gain access to novel technologies and expand their market share. For instance, a series of billion-dollar acquisitions in the last five years have reshaped the competitive landscape, consolidating market power and fostering a more integrated approach to fat grafting solutions. The market is dynamic, with ongoing patent filings and a consistent stream of new product introductions aimed at enhancing efficacy, safety, and patient comfort.
Autologous Fat Grafting Device Market Trends & Opportunities
The global Autologous Fat Grafting Device market is poised for substantial expansion, projected to witness a Compound Annual Growth Rate (CAGR) of over 8.5% from 2025 to 2033, reaching an estimated market size exceeding $3 billion by the end of the forecast period. This robust growth is fueled by a confluence of evolving industry developments, technological sophistication, and shifting consumer preferences towards natural and minimally invasive aesthetic and reconstructive procedures. The increasing prevalence of cosmetic enhancement procedures globally, coupled with a growing awareness of autologous fat grafting as a safe and effective alternative to synthetic fillers and implants, is a primary demand catalyst. Furthermore, advancements in surgical techniques and device technologies are improving the viability and integration of grafted fat, leading to enhanced patient satisfaction and driving adoption across various medical specialties.
Technological shifts are at the forefront of market evolution. Innovations in liposuction and fat processing technologies are continuously enhancing the quality and yield of harvested adipose tissue, making the grafting process more efficient and predictable. For example, the integration of laser-based and ultrasound-assisted technologies with traditional power-assisted liposuction methods is providing practitioners with more precise control over fat harvesting and preparation, leading to superior aesthetic and functional outcomes. This technological convergence is creating new market opportunities for device manufacturers who can offer integrated or complementary solutions. Consumer preferences are increasingly leaning towards natural rejuvenation techniques, driven by a desire for long-lasting results and a reduced risk of allergic reactions or complications associated with synthetic materials. This trend directly benefits the autologous fat grafting market.
The competitive dynamics are intensifying, with established medical device companies and emerging players vying for market dominance. Strategic partnerships, product launches, and the pursuit of intellectual property are key competitive strategies. The market penetration rates are steadily increasing in developed economies, while emerging markets present significant untapped potential due to growing disposable incomes and a rising demand for aesthetic treatments. Industry developments such as the refinement of stem cell enrichment techniques for fat grafts, the development of automated fat processing systems, and the exploration of novel applications beyond cosmetic surgery, such as tissue regeneration and wound healing, are further expanding the market's horizons. The market is also being shaped by increasing investments in clinical trials and research to validate the long-term efficacy and safety of autologous fat grafting, thereby building greater confidence among healthcare providers and patients. The projected market size of over $3 billion reflects the sustained demand and the significant potential for further growth driven by these multifaceted trends and opportunities.
Dominant Markets & Segments in Autologous Fat Grafting Device
The global Autologous Fat Grafting Device market exhibits strong dominance in North America and Europe, with Asia-Pacific emerging as a rapidly growing region. Within these dominant regions, the Clinic segment is currently the largest and most influential application for autologous fat grafting devices, driven by the proliferation of specialized aesthetic and reconstructive surgery clinics. These facilities often invest in advanced, user-friendly devices that streamline procedures and enhance patient throughput. The Hospital segment, while significant, is characterized by broader adoption across various surgical disciplines and a focus on complex reconstructive cases, alongside aesthetic procedures. Key growth drivers in the clinic segment include the increasing demand for elective cosmetic procedures, a higher propensity for adopting new technologies, and a more direct patient-provider relationship fostering trust in fat grafting.
The Type segmentation reveals that Power Assisted autologous fat grafting devices currently hold the largest market share, owing to their established efficacy, versatility, and widespread familiarity among surgeons. However, Ultrasound Assisted and Laser-Based technologies are gaining considerable traction due to their ability to facilitate precise fat harvesting with minimal trauma, leading to improved fat graft viability and reduced donor site morbidity. For instance, ultrasound assistance provides surgeons with enhanced visualization during liposuction, allowing for more targeted fat collection and potentially better-preserved adipocytes. Laser-based systems offer precise energy delivery, which can aid in emulsifying fat for easier extraction and can also potentially stimulate tissue regeneration.
In terms of regional dominance, the United States leads the market, supported by a robust healthcare infrastructure, high disposable incomes, and a culture that embraces cosmetic enhancements. Stringent regulatory pathways for device approval, while a barrier to entry, also ensure the availability of high-quality, safe, and effective devices. In Europe, countries like Germany, France, and the UK contribute significantly to market growth, driven by advanced medical tourism and a strong emphasis on reconstructive surgery. The policies in these regions often encourage innovation and the adoption of advanced medical technologies.
The Asia-Pacific region, particularly China, India, and South Korea, is witnessing rapid expansion due to increasing disposable incomes, a growing middle class, and a rising awareness of aesthetic procedures. Government initiatives to promote medical tourism and advancements in healthcare infrastructure are further fueling growth. Key growth drivers in this segment include the growing patient pool seeking affordable yet effective cosmetic and reconstructive solutions, and the increasing adoption of technologically advanced fat grafting devices by a burgeoning number of practitioners. The combined influence of these dominant regions and segments, coupled with strategic investments in technology and market penetration, is shaping the trajectory of the global autologous fat grafting device market, with an estimated market size projected to surpass $3 billion.
Autologous Fat Grafting Device Product Analysis
Product innovation in Autologous Fat Grafting Devices centers on enhancing fat cell viability, improving harvesting efficiency, and simplifying the overall procedure for both practitioners and patients. Devices are increasingly incorporating advanced features such as precise suction control, integrated filtration systems, and ergonomic designs to minimize tissue trauma. Competitive advantages lie in patented technologies that optimize fat processing, leading to higher survival rates of grafted fat and more predictable, natural-looking results. Applications extend beyond cosmetic augmentation to reconstructive surgery, addressing deformities and defects. The focus is on delivering minimally invasive solutions with shorter recovery times.
Key Drivers, Barriers & Challenges in Autologous Fat Grafting Device
Key Drivers: The autologous fat grafting device market is primarily propelled by the escalating demand for minimally invasive cosmetic and reconstructive procedures, a growing preference for natural augmentation techniques, and continuous technological advancements leading to improved device efficacy and patient outcomes. Economic factors, including increased disposable income and accessibility to financing for aesthetic treatments, also play a crucial role. Furthermore, supportive regulatory frameworks in key markets that facilitate the approval of innovative devices contribute to market expansion.
Barriers & Challenges: Despite its growth potential, the market faces challenges such as stringent regulatory hurdles and lengthy approval processes for new devices, which can significantly impact time-to-market and R&D costs. Supply chain disruptions, particularly for specialized components, can affect manufacturing and availability. Competitive pressures from alternative aesthetic treatments, including synthetic fillers and other regenerative medicine approaches, necessitate ongoing innovation and differentiation. The cost of advanced devices can also be a barrier for smaller clinics.
Growth Drivers in the Autologous Fat Grafting Device Market
The autologous fat grafting device market is experiencing robust growth driven by several key factors. Technological advancements, including the development of refined liposuction techniques and sophisticated fat processing systems, are enhancing procedural efficiency and patient outcomes, leading to increased adoption. The growing consumer preference for natural and organic aesthetic solutions over synthetic alternatives is a significant economic driver, boosting demand for autologous fat grafting. Supportive regulatory policies in major markets, which streamline the approval of innovative medical devices, also foster market expansion. Furthermore, increasing healthcare expenditure and rising disposable incomes globally are making these procedures more accessible, contributing to market penetration.
Challenges Impacting Autologous Fat Grafting Device Growth
Several challenges can impact the growth of the autologous fat grafting device market. Regulatory complexities, including the lengthy and rigorous approval processes for new medical devices, can hinder the introduction of innovative technologies and increase development costs. Supply chain issues, such as the availability of specialized components and manufacturing capacity, can lead to production delays and affect device accessibility. Intense competitive pressures from alternative aesthetic treatments, such as dermal fillers and minimally invasive surgical procedures, necessitate continuous product differentiation and market education. The high initial cost of advanced fat grafting systems can also pose a barrier to adoption for smaller clinics and in emerging markets.
Key Players Shaping the Autologous Fat Grafting Device Market
- Solta Medical
- Cynosure Inc.
- Alma Lasers
- AbbVie
- Cutera
- Erchonia
- Bimini Health Tech
- Candela Medical
- Wells Johnson Co.
- Genesis Biosystems
- Human Med
- Moeller Medical
- MicroAire Surgical
- Smart Lipo
- MTF Biologics
Significant Autologous Fat Grafting Device Industry Milestones
- 2019: Introduction of advanced micro-cannula technology for enhanced fat harvesting precision.
- 2020: Increased adoption of ultrasound-assisted liposuction devices for improved fat yield and quality.
- 2021: Launch of integrated systems combining fat harvesting, processing, and injection capabilities.
- 2022: Emergence of stem cell-enriched fat grafting techniques gaining traction in research and clinical trials.
- 2023: Significant investment in AI-driven analytics for optimizing fat grafting outcomes.
- 2024: Expansion of device applications into reconstructive surgery and tissue regeneration.
Future Outlook for Autologous Fat Grafting Device Market
The future outlook for the Autologous Fat Grafting Device market is exceptionally bright, projected for continued substantial growth driven by ongoing technological innovation and escalating patient demand for natural, long-lasting aesthetic and reconstructive solutions. Strategic opportunities lie in the development of more automated and AI-integrated systems to further enhance precision and ease of use, as well as the expansion of applications into broader regenerative medicine fields. Emerging markets present significant untapped potential, offering avenues for increased market penetration. The market is poised to witness advancements in fat processing, leading to even higher graft survival rates, further solidifying its position as a cornerstone of modern aesthetic and reconstructive surgery.
Autologous Fat Grafting Device Segmentation
-
1. Application
- 1.1. Hospital
- 1.2. Clinic
-
2. Type
- 2.1. Laser-Based
- 2.2. Ultrasound Assisted
- 2.3. Power Assisted
Autologous Fat Grafting Device Segmentation By Geography
-
1. North America
- 1.1. United States
- 1.2. Canada
- 1.3. Mexico
-
2. South America
- 2.1. Brazil
- 2.2. Argentina
- 2.3. Rest of South America
-
3. Europe
- 3.1. United Kingdom
- 3.2. Germany
- 3.3. France
- 3.4. Italy
- 3.5. Spain
- 3.6. Russia
- 3.7. Benelux
- 3.8. Nordics
- 3.9. Rest of Europe
-
4. Middle East & Africa
- 4.1. Turkey
- 4.2. Israel
- 4.3. GCC
- 4.4. North Africa
- 4.5. South Africa
- 4.6. Rest of Middle East & Africa
-
5. Asia Pacific
- 5.1. China
- 5.2. India
- 5.3. Japan
- 5.4. South Korea
- 5.5. ASEAN
- 5.6. Oceania
- 5.7. Rest of Asia Pacific
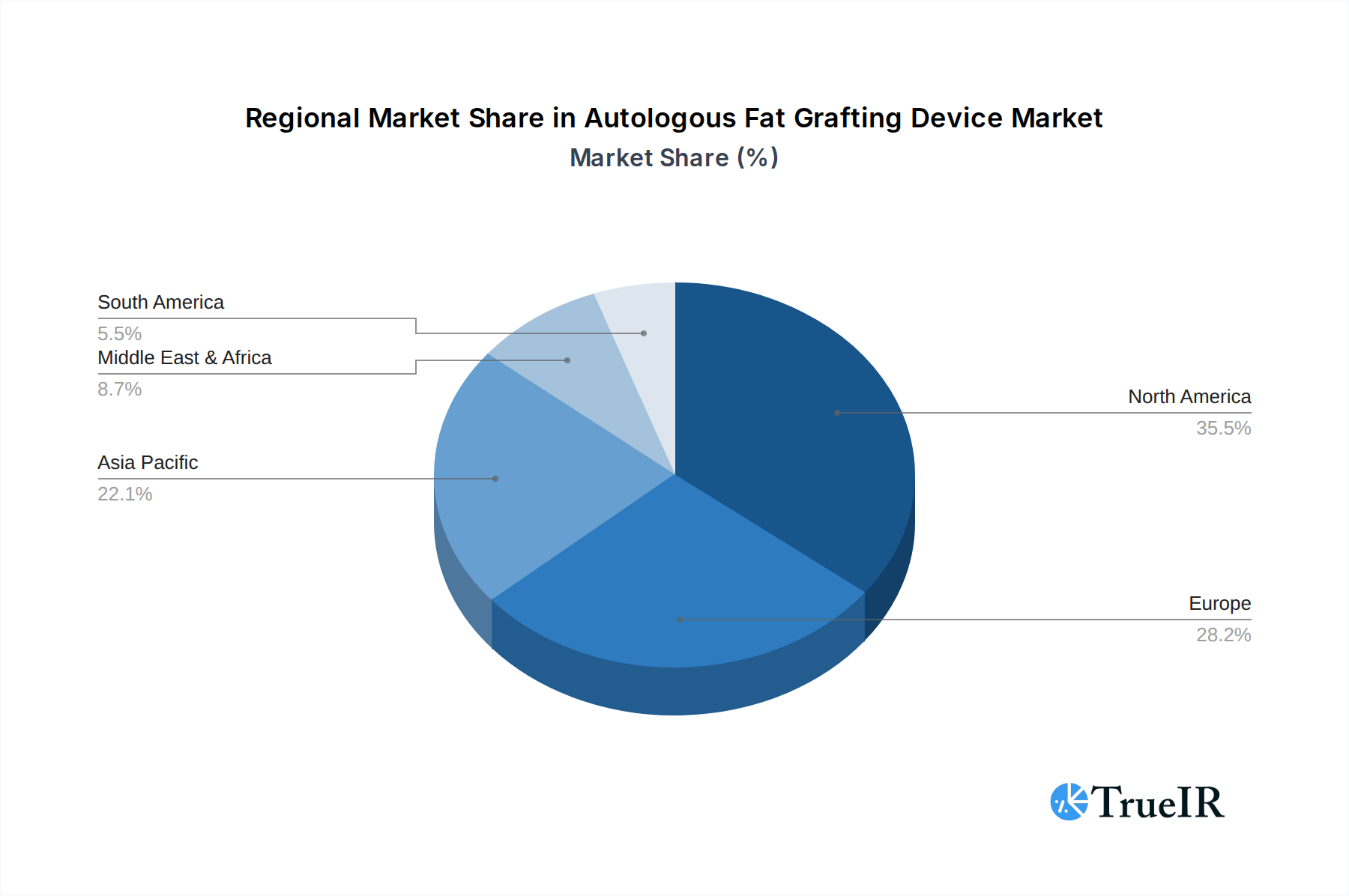
Autologous Fat Grafting Device Regional Market Share

Geographic Coverage of Autologous Fat Grafting Device
Autologous Fat Grafting Device REPORT HIGHLIGHTS
| Aspects | Details |
|---|---|
| Study Period | 2020-2034 |
| Base Year | 2025 |
| Estimated Year | 2026 |
| Forecast Period | 2026-2034 |
| Historical Period | 2020-2025 |
| Growth Rate | CAGR of 16.04% from 2020-2034 |
| Segmentation |
|
Table of Contents
- 1. Introduction
- 1.1. Research Scope
- 1.2. Market Segmentation
- 1.3. Research Methodology
- 1.4. Definitions and Assumptions
- 2. Executive Summary
- 2.1. Introduction
- 3. Market Dynamics
- 3.1. Introduction
- 3.2. Market Drivers
- 3.3. Market Restrains
- 3.4. Market Trends
- 4. Market Factor Analysis
- 4.1. Porters Five Forces
- 4.2. Supply/Value Chain
- 4.3. PESTEL analysis
- 4.4. Market Entropy
- 4.5. Patent/Trademark Analysis
- 5. Global Autologous Fat Grafting Device Analysis, Insights and Forecast, 2020-2032
- 5.1. Market Analysis, Insights and Forecast - by Application
- 5.1.1. Hospital
- 5.1.2. Clinic
- 5.2. Market Analysis, Insights and Forecast - by Type
- 5.2.1. Laser-Based
- 5.2.2. Ultrasound Assisted
- 5.2.3. Power Assisted
- 5.3. Market Analysis, Insights and Forecast - by Region
- 5.3.1. North America
- 5.3.2. South America
- 5.3.3. Europe
- 5.3.4. Middle East & Africa
- 5.3.5. Asia Pacific
- 5.1. Market Analysis, Insights and Forecast - by Application
- 6. North America Autologous Fat Grafting Device Analysis, Insights and Forecast, 2020-2032
- 6.1. Market Analysis, Insights and Forecast - by Application
- 6.1.1. Hospital
- 6.1.2. Clinic
- 6.2. Market Analysis, Insights and Forecast - by Type
- 6.2.1. Laser-Based
- 6.2.2. Ultrasound Assisted
- 6.2.3. Power Assisted
- 6.1. Market Analysis, Insights and Forecast - by Application
- 7. South America Autologous Fat Grafting Device Analysis, Insights and Forecast, 2020-2032
- 7.1. Market Analysis, Insights and Forecast - by Application
- 7.1.1. Hospital
- 7.1.2. Clinic
- 7.2. Market Analysis, Insights and Forecast - by Type
- 7.2.1. Laser-Based
- 7.2.2. Ultrasound Assisted
- 7.2.3. Power Assisted
- 7.1. Market Analysis, Insights and Forecast - by Application
- 8. Europe Autologous Fat Grafting Device Analysis, Insights and Forecast, 2020-2032
- 8.1. Market Analysis, Insights and Forecast - by Application
- 8.1.1. Hospital
- 8.1.2. Clinic
- 8.2. Market Analysis, Insights and Forecast - by Type
- 8.2.1. Laser-Based
- 8.2.2. Ultrasound Assisted
- 8.2.3. Power Assisted
- 8.1. Market Analysis, Insights and Forecast - by Application
- 9. Middle East & Africa Autologous Fat Grafting Device Analysis, Insights and Forecast, 2020-2032
- 9.1. Market Analysis, Insights and Forecast - by Application
- 9.1.1. Hospital
- 9.1.2. Clinic
- 9.2. Market Analysis, Insights and Forecast - by Type
- 9.2.1. Laser-Based
- 9.2.2. Ultrasound Assisted
- 9.2.3. Power Assisted
- 9.1. Market Analysis, Insights and Forecast - by Application
- 10. Asia Pacific Autologous Fat Grafting Device Analysis, Insights and Forecast, 2020-2032
- 10.1. Market Analysis, Insights and Forecast - by Application
- 10.1.1. Hospital
- 10.1.2. Clinic
- 10.2. Market Analysis, Insights and Forecast - by Type
- 10.2.1. Laser-Based
- 10.2.2. Ultrasound Assisted
- 10.2.3. Power Assisted
- 10.1. Market Analysis, Insights and Forecast - by Application
- 11. Competitive Analysis
- 11.1. Global Market Share Analysis 2025
- 11.2. Company Profiles
- 11.2.1 Solta Medical
- 11.2.1.1. Overview
- 11.2.1.2. Products
- 11.2.1.3. SWOT Analysis
- 11.2.1.4. Recent Developments
- 11.2.1.5. Financials (Based on Availability)
- 11.2.2 Cynosure Inc.
- 11.2.2.1. Overview
- 11.2.2.2. Products
- 11.2.2.3. SWOT Analysis
- 11.2.2.4. Recent Developments
- 11.2.2.5. Financials (Based on Availability)
- 11.2.3 Alma Lasers
- 11.2.3.1. Overview
- 11.2.3.2. Products
- 11.2.3.3. SWOT Analysis
- 11.2.3.4. Recent Developments
- 11.2.3.5. Financials (Based on Availability)
- 11.2.4 AbbVie
- 11.2.4.1. Overview
- 11.2.4.2. Products
- 11.2.4.3. SWOT Analysis
- 11.2.4.4. Recent Developments
- 11.2.4.5. Financials (Based on Availability)
- 11.2.5 Cutera
- 11.2.5.1. Overview
- 11.2.5.2. Products
- 11.2.5.3. SWOT Analysis
- 11.2.5.4. Recent Developments
- 11.2.5.5. Financials (Based on Availability)
- 11.2.6 Erchonia
- 11.2.6.1. Overview
- 11.2.6.2. Products
- 11.2.6.3. SWOT Analysis
- 11.2.6.4. Recent Developments
- 11.2.6.5. Financials (Based on Availability)
- 11.2.7 Bimini Health Tech
- 11.2.7.1. Overview
- 11.2.7.2. Products
- 11.2.7.3. SWOT Analysis
- 11.2.7.4. Recent Developments
- 11.2.7.5. Financials (Based on Availability)
- 11.2.8 Candela Medical
- 11.2.8.1. Overview
- 11.2.8.2. Products
- 11.2.8.3. SWOT Analysis
- 11.2.8.4. Recent Developments
- 11.2.8.5. Financials (Based on Availability)
- 11.2.9 Wells Johnson Co.
- 11.2.9.1. Overview
- 11.2.9.2. Products
- 11.2.9.3. SWOT Analysis
- 11.2.9.4. Recent Developments
- 11.2.9.5. Financials (Based on Availability)
- 11.2.10 Genesis Biosystems
- 11.2.10.1. Overview
- 11.2.10.2. Products
- 11.2.10.3. SWOT Analysis
- 11.2.10.4. Recent Developments
- 11.2.10.5. Financials (Based on Availability)
- 11.2.11 Human Med
- 11.2.11.1. Overview
- 11.2.11.2. Products
- 11.2.11.3. SWOT Analysis
- 11.2.11.4. Recent Developments
- 11.2.11.5. Financials (Based on Availability)
- 11.2.12 Moeller Medical
- 11.2.12.1. Overview
- 11.2.12.2. Products
- 11.2.12.3. SWOT Analysis
- 11.2.12.4. Recent Developments
- 11.2.12.5. Financials (Based on Availability)
- 11.2.13 MicroAire Surgical
- 11.2.13.1. Overview
- 11.2.13.2. Products
- 11.2.13.3. SWOT Analysis
- 11.2.13.4. Recent Developments
- 11.2.13.5. Financials (Based on Availability)
- 11.2.14 Smart Lipo
- 11.2.14.1. Overview
- 11.2.14.2. Products
- 11.2.14.3. SWOT Analysis
- 11.2.14.4. Recent Developments
- 11.2.14.5. Financials (Based on Availability)
- 11.2.15 MTF Biologics
- 11.2.15.1. Overview
- 11.2.15.2. Products
- 11.2.15.3. SWOT Analysis
- 11.2.15.4. Recent Developments
- 11.2.15.5. Financials (Based on Availability)
- 11.2.1 Solta Medical
List of Figures
- Figure 1: Global Autologous Fat Grafting Device Revenue Breakdown (undefined, %) by Region 2025 & 2033
- Figure 2: North America Autologous Fat Grafting Device Revenue (undefined), by Application 2025 & 2033
- Figure 3: North America Autologous Fat Grafting Device Revenue Share (%), by Application 2025 & 2033
- Figure 4: North America Autologous Fat Grafting Device Revenue (undefined), by Type 2025 & 2033
- Figure 5: North America Autologous Fat Grafting Device Revenue Share (%), by Type 2025 & 2033
- Figure 6: North America Autologous Fat Grafting Device Revenue (undefined), by Country 2025 & 2033
- Figure 7: North America Autologous Fat Grafting Device Revenue Share (%), by Country 2025 & 2033
- Figure 8: South America Autologous Fat Grafting Device Revenue (undefined), by Application 2025 & 2033
- Figure 9: South America Autologous Fat Grafting Device Revenue Share (%), by Application 2025 & 2033
- Figure 10: South America Autologous Fat Grafting Device Revenue (undefined), by Type 2025 & 2033
- Figure 11: South America Autologous Fat Grafting Device Revenue Share (%), by Type 2025 & 2033
- Figure 12: South America Autologous Fat Grafting Device Revenue (undefined), by Country 2025 & 2033
- Figure 13: South America Autologous Fat Grafting Device Revenue Share (%), by Country 2025 & 2033
- Figure 14: Europe Autologous Fat Grafting Device Revenue (undefined), by Application 2025 & 2033
- Figure 15: Europe Autologous Fat Grafting Device Revenue Share (%), by Application 2025 & 2033
- Figure 16: Europe Autologous Fat Grafting Device Revenue (undefined), by Type 2025 & 2033
- Figure 17: Europe Autologous Fat Grafting Device Revenue Share (%), by Type 2025 & 2033
- Figure 18: Europe Autologous Fat Grafting Device Revenue (undefined), by Country 2025 & 2033
- Figure 19: Europe Autologous Fat Grafting Device Revenue Share (%), by Country 2025 & 2033
- Figure 20: Middle East & Africa Autologous Fat Grafting Device Revenue (undefined), by Application 2025 & 2033
- Figure 21: Middle East & Africa Autologous Fat Grafting Device Revenue Share (%), by Application 2025 & 2033
- Figure 22: Middle East & Africa Autologous Fat Grafting Device Revenue (undefined), by Type 2025 & 2033
- Figure 23: Middle East & Africa Autologous Fat Grafting Device Revenue Share (%), by Type 2025 & 2033
- Figure 24: Middle East & Africa Autologous Fat Grafting Device Revenue (undefined), by Country 2025 & 2033
- Figure 25: Middle East & Africa Autologous Fat Grafting Device Revenue Share (%), by Country 2025 & 2033
- Figure 26: Asia Pacific Autologous Fat Grafting Device Revenue (undefined), by Application 2025 & 2033
- Figure 27: Asia Pacific Autologous Fat Grafting Device Revenue Share (%), by Application 2025 & 2033
- Figure 28: Asia Pacific Autologous Fat Grafting Device Revenue (undefined), by Type 2025 & 2033
- Figure 29: Asia Pacific Autologous Fat Grafting Device Revenue Share (%), by Type 2025 & 2033
- Figure 30: Asia Pacific Autologous Fat Grafting Device Revenue (undefined), by Country 2025 & 2033
- Figure 31: Asia Pacific Autologous Fat Grafting Device Revenue Share (%), by Country 2025 & 2033
List of Tables
- Table 1: Global Autologous Fat Grafting Device Revenue undefined Forecast, by Application 2020 & 2033
- Table 2: Global Autologous Fat Grafting Device Revenue undefined Forecast, by Type 2020 & 2033
- Table 3: Global Autologous Fat Grafting Device Revenue undefined Forecast, by Region 2020 & 2033
- Table 4: Global Autologous Fat Grafting Device Revenue undefined Forecast, by Application 2020 & 2033
- Table 5: Global Autologous Fat Grafting Device Revenue undefined Forecast, by Type 2020 & 2033
- Table 6: Global Autologous Fat Grafting Device Revenue undefined Forecast, by Country 2020 & 2033
- Table 7: United States Autologous Fat Grafting Device Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 8: Canada Autologous Fat Grafting Device Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 9: Mexico Autologous Fat Grafting Device Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 10: Global Autologous Fat Grafting Device Revenue undefined Forecast, by Application 2020 & 2033
- Table 11: Global Autologous Fat Grafting Device Revenue undefined Forecast, by Type 2020 & 2033
- Table 12: Global Autologous Fat Grafting Device Revenue undefined Forecast, by Country 2020 & 2033
- Table 13: Brazil Autologous Fat Grafting Device Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 14: Argentina Autologous Fat Grafting Device Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 15: Rest of South America Autologous Fat Grafting Device Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 16: Global Autologous Fat Grafting Device Revenue undefined Forecast, by Application 2020 & 2033
- Table 17: Global Autologous Fat Grafting Device Revenue undefined Forecast, by Type 2020 & 2033
- Table 18: Global Autologous Fat Grafting Device Revenue undefined Forecast, by Country 2020 & 2033
- Table 19: United Kingdom Autologous Fat Grafting Device Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 20: Germany Autologous Fat Grafting Device Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 21: France Autologous Fat Grafting Device Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 22: Italy Autologous Fat Grafting Device Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 23: Spain Autologous Fat Grafting Device Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 24: Russia Autologous Fat Grafting Device Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 25: Benelux Autologous Fat Grafting Device Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 26: Nordics Autologous Fat Grafting Device Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 27: Rest of Europe Autologous Fat Grafting Device Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 28: Global Autologous Fat Grafting Device Revenue undefined Forecast, by Application 2020 & 2033
- Table 29: Global Autologous Fat Grafting Device Revenue undefined Forecast, by Type 2020 & 2033
- Table 30: Global Autologous Fat Grafting Device Revenue undefined Forecast, by Country 2020 & 2033
- Table 31: Turkey Autologous Fat Grafting Device Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 32: Israel Autologous Fat Grafting Device Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 33: GCC Autologous Fat Grafting Device Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 34: North Africa Autologous Fat Grafting Device Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 35: South Africa Autologous Fat Grafting Device Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 36: Rest of Middle East & Africa Autologous Fat Grafting Device Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 37: Global Autologous Fat Grafting Device Revenue undefined Forecast, by Application 2020 & 2033
- Table 38: Global Autologous Fat Grafting Device Revenue undefined Forecast, by Type 2020 & 2033
- Table 39: Global Autologous Fat Grafting Device Revenue undefined Forecast, by Country 2020 & 2033
- Table 40: China Autologous Fat Grafting Device Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 41: India Autologous Fat Grafting Device Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 42: Japan Autologous Fat Grafting Device Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 43: South Korea Autologous Fat Grafting Device Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 44: ASEAN Autologous Fat Grafting Device Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 45: Oceania Autologous Fat Grafting Device Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 46: Rest of Asia Pacific Autologous Fat Grafting Device Revenue (undefined) Forecast, by Application 2020 & 2033
Frequently Asked Questions
1. What is the projected Compound Annual Growth Rate (CAGR) of the Autologous Fat Grafting Device?
The projected CAGR is approximately 16.04%.
2. Which companies are prominent players in the Autologous Fat Grafting Device?
Key companies in the market include Solta Medical, Cynosure Inc., Alma Lasers, AbbVie, Cutera, Erchonia, Bimini Health Tech, Candela Medical, Wells Johnson Co., Genesis Biosystems, Human Med, Moeller Medical, MicroAire Surgical, Smart Lipo, MTF Biologics.
3. What are the main segments of the Autologous Fat Grafting Device?
The market segments include Application, Type.
4. Can you provide details about the market size?
The market size is estimated to be USD XXX N/A as of 2022.
5. What are some drivers contributing to market growth?
N/A
6. What are the notable trends driving market growth?
N/A
7. Are there any restraints impacting market growth?
N/A
8. Can you provide examples of recent developments in the market?
N/A
9. What pricing options are available for accessing the report?
Pricing options include single-user, multi-user, and enterprise licenses priced at USD 2900.00, USD 4350.00, and USD 5800.00 respectively.
10. Is the market size provided in terms of value or volume?
The market size is provided in terms of value, measured in N/A.
11. Are there any specific market keywords associated with the report?
Yes, the market keyword associated with the report is "Autologous Fat Grafting Device," which aids in identifying and referencing the specific market segment covered.
12. How do I determine which pricing option suits my needs best?
The pricing options vary based on user requirements and access needs. Individual users may opt for single-user licenses, while businesses requiring broader access may choose multi-user or enterprise licenses for cost-effective access to the report.
13. Are there any additional resources or data provided in the Autologous Fat Grafting Device report?
While the report offers comprehensive insights, it's advisable to review the specific contents or supplementary materials provided to ascertain if additional resources or data are available.
14. How can I stay updated on further developments or reports in the Autologous Fat Grafting Device?
To stay informed about further developments, trends, and reports in the Autologous Fat Grafting Device, consider subscribing to industry newsletters, following relevant companies and organizations, or regularly checking reputable industry news sources and publications.
Methodology
Step 1 - Identification of Relevant Samples Size from Population Database



Step 2 - Approaches for Defining Global Market Size (Value, Volume* & Price*)

Note*: In applicable scenarios
Step 3 - Data Sources
Primary Research
- Web Analytics
- Survey Reports
- Research Institute
- Latest Research Reports
- Opinion Leaders
Secondary Research
- Annual Reports
- White Paper
- Latest Press Release
- Industry Association
- Paid Database
- Investor Presentations

Step 4 - Data Triangulation
Involves using different sources of information in order to increase the validity of a study
These sources are likely to be stakeholders in a program - participants, other researchers, program staff, other community members, and so on.
Then we put all data in single framework & apply various statistical tools to find out the dynamic on the market.
During the analysis stage, feedback from the stakeholder groups would be compared to determine areas of agreement as well as areas of divergence


