Key Insights
The global Guide RNA market is poised for substantial expansion, projected to reach $0.7 billion in 2025. This robust growth is underpinned by an impressive Compound Annual Growth Rate (CAGR) of 18.4% during the forecast period of 2025-2033. The market's dynamism is driven by a confluence of factors, most notably the burgeoning applications within bioengineering and medicine. The increasing adoption of CRISPR-based gene editing technologies in drug discovery, development of novel therapeutics, and advancements in genetic research are primary catalysts. Furthermore, the expanding research and development activities globally, coupled with a rising demand for personalized medicine, are significantly contributing to this upward trajectory. The market is segmented into various types of guide RNAs, including sgRNA and Guide RNA in RNA Editing, with each segment witnessing increasing innovation and adoption.
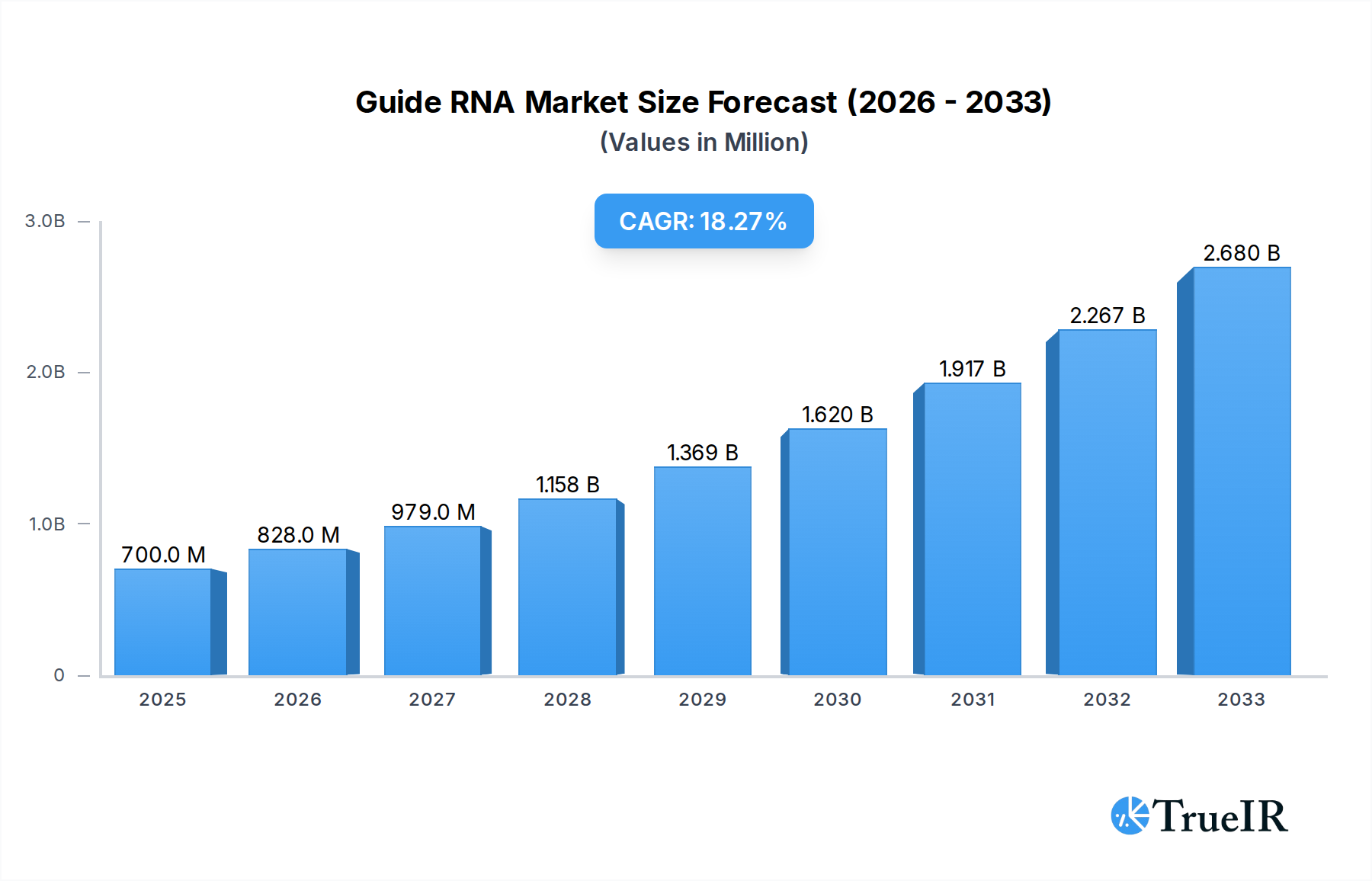
Guide RNA Market Size (In Million)

The market's impressive growth is further bolstered by key trends such as the development of more efficient and specific guide RNA designs, enabling precise gene modifications. The growing investment in life sciences research and the increasing prevalence of genetic disorders are also fueling the demand for advanced gene editing tools. While the market presents a highly optimistic outlook, potential restraints such as the high cost of specialized reagents and the need for stringent regulatory approvals for therapeutic applications warrant consideration. However, the overall market landscape is characterized by intense innovation and a growing number of collaborations among key players, including Thermo Fisher Scientific, Agilent Technologies, and Synthego Corporation, who are instrumental in driving technological advancements and expanding market accessibility. This dynamic environment indicates a highly promising future for the Guide RNA market.
Guide RNA Company Market Share

Unlocking the Potential: A Comprehensive Guide RNA Market Analysis (2019-2033)
This in-depth report provides a dynamic, SEO-optimized analysis of the global Guide RNA market, leveraging high-volume keywords to engage industry professionals and enhance search engine visibility. Covering the period from 2019 to 2033, with a base year of 2025 and a forecast period extending to 2033, this report offers a comprehensive understanding of market structure, trends, opportunities, dominant segments, product innovations, key drivers, challenges, and the competitive landscape.
Guide RNA Market Structure & Competitive Landscape
The Guide RNA market exhibits a moderate to high concentration, driven by significant innovation and substantial capital investment. Key innovation drivers include advancements in CRISPR-based gene editing technologies, enhanced delivery mechanisms, and the development of novel guide RNA designs for increased specificity and efficacy. Regulatory impacts, while generally supportive of scientific advancement, can introduce complexities in preclinical and clinical trial approvals, influencing market entry and product development timelines. Product substitutes, such as other gene editing modalities, exist but currently lack the widespread adoption and versatility of guide RNA. End-user segmentation reveals a strong demand from research institutions and pharmaceutical companies, with growing interest from contract research organizations (CROs) and academic laboratories. Merger and acquisition (M&A) trends are moderately active, indicating a strategic consolidation phase as larger players seek to acquire specialized technologies and expand their portfolios. M&A volumes are projected to increase by approximately 10-15% over the forecast period, with an estimated 30-40 M&A deals annually, reflecting a strategic drive towards portfolio expansion and market dominance.
- Key Innovation Drivers:
- CRISPR-Cas gene editing advancements
- Improved guide RNA design and synthesis
- Novel delivery systems (e.g., viral vectors, lipid nanoparticles)
- Development of multiplexed editing strategies
- Regulatory Impacts:
- FDA and EMA approval pathways for gene therapies
- Ethical considerations in germline editing research
- Intellectual property landscape surrounding guide RNA technologies
- End-User Segmentation:
- Academic and research institutions
- Biotechnology and pharmaceutical companies
- Contract Research Organizations (CROs)
- Diagnostic laboratories
- M&A Trends:
- Acquisition of novel CRISPR platforms
- Integration of guide RNA manufacturing capabilities
- Strategic partnerships to accelerate drug development
Guide RNA Market Trends & Opportunities
The global Guide RNA market is poised for robust growth, projected to expand from an estimated $1.5 billion in 2025 to a projected $8.5 billion by 2033, exhibiting a Compound Annual Growth Rate (CAGR) of approximately 20%. This significant expansion is fueled by the burgeoning field of precision medicine, the increasing adoption of gene editing technologies in therapeutic development, and the expanding applications in bioengineering and diagnostics. Technological shifts are central to this growth, with ongoing refinements in guide RNA synthesis, delivery, and specificity leading to enhanced therapeutic outcomes and expanded research capabilities. The market is witnessing a surge in demand for custom-designed guide RNAs tailored to specific gene targets and experimental conditions. Consumer preferences are increasingly leaning towards higher throughput screening and more efficient gene modification tools, driving innovation in multiplexed guide RNA libraries and automated synthesis platforms. Competitive dynamics are intensifying, characterized by strategic collaborations, product launches, and increasing investment in research and development by key players. Market penetration rates for guide RNA in therapeutic applications are expected to climb from approximately 25% in 2025 to over 50% by 2033, as more gene editing-based therapies advance through clinical trials and gain regulatory approval. The increasing prevalence of genetic disorders, coupled with a growing understanding of their molecular basis, presents a substantial opportunity for gene editing therapies utilizing guide RNA. Furthermore, the application of guide RNA in agricultural biotechnology for crop improvement and in industrial biotechnology for the production of biofuels and biomaterials is an emerging area with significant growth potential. The development of more efficient and cost-effective manufacturing processes for synthetic guide RNAs will also be crucial in driving broader market adoption.
Dominant Markets & Segments in Guide RNA
The Medicine application segment is the dominant force within the Guide RNA market, driven by the revolutionary potential of gene editing in treating a wide array of genetic and acquired diseases. Within this segment, the sgRNA (single-guide RNA) type commands the largest market share, owing to its established efficacy and widespread use in CRISPR-Cas9 gene editing systems. Geographically, North America, particularly the United States, is the leading market due to its robust biopharmaceutical industry, extensive research funding, and favorable regulatory environment for gene therapy development.
- Dominant Application:
- Medicine: This segment is propelled by the development of novel gene therapies for oncology, rare genetic disorders, infectious diseases, and cardiovascular conditions. The ability to precisely correct disease-causing mutations offers unprecedented therapeutic possibilities, attracting substantial investment and research focus.
- Dominant Type:
- sgRNA: Single-guide RNAs are the workhorse of most CRISPR-based gene editing applications. Their relative simplicity in design and established effectiveness in guiding Cas nucleases to target DNA sequences make them the preferred choice for a vast majority of research and therapeutic endeavors.
- Leading Region:
- North America: The region's leadership is attributed to its strong presence of leading biotechnology companies, significant government and private funding for life sciences research, and a well-established regulatory framework that supports the advancement of gene-editing technologies. The presence of key research institutions and a skilled workforce further bolsters its dominance.
- Key Growth Drivers in Medicine Application:
- Advancements in CRISPR-Cas gene editing technology.
- Increasing prevalence of genetic diseases and a growing demand for targeted therapies.
- Significant investments in gene therapy research and development.
- Favorable reimbursement policies for innovative treatments.
- Key Growth Drivers for sgRNA Type:
- High specificity and efficiency in gene editing.
- Ease of design and synthesis for various applications.
- Widespread adoption in research and preclinical development.
- Key Growth Drivers in North America:
- Strong funding for biotechnology and pharmaceutical R&D.
- Presence of leading gene editing companies and research institutions.
- Streamlined regulatory pathways for gene therapy approvals.
- Access to a large patient population for clinical trials.
Guide RNA Product Analysis
Guide RNA products are characterized by their increasing sophistication, enabling precise and efficient gene editing across a spectrum of applications. Innovations focus on enhanced specificity, reduced off-target effects, and improved delivery mechanisms, often in customized formats for specific research needs or therapeutic targets. Competitive advantages lie in proprietary design algorithms, advanced synthesis chemistries that ensure high purity and yield, and robust quality control measures. The market is seeing a proliferation of catalog products for common gene targets alongside bespoke synthesis services for novel applications in bioengineering, medicine, and beyond, ensuring precise genetic manipulation for groundbreaking research and therapy development.
Key Drivers, Barriers & Challenges in Guide RNA
The Guide RNA market is propelled by the transformative potential of gene editing technologies, driving demand for precise genetic manipulation in both research and therapeutic applications. Technological advancements in CRISPR-Cas systems, coupled with increasing investments in biotechnology R&D, are key growth catalysts. Favorable regulatory environments for gene therapies in select regions and the growing understanding of genetic disease mechanisms further fuel market expansion.
However, significant challenges persist. High manufacturing costs for synthetic guide RNAs can limit accessibility. Regulatory complexities surrounding gene editing therapies, particularly concerning long-term safety and ethical implications, pose barriers to market entry. Intellectual property disputes and competition from alternative gene editing modalities also present hurdles. Supply chain disruptions and the need for specialized infrastructure for large-scale production can impact market growth.
Growth Drivers in the Guide RNA Market
The Guide RNA market's growth is underpinned by several key drivers. Technological advancements in CRISPR-Cas gene editing continue to enhance precision, efficiency, and safety, expanding the therapeutic and research utility of guide RNAs. Increased funding and investment in biotechnology and pharmaceutical R&D, particularly in gene therapy, are significantly boosting demand. The growing understanding of genetic diseases and a corresponding push for targeted therapies create a fertile ground for guide RNA applications. Furthermore, supportive government policies and initiatives aimed at fostering innovation in genetic medicine are creating a favorable ecosystem for market expansion. The development of more cost-effective synthesis methods is also a critical growth driver.
Challenges Impacting Guide RNA Growth
Several challenges can impact the growth trajectory of the Guide RNA market. Regulatory hurdles, including evolving guidelines for gene therapy approval and ethical considerations, can slow down product development and market entry. Supply chain issues, such as the availability of raw materials and the capacity for large-scale, high-quality synthetic guide RNA production, can constrain market expansion. Intense competition from other gene editing technologies and the presence of numerous market players can lead to pricing pressures and the need for continuous innovation. Off-target effects and the need for robust validation of guide RNA specificity in therapeutic applications remain ongoing scientific challenges that require diligent research and development.
Key Players Shaping the Guide RNA Market
- Thermo Fisher Scientific
- Agilent Technologies
- System Biosciences
- Jena Bioscience GmbH
- Merck KGaA
- Azenta
- Takara Bio
- Synbio Technologies
- Revvity
- Synthego Corporation
- GenScript Biotech Corporation
- Omicsbio
- SBS Genetech
Significant Guide RNA Industry Milestones
- 2019: Launch of advanced CRISPR-based therapeutic candidates targeting rare genetic diseases.
- 2020: Significant increase in venture capital funding for gene editing startups.
- 2021: FDA approval of the first CRISPR-based gene therapy, signaling a new era for the field.
- 2022: Development of novel guide RNA designs with enhanced specificity and reduced off-target effects.
- 2023: Expansion of guide RNA applications into agricultural biotechnology for crop improvement.
- 2024: Advancements in automated synthesis platforms for high-throughput guide RNA production.
Future Outlook for Guide RNA Market
The future outlook for the Guide RNA market is exceptionally bright, characterized by sustained high growth fueled by ongoing technological innovation and expanding therapeutic applications. Strategic opportunities lie in the development of more precise and safer gene editing tools, the exploration of novel delivery systems, and the expansion into emerging markets and applications beyond traditional medicine, such as agriculture and industrial biotechnology. The increasing focus on personalized medicine and the development of gene therapies for a wider range of diseases will continue to drive demand. Advancements in manufacturing efficiency and cost reduction are expected to further democratize access to these transformative technologies, solidifying the critical role of guide RNA in the future of life sciences.
Guide RNA Segmentation
-
1. Application
- 1.1. Bioengineering
- 1.2. Medicine
- 1.3. Others
-
2. Types
- 2.1. sgRNA
- 2.2. Guide RNA in RNA Editing
- 2.3. Others
Guide RNA Segmentation By Geography
-
1. North America
- 1.1. United States
- 1.2. Canada
- 1.3. Mexico
-
2. South America
- 2.1. Brazil
- 2.2. Argentina
- 2.3. Rest of South America
-
3. Europe
- 3.1. United Kingdom
- 3.2. Germany
- 3.3. France
- 3.4. Italy
- 3.5. Spain
- 3.6. Russia
- 3.7. Benelux
- 3.8. Nordics
- 3.9. Rest of Europe
-
4. Middle East & Africa
- 4.1. Turkey
- 4.2. Israel
- 4.3. GCC
- 4.4. North Africa
- 4.5. South Africa
- 4.6. Rest of Middle East & Africa
-
5. Asia Pacific
- 5.1. China
- 5.2. India
- 5.3. Japan
- 5.4. South Korea
- 5.5. ASEAN
- 5.6. Oceania
- 5.7. Rest of Asia Pacific
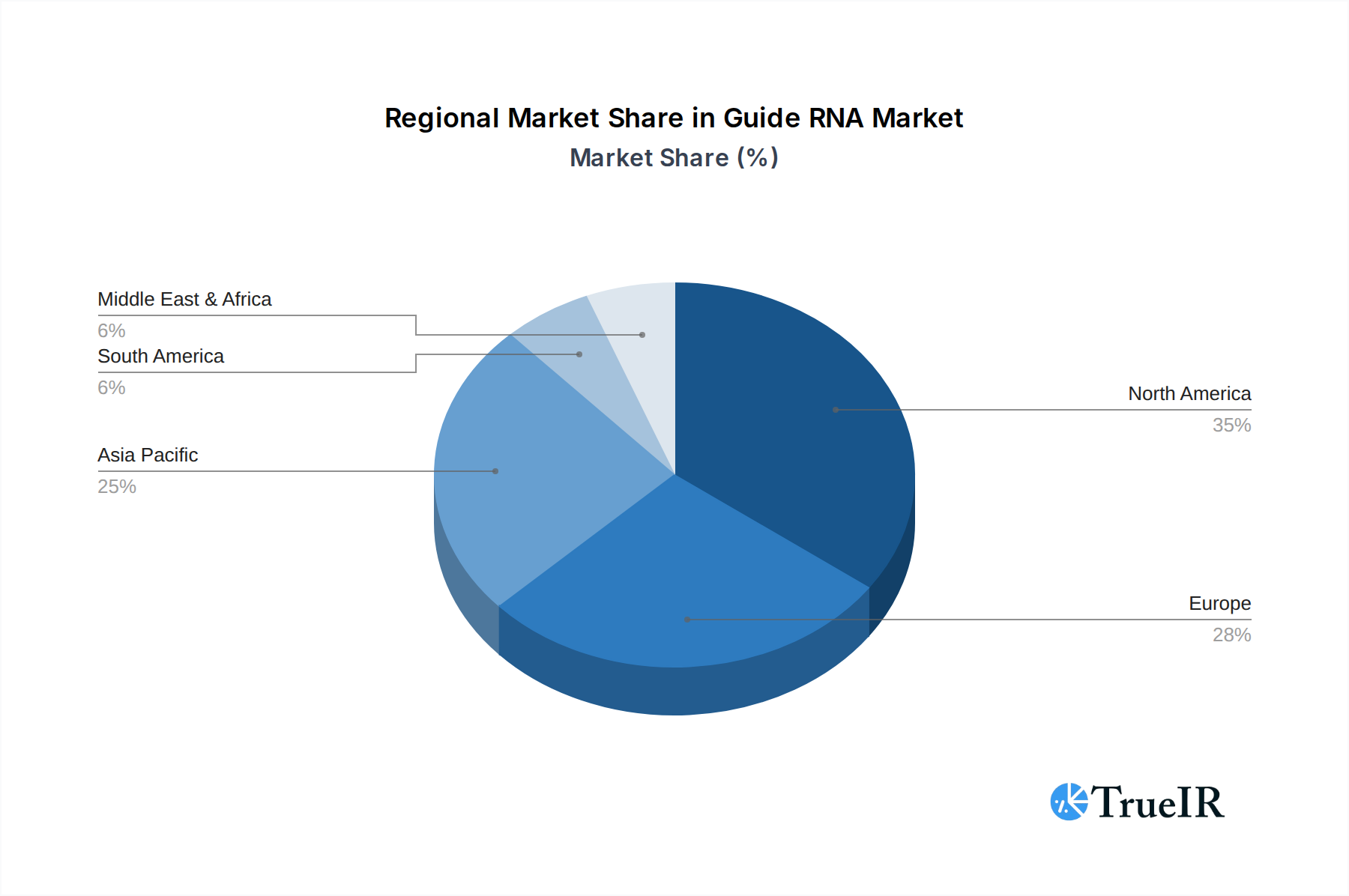
Guide RNA Regional Market Share

Geographic Coverage of Guide RNA
Guide RNA REPORT HIGHLIGHTS
| Aspects | Details |
|---|---|
| Study Period | 2020-2034 |
| Base Year | 2025 |
| Estimated Year | 2026 |
| Forecast Period | 2026-2034 |
| Historical Period | 2020-2025 |
| Growth Rate | CAGR of 18.4% from 2020-2034 |
| Segmentation |
|
Table of Contents
- 1. Introduction
- 1.1. Research Scope
- 1.2. Market Segmentation
- 1.3. Research Objective
- 1.4. Definitions and Assumptions
- 2. Executive Summary
- 2.1. Market Snapshot
- 3. Market Dynamics
- 3.1. Market Drivers
- 3.2. Market Restrains
- 3.3. Market Trends
- 3.4. Market Opportunities
- 4. Market Factor Analysis
- 4.1. Porters Five Forces
- 4.1.1. Bargaining Power of Suppliers
- 4.1.2. Bargaining Power of Buyers
- 4.1.3. Threat of New Entrants
- 4.1.4. Threat of Substitutes
- 4.1.5. Competitive Rivalry
- 4.2. PESTEL analysis
- 4.3. BCG Analysis
- 4.3.1. Stars (High Growth, High Market Share)
- 4.3.2. Cash Cows (Low Growth, High Market Share)
- 4.3.3. Question Mark (High Growth, Low Market Share)
- 4.3.4. Dogs (Low Growth, Low Market Share)
- 4.4. Ansoff Matrix Analysis
- 4.5. Supply Chain Analysis
- 4.6. Regulatory Landscape
- 4.7. Current Market Potential and Opportunity Assessment (TAM–SAM–SOM Framework)
- 4.8. TIR Analyst Note
- 4.1. Porters Five Forces
- 5. Market Analysis, Insights and Forecast 2021-2033
- 5.1. Market Analysis, Insights and Forecast - by Application
- 5.1.1. Bioengineering
- 5.1.2. Medicine
- 5.1.3. Others
- 5.2. Market Analysis, Insights and Forecast - by Types
- 5.2.1. sgRNA
- 5.2.2. Guide RNA in RNA Editing
- 5.2.3. Others
- 5.3. Market Analysis, Insights and Forecast - by Region
- 5.3.1. North America
- 5.3.2. South America
- 5.3.3. Europe
- 5.3.4. Middle East & Africa
- 5.3.5. Asia Pacific
- 5.1. Market Analysis, Insights and Forecast - by Application
- 6. Global Guide RNA Analysis, Insights and Forecast, 2021-2033
- 6.1. Market Analysis, Insights and Forecast - by Application
- 6.1.1. Bioengineering
- 6.1.2. Medicine
- 6.1.3. Others
- 6.2. Market Analysis, Insights and Forecast - by Types
- 6.2.1. sgRNA
- 6.2.2. Guide RNA in RNA Editing
- 6.2.3. Others
- 6.1. Market Analysis, Insights and Forecast - by Application
- 7. North America Guide RNA Analysis, Insights and Forecast, 2020-2032
- 7.1. Market Analysis, Insights and Forecast - by Application
- 7.1.1. Bioengineering
- 7.1.2. Medicine
- 7.1.3. Others
- 7.2. Market Analysis, Insights and Forecast - by Types
- 7.2.1. sgRNA
- 7.2.2. Guide RNA in RNA Editing
- 7.2.3. Others
- 7.1. Market Analysis, Insights and Forecast - by Application
- 8. South America Guide RNA Analysis, Insights and Forecast, 2020-2032
- 8.1. Market Analysis, Insights and Forecast - by Application
- 8.1.1. Bioengineering
- 8.1.2. Medicine
- 8.1.3. Others
- 8.2. Market Analysis, Insights and Forecast - by Types
- 8.2.1. sgRNA
- 8.2.2. Guide RNA in RNA Editing
- 8.2.3. Others
- 8.1. Market Analysis, Insights and Forecast - by Application
- 9. Europe Guide RNA Analysis, Insights and Forecast, 2020-2032
- 9.1. Market Analysis, Insights and Forecast - by Application
- 9.1.1. Bioengineering
- 9.1.2. Medicine
- 9.1.3. Others
- 9.2. Market Analysis, Insights and Forecast - by Types
- 9.2.1. sgRNA
- 9.2.2. Guide RNA in RNA Editing
- 9.2.3. Others
- 9.1. Market Analysis, Insights and Forecast - by Application
- 10. Middle East & Africa Guide RNA Analysis, Insights and Forecast, 2020-2032
- 10.1. Market Analysis, Insights and Forecast - by Application
- 10.1.1. Bioengineering
- 10.1.2. Medicine
- 10.1.3. Others
- 10.2. Market Analysis, Insights and Forecast - by Types
- 10.2.1. sgRNA
- 10.2.2. Guide RNA in RNA Editing
- 10.2.3. Others
- 10.1. Market Analysis, Insights and Forecast - by Application
- 11. Asia Pacific Guide RNA Analysis, Insights and Forecast, 2020-2032
- 11.1. Market Analysis, Insights and Forecast - by Application
- 11.1.1. Bioengineering
- 11.1.2. Medicine
- 11.1.3. Others
- 11.2. Market Analysis, Insights and Forecast - by Types
- 11.2.1. sgRNA
- 11.2.2. Guide RNA in RNA Editing
- 11.2.3. Others
- 11.1. Market Analysis, Insights and Forecast - by Application
- 12. Competitive Analysis
- 12.1. Company Profiles
- 12.1.1 Thermo Fisher Scientific
- 12.1.1.1. Company Overview
- 12.1.1.2. Products
- 12.1.1.3. Company Financials
- 12.1.1.4. SWOT Analysis
- 12.1.2 Agilent Technologies
- 12.1.2.1. Company Overview
- 12.1.2.2. Products
- 12.1.2.3. Company Financials
- 12.1.2.4. SWOT Analysis
- 12.1.3 System Biosciences
- 12.1.3.1. Company Overview
- 12.1.3.2. Products
- 12.1.3.3. Company Financials
- 12.1.3.4. SWOT Analysis
- 12.1.4 Jena Bioscience GmbH
- 12.1.4.1. Company Overview
- 12.1.4.2. Products
- 12.1.4.3. Company Financials
- 12.1.4.4. SWOT Analysis
- 12.1.5 Merck KGaA
- 12.1.5.1. Company Overview
- 12.1.5.2. Products
- 12.1.5.3. Company Financials
- 12.1.5.4. SWOT Analysis
- 12.1.6 Azenta
- 12.1.6.1. Company Overview
- 12.1.6.2. Products
- 12.1.6.3. Company Financials
- 12.1.6.4. SWOT Analysis
- 12.1.7 Takara Bio
- 12.1.7.1. Company Overview
- 12.1.7.2. Products
- 12.1.7.3. Company Financials
- 12.1.7.4. SWOT Analysis
- 12.1.8 Synbio Technologies
- 12.1.8.1. Company Overview
- 12.1.8.2. Products
- 12.1.8.3. Company Financials
- 12.1.8.4. SWOT Analysis
- 12.1.9 Revvity
- 12.1.9.1. Company Overview
- 12.1.9.2. Products
- 12.1.9.3. Company Financials
- 12.1.9.4. SWOT Analysis
- 12.1.10 Synthego Corporation
- 12.1.10.1. Company Overview
- 12.1.10.2. Products
- 12.1.10.3. Company Financials
- 12.1.10.4. SWOT Analysis
- 12.1.11 GenScript Biotech Corporation
- 12.1.11.1. Company Overview
- 12.1.11.2. Products
- 12.1.11.3. Company Financials
- 12.1.11.4. SWOT Analysis
- 12.1.12 Omicsbio
- 12.1.12.1. Company Overview
- 12.1.12.2. Products
- 12.1.12.3. Company Financials
- 12.1.12.4. SWOT Analysis
- 12.1.13 SBS Genetech
- 12.1.13.1. Company Overview
- 12.1.13.2. Products
- 12.1.13.3. Company Financials
- 12.1.13.4. SWOT Analysis
- 12.1.1 Thermo Fisher Scientific
- 12.2. Market Entropy
- 12.2.1 Company's Key Areas Served
- 12.2.2 Recent Developments
- 12.3. Company Market Share Analysis 2025
- 12.3.1 Top 5 Companies Market Share Analysis
- 12.3.2 Top 3 Companies Market Share Analysis
- 12.4. List of Potential Customers
- 13. Research Methodology
List of Figures
- Figure 1: Global Guide RNA Revenue Breakdown (billion, %) by Region 2025 & 2033
- Figure 2: North America Guide RNA Revenue (billion), by Application 2025 & 2033
- Figure 3: North America Guide RNA Revenue Share (%), by Application 2025 & 2033
- Figure 4: North America Guide RNA Revenue (billion), by Types 2025 & 2033
- Figure 5: North America Guide RNA Revenue Share (%), by Types 2025 & 2033
- Figure 6: North America Guide RNA Revenue (billion), by Country 2025 & 2033
- Figure 7: North America Guide RNA Revenue Share (%), by Country 2025 & 2033
- Figure 8: South America Guide RNA Revenue (billion), by Application 2025 & 2033
- Figure 9: South America Guide RNA Revenue Share (%), by Application 2025 & 2033
- Figure 10: South America Guide RNA Revenue (billion), by Types 2025 & 2033
- Figure 11: South America Guide RNA Revenue Share (%), by Types 2025 & 2033
- Figure 12: South America Guide RNA Revenue (billion), by Country 2025 & 2033
- Figure 13: South America Guide RNA Revenue Share (%), by Country 2025 & 2033
- Figure 14: Europe Guide RNA Revenue (billion), by Application 2025 & 2033
- Figure 15: Europe Guide RNA Revenue Share (%), by Application 2025 & 2033
- Figure 16: Europe Guide RNA Revenue (billion), by Types 2025 & 2033
- Figure 17: Europe Guide RNA Revenue Share (%), by Types 2025 & 2033
- Figure 18: Europe Guide RNA Revenue (billion), by Country 2025 & 2033
- Figure 19: Europe Guide RNA Revenue Share (%), by Country 2025 & 2033
- Figure 20: Middle East & Africa Guide RNA Revenue (billion), by Application 2025 & 2033
- Figure 21: Middle East & Africa Guide RNA Revenue Share (%), by Application 2025 & 2033
- Figure 22: Middle East & Africa Guide RNA Revenue (billion), by Types 2025 & 2033
- Figure 23: Middle East & Africa Guide RNA Revenue Share (%), by Types 2025 & 2033
- Figure 24: Middle East & Africa Guide RNA Revenue (billion), by Country 2025 & 2033
- Figure 25: Middle East & Africa Guide RNA Revenue Share (%), by Country 2025 & 2033
- Figure 26: Asia Pacific Guide RNA Revenue (billion), by Application 2025 & 2033
- Figure 27: Asia Pacific Guide RNA Revenue Share (%), by Application 2025 & 2033
- Figure 28: Asia Pacific Guide RNA Revenue (billion), by Types 2025 & 2033
- Figure 29: Asia Pacific Guide RNA Revenue Share (%), by Types 2025 & 2033
- Figure 30: Asia Pacific Guide RNA Revenue (billion), by Country 2025 & 2033
- Figure 31: Asia Pacific Guide RNA Revenue Share (%), by Country 2025 & 2033
List of Tables
- Table 1: Global Guide RNA Revenue billion Forecast, by Application 2020 & 2033
- Table 2: Global Guide RNA Revenue billion Forecast, by Types 2020 & 2033
- Table 3: Global Guide RNA Revenue billion Forecast, by Region 2020 & 2033
- Table 4: Global Guide RNA Revenue billion Forecast, by Application 2020 & 2033
- Table 5: Global Guide RNA Revenue billion Forecast, by Types 2020 & 2033
- Table 6: Global Guide RNA Revenue billion Forecast, by Country 2020 & 2033
- Table 7: United States Guide RNA Revenue (billion) Forecast, by Application 2020 & 2033
- Table 8: Canada Guide RNA Revenue (billion) Forecast, by Application 2020 & 2033
- Table 9: Mexico Guide RNA Revenue (billion) Forecast, by Application 2020 & 2033
- Table 10: Global Guide RNA Revenue billion Forecast, by Application 2020 & 2033
- Table 11: Global Guide RNA Revenue billion Forecast, by Types 2020 & 2033
- Table 12: Global Guide RNA Revenue billion Forecast, by Country 2020 & 2033
- Table 13: Brazil Guide RNA Revenue (billion) Forecast, by Application 2020 & 2033
- Table 14: Argentina Guide RNA Revenue (billion) Forecast, by Application 2020 & 2033
- Table 15: Rest of South America Guide RNA Revenue (billion) Forecast, by Application 2020 & 2033
- Table 16: Global Guide RNA Revenue billion Forecast, by Application 2020 & 2033
- Table 17: Global Guide RNA Revenue billion Forecast, by Types 2020 & 2033
- Table 18: Global Guide RNA Revenue billion Forecast, by Country 2020 & 2033
- Table 19: United Kingdom Guide RNA Revenue (billion) Forecast, by Application 2020 & 2033
- Table 20: Germany Guide RNA Revenue (billion) Forecast, by Application 2020 & 2033
- Table 21: France Guide RNA Revenue (billion) Forecast, by Application 2020 & 2033
- Table 22: Italy Guide RNA Revenue (billion) Forecast, by Application 2020 & 2033
- Table 23: Spain Guide RNA Revenue (billion) Forecast, by Application 2020 & 2033
- Table 24: Russia Guide RNA Revenue (billion) Forecast, by Application 2020 & 2033
- Table 25: Benelux Guide RNA Revenue (billion) Forecast, by Application 2020 & 2033
- Table 26: Nordics Guide RNA Revenue (billion) Forecast, by Application 2020 & 2033
- Table 27: Rest of Europe Guide RNA Revenue (billion) Forecast, by Application 2020 & 2033
- Table 28: Global Guide RNA Revenue billion Forecast, by Application 2020 & 2033
- Table 29: Global Guide RNA Revenue billion Forecast, by Types 2020 & 2033
- Table 30: Global Guide RNA Revenue billion Forecast, by Country 2020 & 2033
- Table 31: Turkey Guide RNA Revenue (billion) Forecast, by Application 2020 & 2033
- Table 32: Israel Guide RNA Revenue (billion) Forecast, by Application 2020 & 2033
- Table 33: GCC Guide RNA Revenue (billion) Forecast, by Application 2020 & 2033
- Table 34: North Africa Guide RNA Revenue (billion) Forecast, by Application 2020 & 2033
- Table 35: South Africa Guide RNA Revenue (billion) Forecast, by Application 2020 & 2033
- Table 36: Rest of Middle East & Africa Guide RNA Revenue (billion) Forecast, by Application 2020 & 2033
- Table 37: Global Guide RNA Revenue billion Forecast, by Application 2020 & 2033
- Table 38: Global Guide RNA Revenue billion Forecast, by Types 2020 & 2033
- Table 39: Global Guide RNA Revenue billion Forecast, by Country 2020 & 2033
- Table 40: China Guide RNA Revenue (billion) Forecast, by Application 2020 & 2033
- Table 41: India Guide RNA Revenue (billion) Forecast, by Application 2020 & 2033
- Table 42: Japan Guide RNA Revenue (billion) Forecast, by Application 2020 & 2033
- Table 43: South Korea Guide RNA Revenue (billion) Forecast, by Application 2020 & 2033
- Table 44: ASEAN Guide RNA Revenue (billion) Forecast, by Application 2020 & 2033
- Table 45: Oceania Guide RNA Revenue (billion) Forecast, by Application 2020 & 2033
- Table 46: Rest of Asia Pacific Guide RNA Revenue (billion) Forecast, by Application 2020 & 2033
Frequently Asked Questions
1. What is the projected Compound Annual Growth Rate (CAGR) of the Guide RNA?
The projected CAGR is approximately 18.4%.
2. Which companies are prominent players in the Guide RNA?
Key companies in the market include Thermo Fisher Scientific, Agilent Technologies, System Biosciences, Jena Bioscience GmbH, Merck KGaA, Azenta, Takara Bio, Synbio Technologies, Revvity, Synthego Corporation, GenScript Biotech Corporation, Omicsbio, SBS Genetech.
3. What are the main segments of the Guide RNA?
The market segments include Application, Types.
4. Can you provide details about the market size?
The market size is estimated to be USD 0.7 billion as of 2022.
5. What are some drivers contributing to market growth?
N/A
6. What are the notable trends driving market growth?
N/A
7. Are there any restraints impacting market growth?
N/A
8. Can you provide examples of recent developments in the market?
N/A
9. What pricing options are available for accessing the report?
Pricing options include single-user, multi-user, and enterprise licenses priced at USD 2900.00, USD 4350.00, and USD 5800.00 respectively.
10. Is the market size provided in terms of value or volume?
The market size is provided in terms of value, measured in billion.
11. Are there any specific market keywords associated with the report?
Yes, the market keyword associated with the report is "Guide RNA," which aids in identifying and referencing the specific market segment covered.
12. How do I determine which pricing option suits my needs best?
The pricing options vary based on user requirements and access needs. Individual users may opt for single-user licenses, while businesses requiring broader access may choose multi-user or enterprise licenses for cost-effective access to the report.
13. Are there any additional resources or data provided in the Guide RNA report?
While the report offers comprehensive insights, it's advisable to review the specific contents or supplementary materials provided to ascertain if additional resources or data are available.
14. How can I stay updated on further developments or reports in the Guide RNA?
To stay informed about further developments, trends, and reports in the Guide RNA, consider subscribing to industry newsletters, following relevant companies and organizations, or regularly checking reputable industry news sources and publications.
Methodology
Step 1 - Identification of Relevant Samples Size from Population Database



Step 2 - Approaches for Defining Global Market Size (Value, Volume* & Price*)

Note*: In applicable scenarios
Step 3 - Data Sources
Primary Research
- Web Analytics
- Survey Reports
- Research Institute
- Latest Research Reports
- Opinion Leaders
Secondary Research
- Annual Reports
- White Paper
- Latest Press Release
- Industry Association
- Paid Database
- Investor Presentations

Step 4 - Data Triangulation
Involves using different sources of information in order to increase the validity of a study
These sources are likely to be stakeholders in a program - participants, other researchers, program staff, other community members, and so on.
Then we put all data in single framework & apply various statistical tools to find out the dynamic on the market.
During the analysis stage, feedback from the stakeholder groups would be compared to determine areas of agreement as well as areas of divergence


