Key Insights
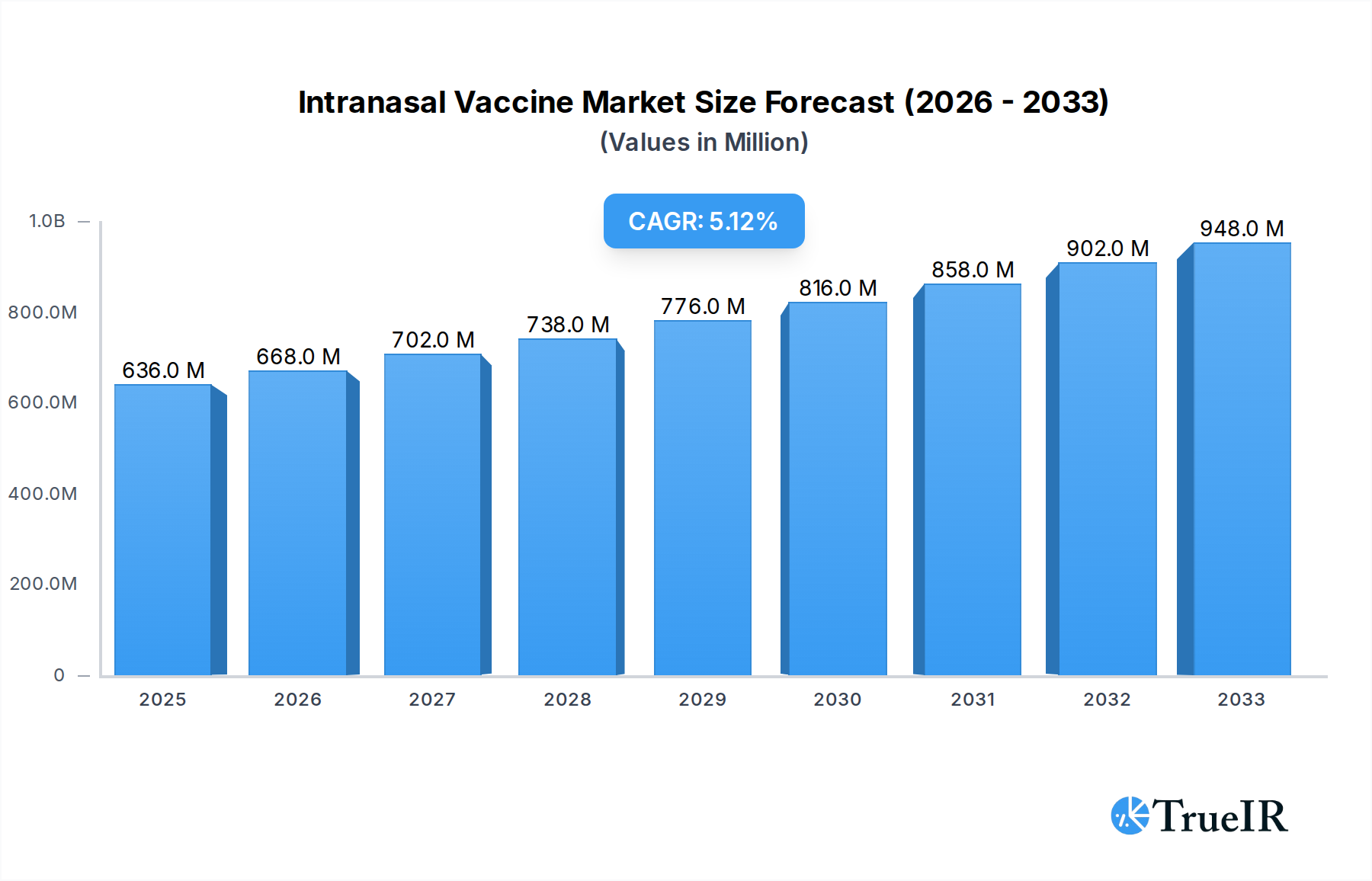
The global intranasal vaccine market is poised for significant expansion, projected to reach an estimated USD 636 million in 2025 and grow at a robust Compound Annual Growth Rate (CAGR) of 5.2% from 2025 to 2033. This upward trajectory is primarily fueled by the inherent advantages of intranasal delivery, including needle-free administration, enhanced patient compliance, and the potential for rapid immune response through mucosal immunity. The increasing prevalence of infectious diseases, coupled with the ongoing need for effective and accessible vaccination strategies, particularly for pediatric and elderly populations, are key market drivers. Furthermore, the development and approval of novel intranasal vaccines for prevalent diseases like COVID-19 and influenza are expected to significantly boost market adoption and revenue. Emerging economies, with their large and growing populations and increasing healthcare investments, present substantial opportunities for market players. The focus on preventative healthcare measures globally is further bolstering the demand for innovative vaccine delivery systems like intranasal vaccines.
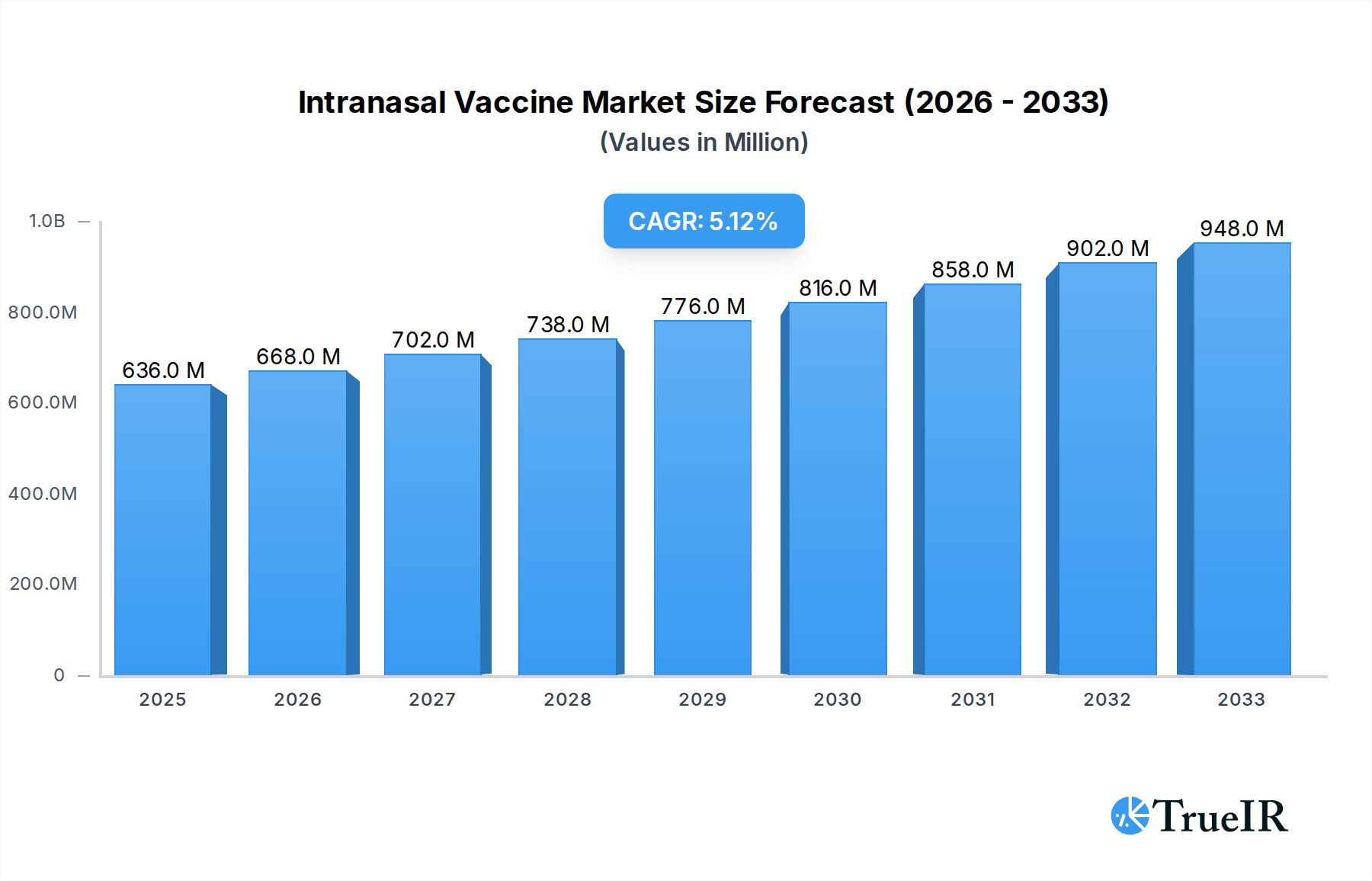
Intranasal Vaccine Market Size (In Million)

The market is segmented into distinct applications, with hospitals and clinics emerging as the primary end-users due to their established healthcare infrastructure and direct patient access. On the types front, flu vaccines and COVID-19 vaccines represent the most significant segments, reflecting the current global health priorities and the continuous demand for immunization against these viral threats. While the market demonstrates strong growth potential, certain restraints may impede its full realization. These could include regulatory hurdles for new vaccine approvals, the initial cost of developing and manufacturing intranasal vaccine technologies, and potential public perception challenges regarding the efficacy and safety of novel delivery methods compared to traditional injectable vaccines. Despite these challenges, the inherent benefits of intranasal vaccines in terms of ease of administration and improved immune responses are expected to outweigh these concerns, driving sustained market growth and innovation.
Intranasal Vaccine Company Market Share

Here is a dynamic, SEO-optimized report description for the Intranasal Vaccine market, designed for immediate use and maximum impact:
Intranasal Vaccine Market Structure & Competitive Landscape
The global intranasal vaccine market, a rapidly evolving sector poised for significant expansion, exhibits a complex and dynamic competitive landscape. Market concentration is currently moderate, with leading players like Sanofi, AstraZeneca, and GlaxoSmithKline alongside emerging innovators such as Bharat Biotech and Beijing Wantai Biopharmaceutical Co., Ltd. The study period from 2019 to 2033, with a base and estimated year of 2025, reveals a market driven by intense innovation in vaccine delivery systems. Key drivers include the pursuit of needle-free vaccination, enhanced patient compliance, and faster immune responses. Regulatory impacts are paramount, with stringent approval processes by global health authorities shaping product development and market entry. Product substitutes, primarily traditional injectable vaccines, remain a significant factor, though intranasal vaccines offer distinct advantages in ease of administration and potential for mucosal immunity.
The end-user segmentation of the market includes hospitals and clinics, with future potential in community settings. Mergers & Acquisitions (M&A) trends are increasingly evident as larger pharmaceutical companies seek to bolster their portfolios with novel intranasal technologies. M&A volumes are projected to rise, with an estimated XX million in deal value anticipated during the forecast period (2025–2033). Concentration ratios are expected to shift as strategic partnerships and acquisitions consolidate market share. The competitive environment is characterized by a constant drive for differentiation through advanced formulation, broader spectrum of protection, and cost-effectiveness.
Intranasal Vaccine Market Trends & Opportunities
The global intranasal vaccine market is on an impressive trajectory, projected to witness substantial growth driven by an increasing demand for convenient and effective immunization strategies. The market size, estimated to be in the billions of dollars for 2025, is forecast to expand at a robust Compound Annual Growth Rate (CAGR) of approximately XX% from 2025 to 2033. This expansion is fueled by a confluence of technological advancements, evolving consumer preferences, and a growing recognition of the benefits offered by non-invasive vaccine delivery.
Technological shifts are at the forefront of this market's evolution. Innovations in nanoparticle delivery systems, adjuvant technologies, and viral vector platforms are enabling the development of intranasal vaccines with enhanced immunogenicity and stability. The emphasis on mucosal immunity, which intranasal vaccines are uniquely positioned to stimulate, is a key trend, offering a promising avenue for protection against respiratory pathogens. Consumer preferences are increasingly leaning towards less painful and more accessible vaccination methods, thereby boosting the appeal of intranasal vaccines, particularly among pediatric and elderly populations, as well as individuals with needle phobia. Market penetration rates are expected to accelerate as regulatory approvals broaden and public awareness of intranasal vaccine benefits grows.
Competitive dynamics are becoming more pronounced, with both established pharmaceutical giants like Sanofi and emerging biopharmaceutical companies actively investing in research and development. This competition fosters a continuous cycle of innovation, leading to the introduction of novel vaccine candidates for a range of infectious diseases, including influenza and COVID-19. The increasing prevalence of respiratory infections globally, coupled with the ongoing need for effective pandemic preparedness, further solidifies the market's growth potential. The development of thermostable intranasal vaccines also presents significant opportunities, particularly in regions with limited cold chain infrastructure, thereby expanding access to critical immunizations. The market is ripe for strategic collaborations and partnerships aimed at accelerating product development and commercialization, further driving growth and market penetration. The estimated market size for 2025 is valued at over $XX million, with projections indicating a significant increase by 2033.
Dominant Markets & Segments in Intranasal Vaccine
The global intranasal vaccine market exhibits significant regional variations and segment dominance, driven by a complex interplay of healthcare infrastructure, public health policies, and disease prevalence. The Hospital segment is currently the dominant application, owing to the established infrastructure for vaccine administration, skilled healthcare professionals, and the centralized procurement of medical supplies. Hospitals are pivotal in the initial rollout and clinical trials of new intranasal vaccine technologies, particularly for high-risk populations and during public health emergencies.
Within the types of vaccines, Flu Vaccines have historically commanded a substantial market share due to their annual demand and widespread adoption. However, the COVID-19 pandemic has dramatically accelerated the development and demand for COVID-19 Vaccines, positioning them as a rapidly growing and increasingly dominant segment. The urgency to develop effective and accessible COVID-19 vaccines has spurred significant investment in intranasal delivery platforms, with several candidates showing promising results. The potential for self-administration and rapid mass vaccination campaigns further enhances the appeal of intranasal COVID-19 vaccines.
Geographically, North America and Europe are currently leading markets, characterized by high disposable incomes, advanced healthcare systems, and robust research and development capabilities. However, Asia-Pacific is emerging as a critical growth engine, driven by a large population, increasing healthcare expenditure, and the presence of major vaccine manufacturers like Serum Institute of India and Bharat Biotech, who are actively involved in intranasal vaccine development. Government initiatives promoting vaccination programs and the rising burden of infectious diseases in these regions are key growth drivers. Favorable policies supporting vaccine innovation and adoption, coupled with the increasing prevalence of target diseases such as influenza and the ongoing need for COVID-19 boosters, are expected to propel these segments and regions to new heights. The overall market is projected to reach over $XX million by 2033.
Intranasal Vaccine Product Analysis
Intranasal vaccine product development is characterized by a surge in innovative delivery systems designed to enhance immunogenicity and patient compliance. Innovations focus on needle-free administration, utilizing spray devices that deliver vaccines directly to the nasal mucosa. This approach leverages the natural immune defenses of the nasal passages, offering potential for robust mucosal immunity against respiratory pathogens. Key competitive advantages include ease of administration, suitability for mass vaccination campaigns, reduced risk of needle-stick injuries, and improved tolerability for diverse patient populations, including children and the elderly. Companies are actively developing intranasal formulations for influenza, COVID-19, and other infectious diseases, aiming to provide broader protection and more convenient vaccination options than traditional injectable vaccines, with an estimated XX million in development funding invested.
Key Drivers, Barriers & Challenges in Intranasal Vaccine
Key Drivers propelling the intranasal vaccine market include the growing demand for needle-free vaccination solutions driven by patient preference and reduced administration costs, technological advancements in delivery systems and adjuvant technologies enhancing efficacy, and the urgent need for rapid and scalable immunization strategies against emerging infectious diseases. Government initiatives and global health organizations' focus on improving vaccine accessibility and public health outcomes also serve as significant catalysts. The intrinsic ability of intranasal vaccines to induce mucosal immunity is a critical driver for combating respiratory infections.
Challenges impacting intranasal vaccine growth include the stringent regulatory hurdles for novel delivery systems, the need for extensive clinical trials to establish non-inferiority to injectable vaccines, and potential manufacturing complexities and scalability issues. Public perception and vaccine hesitancy, though potentially mitigated by the needle-free aspect, remain a concern requiring effective public education campaigns. Competitive pressures from established injectable vaccine markets and the high cost of research and development also present significant restraints, with estimated R&D investment reaching $XX million annually.
Growth Drivers in the Intranasal Vaccine Market
The intranasal vaccine market is experiencing robust growth driven by several key factors. Technological advancements in nasal spray devices, adjuvant formulations, and platform technologies are enabling the development of highly effective and stable intranasal vaccines. Economic factors, such as the potential for lower administration costs and wider reach in resource-limited settings, are also significant. Regulatory support and increasing government mandates for vaccination programs, coupled with a growing emphasis on pandemic preparedness, are creating a favorable environment. The inherent advantages of non-invasive delivery, such as improved patient compliance and the potential for enhanced mucosal immunity, are critical growth drivers, positioning intranasal vaccines as a complementary or alternative approach to traditional immunizations. The market is projected to see an influx of over $XX million in new investments by 2033.
Challenges Impacting Intranasal Vaccine Growth
Despite the promising outlook, the intranasal vaccine market faces several significant challenges. Regulatory complexities surrounding novel drug delivery systems require extensive validation and can lead to prolonged approval timelines, impacting market entry. Supply chain issues, including the manufacturing scalability of specialized nasal spray devices and the stability of formulations under varying environmental conditions, can pose logistical hurdles. Competitive pressures from the well-established injectable vaccine market, which enjoys widespread trust and infrastructure, necessitate substantial efforts to demonstrate clear advantages and gain market acceptance. Furthermore, the high cost of research and development for novel intranasal vaccine candidates, estimated at over $XX million per product, can deter smaller players and slow down innovation.
Key Players Shaping the Intranasal Vaccine Market
- Sanofi
- AstraZeneca
- Abbott
- GlaxoSmithKline
- Serum Institute of India
- Bharat Biotech
- Antares Pharma
- INJEX Pharma GmbH
- BioDiem
- PharmaJet
- Crossject
- Beijing Wantai Biopharmaceutical Co.,Ltd.
- Changchun Baike Company
Significant Intranasal Vaccine Industry Milestones
- 2019: Initiation of Phase I clinical trials for novel intranasal flu vaccine candidates by major pharmaceutical companies.
- 2020: Increased investment in intranasal vaccine R&D for COVID-19 following the global pandemic.
- 2021: Promising preclinical results reported for several intranasal COVID-19 vaccine candidates, demonstrating robust immune responses.
- 2022: Emergence of strategic partnerships between pharmaceutical giants and biotech firms specializing in nasal drug delivery technologies.
- 2023: Anticipated commencement of late-stage clinical trials for leading intranasal COVID-19 vaccines, with regulatory submissions expected shortly thereafter.
- 2024: First potential regulatory approvals for intranasal COVID-19 vaccines in select markets, marking a significant shift in vaccination strategies.
- 2025: Expansion of intranasal vaccine pipelines to include other infectious diseases, such as RSV and influenza, with estimated market penetration of XX%.
Future Outlook for Intranasal Vaccine Market
The future outlook for the intranasal vaccine market is exceptionally bright, driven by an ongoing commitment to innovation and a growing recognition of its therapeutic potential. Strategic opportunities lie in the development of universal vaccines for influenza and broad-spectrum antivirals delivered via the nasal route. Expansion into emerging markets with less developed healthcare infrastructure presents substantial untapped potential. The increasing focus on herd immunity and preventative healthcare strategies will further bolster demand. Continued investment in R&D, coupled with favorable regulatory pathways, is expected to drive significant market growth, with an estimated market size exceeding $XX million by 2033, offering substantial returns on investment.
Intranasal Vaccine Segmentation
-
1. Application
- 1.1. Hospital
- 1.2. Clinic
-
2. Types
- 2.1. Flu Vaccine
- 2.2. COVID-19 Vaccines
Intranasal Vaccine Segmentation By Geography
-
1. North America
- 1.1. United States
- 1.2. Canada
- 1.3. Mexico
-
2. South America
- 2.1. Brazil
- 2.2. Argentina
- 2.3. Rest of South America
-
3. Europe
- 3.1. United Kingdom
- 3.2. Germany
- 3.3. France
- 3.4. Italy
- 3.5. Spain
- 3.6. Russia
- 3.7. Benelux
- 3.8. Nordics
- 3.9. Rest of Europe
-
4. Middle East & Africa
- 4.1. Turkey
- 4.2. Israel
- 4.3. GCC
- 4.4. North Africa
- 4.5. South Africa
- 4.6. Rest of Middle East & Africa
-
5. Asia Pacific
- 5.1. China
- 5.2. India
- 5.3. Japan
- 5.4. South Korea
- 5.5. ASEAN
- 5.6. Oceania
- 5.7. Rest of Asia Pacific
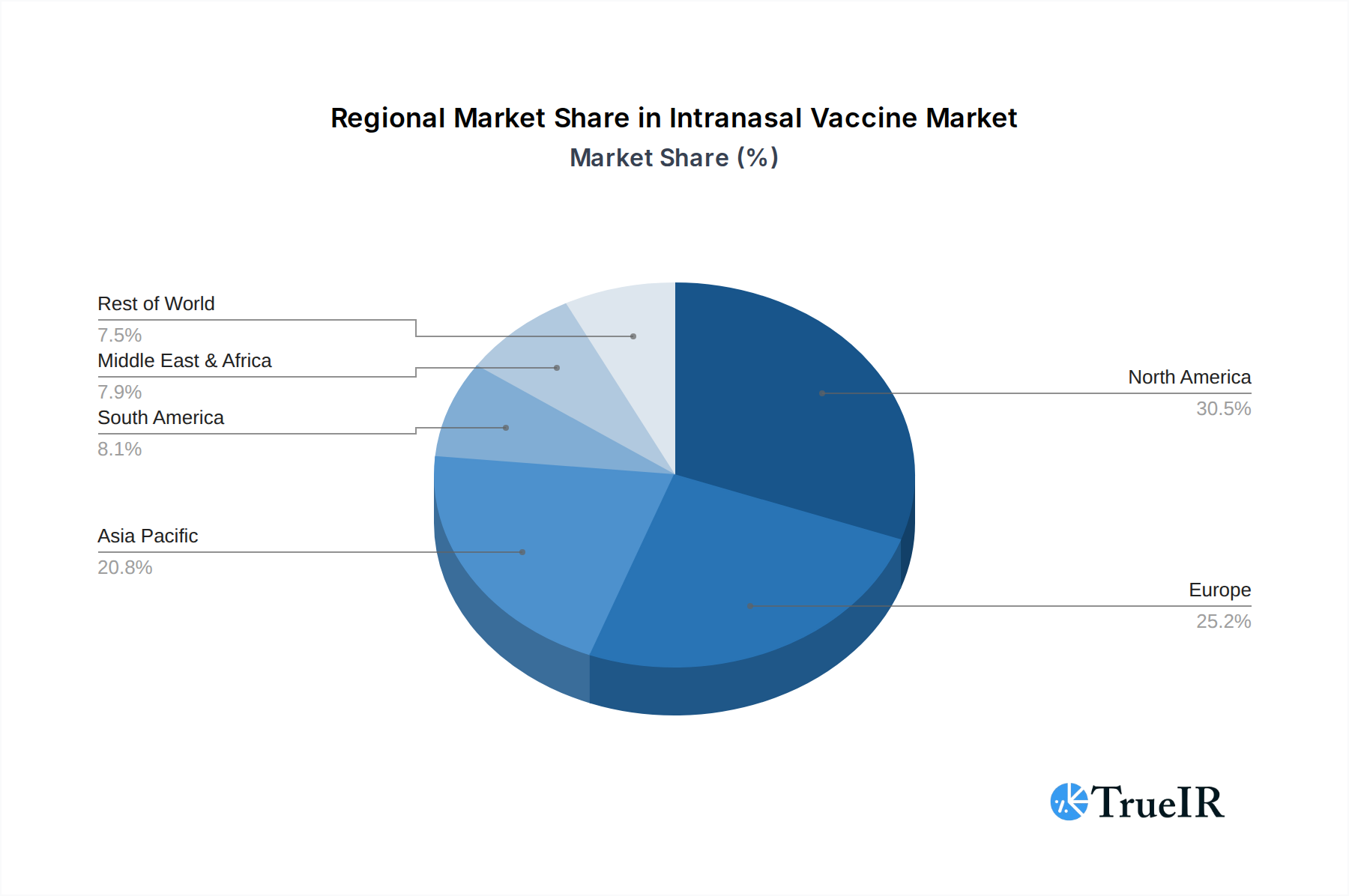
Intranasal Vaccine Regional Market Share

Geographic Coverage of Intranasal Vaccine
Intranasal Vaccine REPORT HIGHLIGHTS
| Aspects | Details |
|---|---|
| Study Period | 2020-2034 |
| Base Year | 2025 |
| Estimated Year | 2026 |
| Forecast Period | 2026-2034 |
| Historical Period | 2020-2025 |
| Growth Rate | CAGR of 5.2% from 2020-2034 |
| Segmentation |
|
Table of Contents
- 1. Introduction
- 1.1. Research Scope
- 1.2. Market Segmentation
- 1.3. Research Methodology
- 1.4. Definitions and Assumptions
- 2. Executive Summary
- 2.1. Introduction
- 3. Market Dynamics
- 3.1. Introduction
- 3.2. Market Drivers
- 3.3. Market Restrains
- 3.4. Market Trends
- 4. Market Factor Analysis
- 4.1. Porters Five Forces
- 4.2. Supply/Value Chain
- 4.3. PESTEL analysis
- 4.4. Market Entropy
- 4.5. Patent/Trademark Analysis
- 5. Global Intranasal Vaccine Analysis, Insights and Forecast, 2020-2032
- 5.1. Market Analysis, Insights and Forecast - by Application
- 5.1.1. Hospital
- 5.1.2. Clinic
- 5.2. Market Analysis, Insights and Forecast - by Types
- 5.2.1. Flu Vaccine
- 5.2.2. COVID-19 Vaccines
- 5.3. Market Analysis, Insights and Forecast - by Region
- 5.3.1. North America
- 5.3.2. South America
- 5.3.3. Europe
- 5.3.4. Middle East & Africa
- 5.3.5. Asia Pacific
- 5.1. Market Analysis, Insights and Forecast - by Application
- 6. North America Intranasal Vaccine Analysis, Insights and Forecast, 2020-2032
- 6.1. Market Analysis, Insights and Forecast - by Application
- 6.1.1. Hospital
- 6.1.2. Clinic
- 6.2. Market Analysis, Insights and Forecast - by Types
- 6.2.1. Flu Vaccine
- 6.2.2. COVID-19 Vaccines
- 6.1. Market Analysis, Insights and Forecast - by Application
- 7. South America Intranasal Vaccine Analysis, Insights and Forecast, 2020-2032
- 7.1. Market Analysis, Insights and Forecast - by Application
- 7.1.1. Hospital
- 7.1.2. Clinic
- 7.2. Market Analysis, Insights and Forecast - by Types
- 7.2.1. Flu Vaccine
- 7.2.2. COVID-19 Vaccines
- 7.1. Market Analysis, Insights and Forecast - by Application
- 8. Europe Intranasal Vaccine Analysis, Insights and Forecast, 2020-2032
- 8.1. Market Analysis, Insights and Forecast - by Application
- 8.1.1. Hospital
- 8.1.2. Clinic
- 8.2. Market Analysis, Insights and Forecast - by Types
- 8.2.1. Flu Vaccine
- 8.2.2. COVID-19 Vaccines
- 8.1. Market Analysis, Insights and Forecast - by Application
- 9. Middle East & Africa Intranasal Vaccine Analysis, Insights and Forecast, 2020-2032
- 9.1. Market Analysis, Insights and Forecast - by Application
- 9.1.1. Hospital
- 9.1.2. Clinic
- 9.2. Market Analysis, Insights and Forecast - by Types
- 9.2.1. Flu Vaccine
- 9.2.2. COVID-19 Vaccines
- 9.1. Market Analysis, Insights and Forecast - by Application
- 10. Asia Pacific Intranasal Vaccine Analysis, Insights and Forecast, 2020-2032
- 10.1. Market Analysis, Insights and Forecast - by Application
- 10.1.1. Hospital
- 10.1.2. Clinic
- 10.2. Market Analysis, Insights and Forecast - by Types
- 10.2.1. Flu Vaccine
- 10.2.2. COVID-19 Vaccines
- 10.1. Market Analysis, Insights and Forecast - by Application
- 11. Competitive Analysis
- 11.1. Global Market Share Analysis 2025
- 11.2. Company Profiles
- 11.2.1 Sanofi
- 11.2.1.1. Overview
- 11.2.1.2. Products
- 11.2.1.3. SWOT Analysis
- 11.2.1.4. Recent Developments
- 11.2.1.5. Financials (Based on Availability)
- 11.2.2 AstraZeneca
- 11.2.2.1. Overview
- 11.2.2.2. Products
- 11.2.2.3. SWOT Analysis
- 11.2.2.4. Recent Developments
- 11.2.2.5. Financials (Based on Availability)
- 11.2.3 Abbott
- 11.2.3.1. Overview
- 11.2.3.2. Products
- 11.2.3.3. SWOT Analysis
- 11.2.3.4. Recent Developments
- 11.2.3.5. Financials (Based on Availability)
- 11.2.4 GlaxoSmithKline
- 11.2.4.1. Overview
- 11.2.4.2. Products
- 11.2.4.3. SWOT Analysis
- 11.2.4.4. Recent Developments
- 11.2.4.5. Financials (Based on Availability)
- 11.2.5 Serum Institute of India
- 11.2.5.1. Overview
- 11.2.5.2. Products
- 11.2.5.3. SWOT Analysis
- 11.2.5.4. Recent Developments
- 11.2.5.5. Financials (Based on Availability)
- 11.2.6 Bharat Biotech
- 11.2.6.1. Overview
- 11.2.6.2. Products
- 11.2.6.3. SWOT Analysis
- 11.2.6.4. Recent Developments
- 11.2.6.5. Financials (Based on Availability)
- 11.2.7 Antares Pharma
- 11.2.7.1. Overview
- 11.2.7.2. Products
- 11.2.7.3. SWOT Analysis
- 11.2.7.4. Recent Developments
- 11.2.7.5. Financials (Based on Availability)
- 11.2.8 INJEX Pharma GmbH
- 11.2.8.1. Overview
- 11.2.8.2. Products
- 11.2.8.3. SWOT Analysis
- 11.2.8.4. Recent Developments
- 11.2.8.5. Financials (Based on Availability)
- 11.2.9 BioDiem
- 11.2.9.1. Overview
- 11.2.9.2. Products
- 11.2.9.3. SWOT Analysis
- 11.2.9.4. Recent Developments
- 11.2.9.5. Financials (Based on Availability)
- 11.2.10 PharmaJet and Crossject
- 11.2.10.1. Overview
- 11.2.10.2. Products
- 11.2.10.3. SWOT Analysis
- 11.2.10.4. Recent Developments
- 11.2.10.5. Financials (Based on Availability)
- 11.2.11 Beijing Wantai Biopharmaceutical Co.
- 11.2.11.1. Overview
- 11.2.11.2. Products
- 11.2.11.3. SWOT Analysis
- 11.2.11.4. Recent Developments
- 11.2.11.5. Financials (Based on Availability)
- 11.2.12 Ltd.
- 11.2.12.1. Overview
- 11.2.12.2. Products
- 11.2.12.3. SWOT Analysis
- 11.2.12.4. Recent Developments
- 11.2.12.5. Financials (Based on Availability)
- 11.2.13 Changchun Baike Company
- 11.2.13.1. Overview
- 11.2.13.2. Products
- 11.2.13.3. SWOT Analysis
- 11.2.13.4. Recent Developments
- 11.2.13.5. Financials (Based on Availability)
- 11.2.1 Sanofi
List of Figures
- Figure 1: Global Intranasal Vaccine Revenue Breakdown (undefined, %) by Region 2025 & 2033
- Figure 2: North America Intranasal Vaccine Revenue (undefined), by Application 2025 & 2033
- Figure 3: North America Intranasal Vaccine Revenue Share (%), by Application 2025 & 2033
- Figure 4: North America Intranasal Vaccine Revenue (undefined), by Types 2025 & 2033
- Figure 5: North America Intranasal Vaccine Revenue Share (%), by Types 2025 & 2033
- Figure 6: North America Intranasal Vaccine Revenue (undefined), by Country 2025 & 2033
- Figure 7: North America Intranasal Vaccine Revenue Share (%), by Country 2025 & 2033
- Figure 8: South America Intranasal Vaccine Revenue (undefined), by Application 2025 & 2033
- Figure 9: South America Intranasal Vaccine Revenue Share (%), by Application 2025 & 2033
- Figure 10: South America Intranasal Vaccine Revenue (undefined), by Types 2025 & 2033
- Figure 11: South America Intranasal Vaccine Revenue Share (%), by Types 2025 & 2033
- Figure 12: South America Intranasal Vaccine Revenue (undefined), by Country 2025 & 2033
- Figure 13: South America Intranasal Vaccine Revenue Share (%), by Country 2025 & 2033
- Figure 14: Europe Intranasal Vaccine Revenue (undefined), by Application 2025 & 2033
- Figure 15: Europe Intranasal Vaccine Revenue Share (%), by Application 2025 & 2033
- Figure 16: Europe Intranasal Vaccine Revenue (undefined), by Types 2025 & 2033
- Figure 17: Europe Intranasal Vaccine Revenue Share (%), by Types 2025 & 2033
- Figure 18: Europe Intranasal Vaccine Revenue (undefined), by Country 2025 & 2033
- Figure 19: Europe Intranasal Vaccine Revenue Share (%), by Country 2025 & 2033
- Figure 20: Middle East & Africa Intranasal Vaccine Revenue (undefined), by Application 2025 & 2033
- Figure 21: Middle East & Africa Intranasal Vaccine Revenue Share (%), by Application 2025 & 2033
- Figure 22: Middle East & Africa Intranasal Vaccine Revenue (undefined), by Types 2025 & 2033
- Figure 23: Middle East & Africa Intranasal Vaccine Revenue Share (%), by Types 2025 & 2033
- Figure 24: Middle East & Africa Intranasal Vaccine Revenue (undefined), by Country 2025 & 2033
- Figure 25: Middle East & Africa Intranasal Vaccine Revenue Share (%), by Country 2025 & 2033
- Figure 26: Asia Pacific Intranasal Vaccine Revenue (undefined), by Application 2025 & 2033
- Figure 27: Asia Pacific Intranasal Vaccine Revenue Share (%), by Application 2025 & 2033
- Figure 28: Asia Pacific Intranasal Vaccine Revenue (undefined), by Types 2025 & 2033
- Figure 29: Asia Pacific Intranasal Vaccine Revenue Share (%), by Types 2025 & 2033
- Figure 30: Asia Pacific Intranasal Vaccine Revenue (undefined), by Country 2025 & 2033
- Figure 31: Asia Pacific Intranasal Vaccine Revenue Share (%), by Country 2025 & 2033
List of Tables
- Table 1: Global Intranasal Vaccine Revenue undefined Forecast, by Application 2020 & 2033
- Table 2: Global Intranasal Vaccine Revenue undefined Forecast, by Types 2020 & 2033
- Table 3: Global Intranasal Vaccine Revenue undefined Forecast, by Region 2020 & 2033
- Table 4: Global Intranasal Vaccine Revenue undefined Forecast, by Application 2020 & 2033
- Table 5: Global Intranasal Vaccine Revenue undefined Forecast, by Types 2020 & 2033
- Table 6: Global Intranasal Vaccine Revenue undefined Forecast, by Country 2020 & 2033
- Table 7: United States Intranasal Vaccine Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 8: Canada Intranasal Vaccine Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 9: Mexico Intranasal Vaccine Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 10: Global Intranasal Vaccine Revenue undefined Forecast, by Application 2020 & 2033
- Table 11: Global Intranasal Vaccine Revenue undefined Forecast, by Types 2020 & 2033
- Table 12: Global Intranasal Vaccine Revenue undefined Forecast, by Country 2020 & 2033
- Table 13: Brazil Intranasal Vaccine Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 14: Argentina Intranasal Vaccine Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 15: Rest of South America Intranasal Vaccine Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 16: Global Intranasal Vaccine Revenue undefined Forecast, by Application 2020 & 2033
- Table 17: Global Intranasal Vaccine Revenue undefined Forecast, by Types 2020 & 2033
- Table 18: Global Intranasal Vaccine Revenue undefined Forecast, by Country 2020 & 2033
- Table 19: United Kingdom Intranasal Vaccine Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 20: Germany Intranasal Vaccine Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 21: France Intranasal Vaccine Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 22: Italy Intranasal Vaccine Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 23: Spain Intranasal Vaccine Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 24: Russia Intranasal Vaccine Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 25: Benelux Intranasal Vaccine Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 26: Nordics Intranasal Vaccine Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 27: Rest of Europe Intranasal Vaccine Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 28: Global Intranasal Vaccine Revenue undefined Forecast, by Application 2020 & 2033
- Table 29: Global Intranasal Vaccine Revenue undefined Forecast, by Types 2020 & 2033
- Table 30: Global Intranasal Vaccine Revenue undefined Forecast, by Country 2020 & 2033
- Table 31: Turkey Intranasal Vaccine Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 32: Israel Intranasal Vaccine Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 33: GCC Intranasal Vaccine Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 34: North Africa Intranasal Vaccine Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 35: South Africa Intranasal Vaccine Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 36: Rest of Middle East & Africa Intranasal Vaccine Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 37: Global Intranasal Vaccine Revenue undefined Forecast, by Application 2020 & 2033
- Table 38: Global Intranasal Vaccine Revenue undefined Forecast, by Types 2020 & 2033
- Table 39: Global Intranasal Vaccine Revenue undefined Forecast, by Country 2020 & 2033
- Table 40: China Intranasal Vaccine Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 41: India Intranasal Vaccine Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 42: Japan Intranasal Vaccine Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 43: South Korea Intranasal Vaccine Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 44: ASEAN Intranasal Vaccine Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 45: Oceania Intranasal Vaccine Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 46: Rest of Asia Pacific Intranasal Vaccine Revenue (undefined) Forecast, by Application 2020 & 2033
Frequently Asked Questions
1. What is the projected Compound Annual Growth Rate (CAGR) of the Intranasal Vaccine?
The projected CAGR is approximately 5.2%.
2. Which companies are prominent players in the Intranasal Vaccine?
Key companies in the market include Sanofi, AstraZeneca, Abbott, GlaxoSmithKline, Serum Institute of India, Bharat Biotech, Antares Pharma, INJEX Pharma GmbH, BioDiem, PharmaJet and Crossject, Beijing Wantai Biopharmaceutical Co., Ltd., Changchun Baike Company.
3. What are the main segments of the Intranasal Vaccine?
The market segments include Application, Types.
4. Can you provide details about the market size?
The market size is estimated to be USD XXX N/A as of 2022.
5. What are some drivers contributing to market growth?
N/A
6. What are the notable trends driving market growth?
N/A
7. Are there any restraints impacting market growth?
N/A
8. Can you provide examples of recent developments in the market?
N/A
9. What pricing options are available for accessing the report?
Pricing options include single-user, multi-user, and enterprise licenses priced at USD 2900.00, USD 4350.00, and USD 5800.00 respectively.
10. Is the market size provided in terms of value or volume?
The market size is provided in terms of value, measured in N/A.
11. Are there any specific market keywords associated with the report?
Yes, the market keyword associated with the report is "Intranasal Vaccine," which aids in identifying and referencing the specific market segment covered.
12. How do I determine which pricing option suits my needs best?
The pricing options vary based on user requirements and access needs. Individual users may opt for single-user licenses, while businesses requiring broader access may choose multi-user or enterprise licenses for cost-effective access to the report.
13. Are there any additional resources or data provided in the Intranasal Vaccine report?
While the report offers comprehensive insights, it's advisable to review the specific contents or supplementary materials provided to ascertain if additional resources or data are available.
14. How can I stay updated on further developments or reports in the Intranasal Vaccine?
To stay informed about further developments, trends, and reports in the Intranasal Vaccine, consider subscribing to industry newsletters, following relevant companies and organizations, or regularly checking reputable industry news sources and publications.
Methodology
Step 1 - Identification of Relevant Samples Size from Population Database



Step 2 - Approaches for Defining Global Market Size (Value, Volume* & Price*)

Note*: In applicable scenarios
Step 3 - Data Sources
Primary Research
- Web Analytics
- Survey Reports
- Research Institute
- Latest Research Reports
- Opinion Leaders
Secondary Research
- Annual Reports
- White Paper
- Latest Press Release
- Industry Association
- Paid Database
- Investor Presentations

Step 4 - Data Triangulation
Involves using different sources of information in order to increase the validity of a study
These sources are likely to be stakeholders in a program - participants, other researchers, program staff, other community members, and so on.
Then we put all data in single framework & apply various statistical tools to find out the dynamic on the market.
During the analysis stage, feedback from the stakeholder groups would be compared to determine areas of agreement as well as areas of divergence


