Key Insights
The global Neuro Intervention Intermediate Catheter market is poised for robust expansion, with an estimated market size of $2.82 billion in 2025. This growth is underpinned by a projected Compound Annual Growth Rate (CAGR) of 4.7% from 2019 to 2033, indicating sustained demand and increasing adoption of these critical medical devices. A primary driver for this expansion is the escalating global burden of neurological disorders, including stroke, aneurysms, and arteriovenous malformations (AVMs). As the aging population continues to grow worldwide, so does the incidence of these conditions, necessitating advanced minimally invasive neurovascular interventions. Furthermore, significant advancements in catheter technology, characterized by improved trackability, navigability, and smaller profiles, are enabling neurosurgeons and interventional neuroradiologists to perform more complex procedures with greater precision and safety, further fueling market growth.
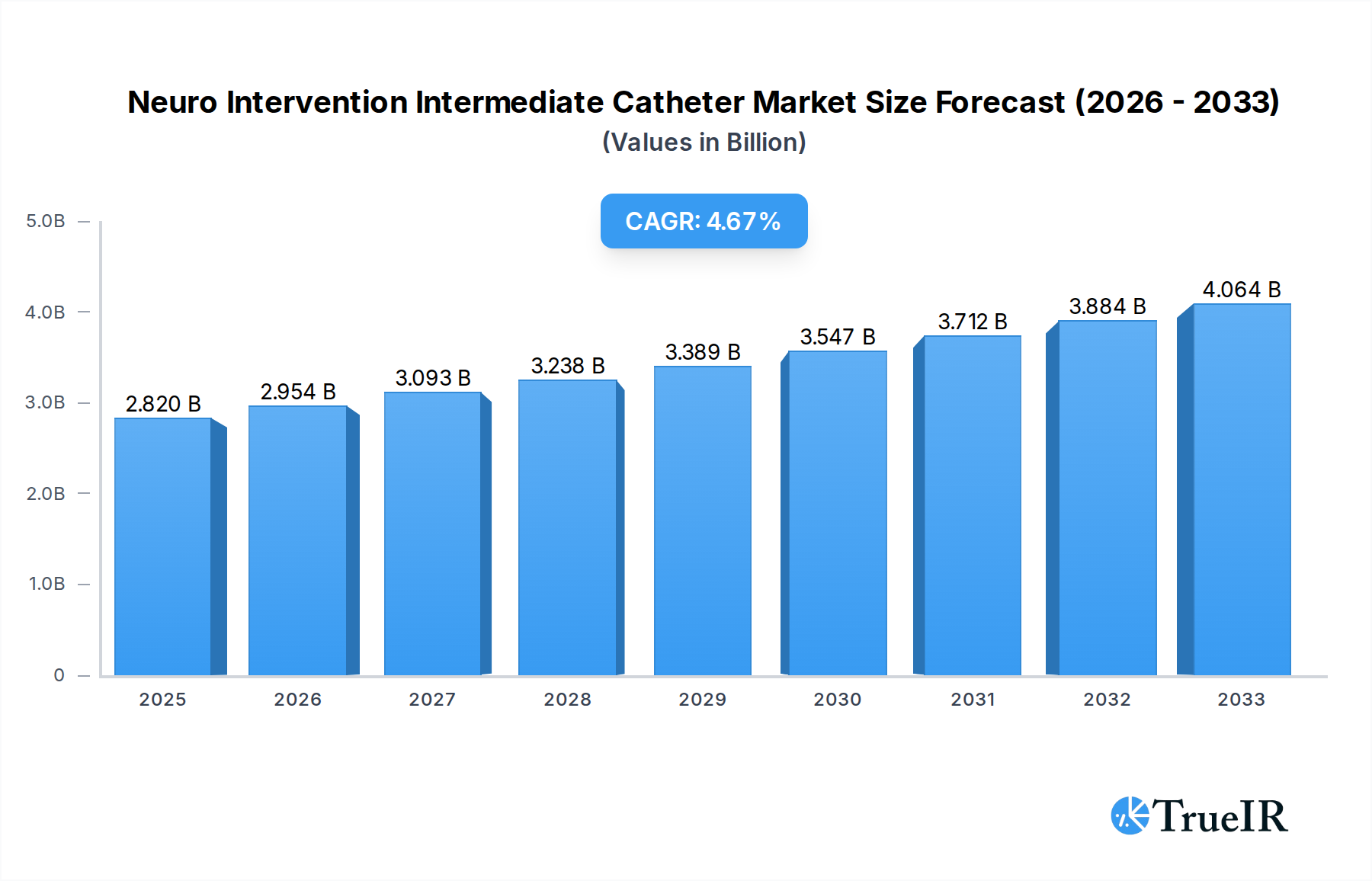
Neuro Intervention Intermediate Catheter Market Size (In Billion)

The market's trajectory is also being shaped by increasing healthcare expenditure, particularly in emerging economies, and a growing preference for minimally invasive surgical techniques over traditional open surgeries due to reduced patient recovery times and hospital stays. Key trends include the development of next-generation catheters with enhanced features like advanced steering capabilities, integrated imaging, and drug-eluting properties to prevent restenosis. While the market demonstrates strong growth potential, certain restraints, such as the high cost of advanced neuro intervention procedures and the need for specialized training for medical professionals, could temper the pace of adoption in some regions. Nonetheless, the overwhelming benefits of these devices in treating debilitating neurological conditions and improving patient outcomes are expected to drive sustained market performance across diverse applications, from large hospitals to specialized clinics.
Neuro Intervention Intermediate Catheter Company Market Share

Here is a dynamic, SEO-optimized report description for the Neuro Intervention Intermediate Catheter Market, designed for immediate use without modification.
Neuro Intervention Intermediate Catheter Market Structure & Competitive Landscape
The neuro intervention intermediate catheter market is characterized by a moderately concentrated competitive landscape, with established global players vying for market share alongside emerging regional manufacturers. Key innovation drivers include advancements in catheter design for improved trackability, deliverability, and lesion crossing capabilities, alongside material science innovations for enhanced biocompatibility and pushability. Regulatory impacts, primarily from bodies like the FDA and EMA, significantly shape market entry and product approval processes, influencing the pace of innovation and market access. Product substitutes, while limited in direct neuro interventional applications, can include more traditional surgical approaches in specific complex cases. End-user segmentation is heavily skewed towards hospitals, accounting for approximately 95% of the market, followed by specialized neuro-interventional clinics (4%) and other healthcare settings (1%). Mergers and acquisition (M&A) trends have been moderate, with larger companies strategically acquiring smaller innovative firms to expand their product portfolios and technological capabilities. Recent M&A activity has focused on acquiring platforms with advanced imaging compatibility and microcatheter technologies. For instance, the market is estimated to have seen approximately 5 major M&A transactions in the historical period, with an estimated cumulative deal value of $1.5 billion. The concentration ratio among the top four players is estimated to be around 60%.
Neuro Intervention Intermediate Catheter Market Trends & Opportunities
The global neuro intervention intermediate catheter market is poised for substantial expansion, driven by a confluence of escalating demand for minimally invasive neurovascular treatments and continuous technological innovation. The market size is projected to grow from an estimated $2.3 billion in the base year 2025 to a formidable $6.8 billion by the end of the forecast period in 2033, exhibiting a robust Compound Annual Growth Rate (CAGR) of approximately 14.5% from 2025 to 2033. This significant growth trajectory is underpinned by the increasing prevalence of neurological disorders such as stroke, aneurysms, and arteriovenous malformations (AVMs), which necessitate advanced neuro interventional procedures. Technological shifts are at the forefront of market dynamics, with a relentless pursuit of catheters offering enhanced precision, navigation, and therapeutic delivery. The development of microcatheters with improved coaxial designs, advanced steerability, and compatibility with various imaging modalities (e.g., MRI, CT angiography) is a key trend. Furthermore, advancements in hydrophilic coatings and radiopaque markers are crucial for facilitating complex procedures and ensuring optimal visualization during interventions. Consumer preferences are increasingly tilting towards minimally invasive techniques due to their inherent benefits, including reduced patient trauma, shorter recovery times, and lower healthcare costs compared to open surgery. This preference directly fuels the demand for sophisticated neuro intervention intermediate catheters. Competitive dynamics are intensifying, with both established medical device giants and agile startups investing heavily in research and development. Companies are focusing on developing differentiated products that address specific procedural challenges, such as navigating tortuous vasculature and delivering complex therapeutic agents or devices. The market penetration rate for advanced neuro interventional procedures is steadily increasing, particularly in developed economies, and is expected to see significant growth in emerging markets as healthcare infrastructure and awareness improve. The adoption of new technologies, such as resheathable catheters and those with integrated micro-guidewires, is also a significant trend, offering procedural efficiency and improved patient outcomes. The integration of AI-powered navigation assistance and real-time imaging feedback is an emerging frontier that could further revolutionize the market.
Dominant Markets & Segments in Neuro Intervention Intermediate Catheter
The neuro intervention intermediate catheter market's dominance is shaped by key regional and segment-specific factors. Application-wise, the Hospital segment is by far the most dominant, accounting for an estimated 95% of the market share. This dominance is directly attributable to the comprehensive infrastructure, specialized surgical teams, and advanced diagnostic and therapeutic equipment available in hospital settings, which are essential for complex neuro interventional procedures. The availability of dedicated neuro-interventional suites within hospitals further solidifies this segment's leadership. Clinics, while growing, represent a smaller but important segment at approximately 4%, often focusing on outpatient diagnostics and less complex interventional procedures. The "Others" category, encompassing research institutions and specialized centers, comprises the remaining 1% but plays a crucial role in pioneering new techniques and technologies.
From a Type perspective, both Straight Tip and Pre-bent Tip catheters hold significant market share, catering to different procedural needs. Straight tip catheters offer versatility for initial navigation and guidewire manipulation, while pre-bent tip catheters are crucial for negotiating complex anatomical curves and tortuous vessels encountered in neurovascular anatomy. The market penetration of each type is influenced by the specific pathologies being treated and the surgeon's preference, but both are integral to the neuro intervention workflow.
Regionally, North America and Europe currently dominate the neuro intervention intermediate catheter market, driven by high healthcare spending, advanced technological adoption, and a high prevalence of neurological diseases. The burgeoning healthcare infrastructure and increasing patient awareness in the Asia Pacific region, particularly in countries like China and India, present the fastest-growing markets. Key growth drivers in these dominant and emerging regions include:
- Infrastructure Development: Significant investments in upgrading hospital infrastructure and establishing specialized neuro-interventional centers.
- Policy and Reimbursement: Favorable government policies promoting advanced medical treatments and robust reimbursement frameworks that encourage the adoption of minimally invasive procedures.
- Technological Adoption: A strong appetite for adopting the latest technological advancements in medical devices, including sophisticated catheters.
- Prevalence of Neurological Disorders: A high and growing incidence of conditions like ischemic and hemorrhagic stroke, aneurysms, and AVMs, which are primary indications for neuro interventional procedures.
- Skilled Workforce: A growing pool of highly trained neurologists, interventional neuroradiologists, and neurosurgeons proficient in performing these complex procedures.
The synergistic interplay of these factors ensures sustained demand and market expansion for neuro intervention intermediate catheters across these key segments and regions.
Neuro Intervention Intermediate Catheter Product Analysis
Neuro intervention intermediate catheters are engineered for precise navigation through delicate cerebrovascular anatomy. Innovations focus on enhancing pushability, torque control, and lubricity for seamless advancement to target lesions. Advanced materials like high-strength polymers and composite shafts enable kink resistance and optimal deliverability of adjunctive devices. Competitive advantages lie in features such as hydrophilic coatings for reduced friction, radiopaque markers for enhanced fluoroscopic visualization, and tailored tip designs (straight or pre-bent) to navigate tortuous vasculature. Microcatheter technology, in particular, is a significant area of development, enabling access to distal lesions previously unreachable.
Key Drivers, Barriers & Challenges in Neuro Intervention Intermediate Catheter
Key Drivers:
The neuro intervention intermediate catheter market is propelled by the escalating global burden of neurological disorders, particularly stroke, aneurysms, and AVMs, driving demand for minimally invasive treatments. Technological advancements in catheter design, material science, and imaging compatibility are key catalysts, enabling greater precision and improved patient outcomes. The increasing preference for minimally invasive procedures due to shorter recovery times and reduced patient trauma further fuels market growth. Favorable reimbursement policies and government initiatives promoting advanced healthcare technologies also play a significant role.
Barriers & Challenges:
High research and development costs associated with innovative catheter technologies and stringent regulatory approval processes pose significant barriers. The complexity of neurovascular anatomy requires highly skilled neurointerventionalists, limiting widespread adoption in regions with a shortage of trained professionals. Intense competition among established players and the threat of product obsolescence due to rapid technological evolution also present challenges. Furthermore, supply chain disruptions and the cost of advanced materials can impact manufacturing and pricing.
Growth Drivers in the Neuro Intervention Intermediate Catheter Market
The growth of the neuro intervention intermediate catheter market is significantly propelled by the increasing global prevalence of cerebrovascular diseases such as stroke, cerebral aneurysms, and arteriovenous malformations. This escalating disease burden directly translates into a higher demand for effective and minimally invasive treatment options. Technological innovation is another paramount driver; continuous advancements in catheter design, including improved trackability, steerability, and deliverability, allow for more precise navigation within the intricate cerebrovascular system. The development of advanced materials and hydrophilic coatings further enhances procedural efficiency and patient safety. Furthermore, the growing preference among patients and healthcare providers for minimally invasive procedures, owing to reduced patient trauma, shorter hospital stays, and faster recovery times, significantly boosts market expansion. Supportive government initiatives and favorable reimbursement policies in many regions also encourage the adoption of these advanced interventional techniques.
Challenges Impacting Neuro Intervention Intermediate Catheter Growth
Several challenges impact the growth trajectory of the neuro intervention intermediate catheter market. The extremely high cost of research and development for novel catheter technologies, coupled with the rigorous and lengthy regulatory approval processes mandated by health authorities like the FDA and EMA, can act as significant barriers to entry and market acceleration. The specialized nature of neurointerventional procedures necessitates a highly skilled and trained workforce, and a global shortage of neurointerventionalists and interventional neuroradiologists can limit the widespread adoption and utilization of these advanced catheters in certain regions. Intense competition among established medical device manufacturers, leading to price pressures and the constant threat of product obsolescence due to rapid technological advancements, also poses a considerable challenge. Moreover, potential disruptions in the global supply chain for critical raw materials and manufacturing components can impact production volumes and costs, affecting market dynamics.
Key Players Shaping the Neuro Intervention Intermediate Catheter Market
- Medtronic
- Stryker
- Penumbra
- MicroVention
- TJWY Medical
- Nanjing Precision Medical
- Pejia Medical
Significant Neuro Intervention Intermediate Catheter Industry Milestones
- 2019: Introduction of next-generation hydrophilic coatings enhancing lubricity and reducing friction in neurovascular navigation.
- 2020: Launch of advanced microcatheters with enhanced torque control for improved manipulation in tortuous anatomy.
- 2021: Development of novel braided catheter shaft designs offering superior pushability and kink resistance.
- 2022: Increased focus on catheters with improved compatibility for advanced imaging modalities like cone-beam CT and rotational angiography.
- 2023: Emergence of bioresorbable materials in catheter coatings for potential future applications.
- 2024: Significant advancements in steerable catheter tip designs for enhanced maneuverability in complex neurovascular structures.
Future Outlook for Neuro Intervention Intermediate Catheter Market
The future outlook for the neuro intervention intermediate catheter market is exceptionally bright, driven by persistent innovation and growing demand for minimally invasive neurological treatments. Continued advancements in catheter materials, design, and enhanced imaging integration will unlock new procedural possibilities and improve patient outcomes. The expansion of healthcare infrastructure and increasing awareness of neuro interventional therapies in emerging economies represent substantial growth catalysts. Strategic collaborations and acquisitions are expected to further consolidate the market and accelerate the development of next-generation technologies. The market is poised for sustained growth, driven by the imperative to effectively treat an aging global population facing an increasing incidence of neurovascular conditions.
Neuro Intervention Intermediate Catheter Segmentation
-
1. Application
- 1.1. Hospital
- 1.2. Clinics
- 1.3. Others
-
2. Types
- 2.1. Straight Tip
- 2.2. Pre-bent Tip
Neuro Intervention Intermediate Catheter Segmentation By Geography
-
1. North America
- 1.1. United States
- 1.2. Canada
- 1.3. Mexico
-
2. South America
- 2.1. Brazil
- 2.2. Argentina
- 2.3. Rest of South America
-
3. Europe
- 3.1. United Kingdom
- 3.2. Germany
- 3.3. France
- 3.4. Italy
- 3.5. Spain
- 3.6. Russia
- 3.7. Benelux
- 3.8. Nordics
- 3.9. Rest of Europe
-
4. Middle East & Africa
- 4.1. Turkey
- 4.2. Israel
- 4.3. GCC
- 4.4. North Africa
- 4.5. South Africa
- 4.6. Rest of Middle East & Africa
-
5. Asia Pacific
- 5.1. China
- 5.2. India
- 5.3. Japan
- 5.4. South Korea
- 5.5. ASEAN
- 5.6. Oceania
- 5.7. Rest of Asia Pacific
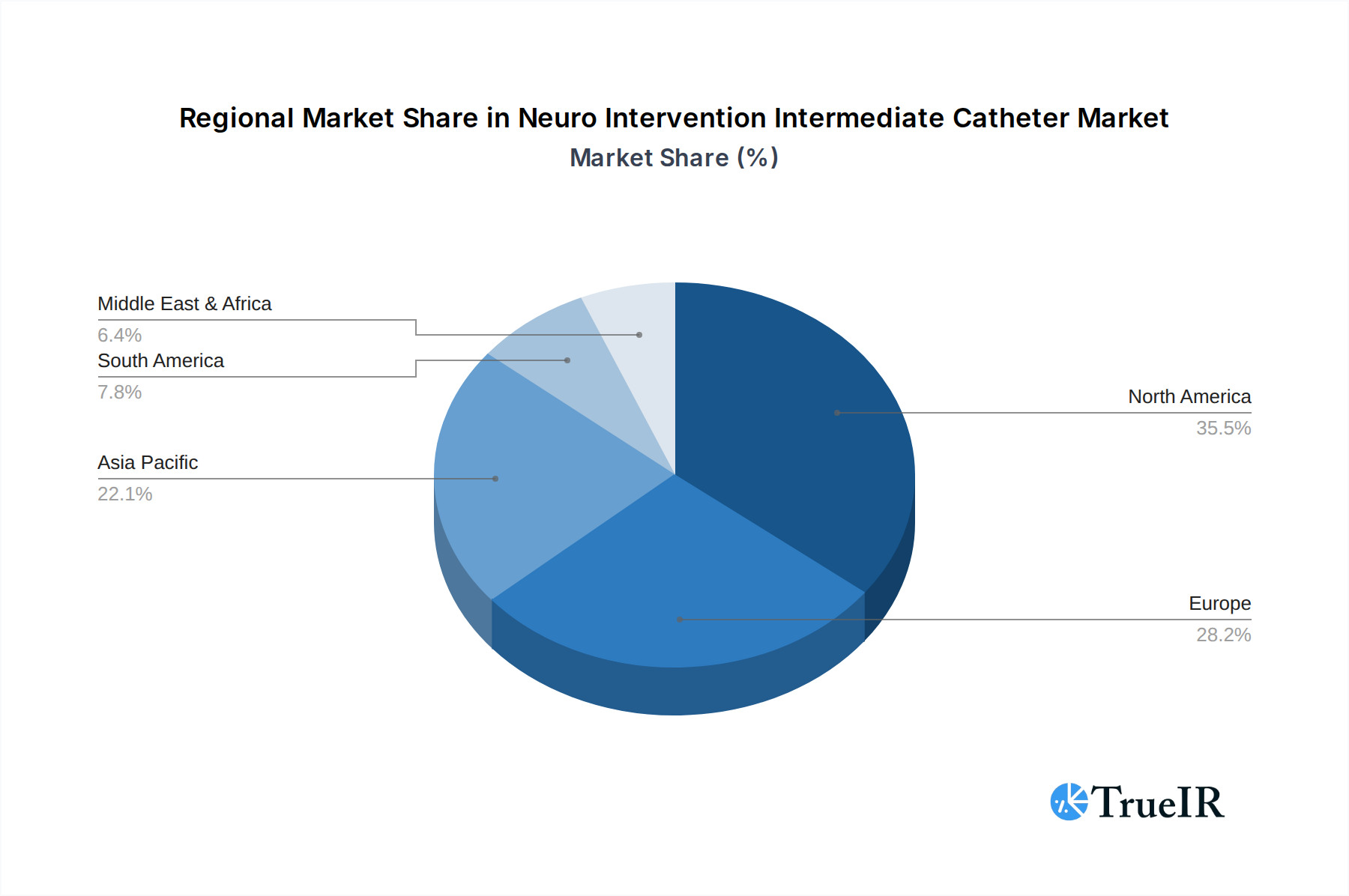
Neuro Intervention Intermediate Catheter Regional Market Share

Geographic Coverage of Neuro Intervention Intermediate Catheter
Neuro Intervention Intermediate Catheter REPORT HIGHLIGHTS
| Aspects | Details |
|---|---|
| Study Period | 2020-2034 |
| Base Year | 2025 |
| Estimated Year | 2026 |
| Forecast Period | 2026-2034 |
| Historical Period | 2020-2025 |
| Growth Rate | CAGR of 4.7% from 2020-2034 |
| Segmentation |
|
Table of Contents
- 1. Introduction
- 1.1. Research Scope
- 1.2. Market Segmentation
- 1.3. Research Objective
- 1.4. Definitions and Assumptions
- 2. Executive Summary
- 2.1. Market Snapshot
- 3. Market Dynamics
- 3.1. Market Drivers
- 3.2. Market Restrains
- 3.3. Market Trends
- 3.4. Market Opportunities
- 4. Market Factor Analysis
- 4.1. Porters Five Forces
- 4.1.1. Bargaining Power of Suppliers
- 4.1.2. Bargaining Power of Buyers
- 4.1.3. Threat of New Entrants
- 4.1.4. Threat of Substitutes
- 4.1.5. Competitive Rivalry
- 4.2. PESTEL analysis
- 4.3. BCG Analysis
- 4.3.1. Stars (High Growth, High Market Share)
- 4.3.2. Cash Cows (Low Growth, High Market Share)
- 4.3.3. Question Mark (High Growth, Low Market Share)
- 4.3.4. Dogs (Low Growth, Low Market Share)
- 4.4. Ansoff Matrix Analysis
- 4.5. Supply Chain Analysis
- 4.6. Regulatory Landscape
- 4.7. Current Market Potential and Opportunity Assessment (TAM–SAM–SOM Framework)
- 4.8. TIR Analyst Note
- 4.1. Porters Five Forces
- 5. Market Analysis, Insights and Forecast 2021-2033
- 5.1. Market Analysis, Insights and Forecast - by Application
- 5.1.1. Hospital
- 5.1.2. Clinics
- 5.1.3. Others
- 5.2. Market Analysis, Insights and Forecast - by Types
- 5.2.1. Straight Tip
- 5.2.2. Pre-bent Tip
- 5.3. Market Analysis, Insights and Forecast - by Region
- 5.3.1. North America
- 5.3.2. South America
- 5.3.3. Europe
- 5.3.4. Middle East & Africa
- 5.3.5. Asia Pacific
- 5.1. Market Analysis, Insights and Forecast - by Application
- 6. Global Neuro Intervention Intermediate Catheter Analysis, Insights and Forecast, 2021-2033
- 6.1. Market Analysis, Insights and Forecast - by Application
- 6.1.1. Hospital
- 6.1.2. Clinics
- 6.1.3. Others
- 6.2. Market Analysis, Insights and Forecast - by Types
- 6.2.1. Straight Tip
- 6.2.2. Pre-bent Tip
- 6.1. Market Analysis, Insights and Forecast - by Application
- 7. North America Neuro Intervention Intermediate Catheter Analysis, Insights and Forecast, 2020-2032
- 7.1. Market Analysis, Insights and Forecast - by Application
- 7.1.1. Hospital
- 7.1.2. Clinics
- 7.1.3. Others
- 7.2. Market Analysis, Insights and Forecast - by Types
- 7.2.1. Straight Tip
- 7.2.2. Pre-bent Tip
- 7.1. Market Analysis, Insights and Forecast - by Application
- 8. South America Neuro Intervention Intermediate Catheter Analysis, Insights and Forecast, 2020-2032
- 8.1. Market Analysis, Insights and Forecast - by Application
- 8.1.1. Hospital
- 8.1.2. Clinics
- 8.1.3. Others
- 8.2. Market Analysis, Insights and Forecast - by Types
- 8.2.1. Straight Tip
- 8.2.2. Pre-bent Tip
- 8.1. Market Analysis, Insights and Forecast - by Application
- 9. Europe Neuro Intervention Intermediate Catheter Analysis, Insights and Forecast, 2020-2032
- 9.1. Market Analysis, Insights and Forecast - by Application
- 9.1.1. Hospital
- 9.1.2. Clinics
- 9.1.3. Others
- 9.2. Market Analysis, Insights and Forecast - by Types
- 9.2.1. Straight Tip
- 9.2.2. Pre-bent Tip
- 9.1. Market Analysis, Insights and Forecast - by Application
- 10. Middle East & Africa Neuro Intervention Intermediate Catheter Analysis, Insights and Forecast, 2020-2032
- 10.1. Market Analysis, Insights and Forecast - by Application
- 10.1.1. Hospital
- 10.1.2. Clinics
- 10.1.3. Others
- 10.2. Market Analysis, Insights and Forecast - by Types
- 10.2.1. Straight Tip
- 10.2.2. Pre-bent Tip
- 10.1. Market Analysis, Insights and Forecast - by Application
- 11. Asia Pacific Neuro Intervention Intermediate Catheter Analysis, Insights and Forecast, 2020-2032
- 11.1. Market Analysis, Insights and Forecast - by Application
- 11.1.1. Hospital
- 11.1.2. Clinics
- 11.1.3. Others
- 11.2. Market Analysis, Insights and Forecast - by Types
- 11.2.1. Straight Tip
- 11.2.2. Pre-bent Tip
- 11.1. Market Analysis, Insights and Forecast - by Application
- 12. Competitive Analysis
- 12.1. Company Profiles
- 12.1.1 Medtronic
- 12.1.1.1. Company Overview
- 12.1.1.2. Products
- 12.1.1.3. Company Financials
- 12.1.1.4. SWOT Analysis
- 12.1.2 Stryker
- 12.1.2.1. Company Overview
- 12.1.2.2. Products
- 12.1.2.3. Company Financials
- 12.1.2.4. SWOT Analysis
- 12.1.3 Penumbra
- 12.1.3.1. Company Overview
- 12.1.3.2. Products
- 12.1.3.3. Company Financials
- 12.1.3.4. SWOT Analysis
- 12.1.4 MicroVention
- 12.1.4.1. Company Overview
- 12.1.4.2. Products
- 12.1.4.3. Company Financials
- 12.1.4.4. SWOT Analysis
- 12.1.5 TJWY Medical
- 12.1.5.1. Company Overview
- 12.1.5.2. Products
- 12.1.5.3. Company Financials
- 12.1.5.4. SWOT Analysis
- 12.1.6 Nanjing Precision Medical
- 12.1.6.1. Company Overview
- 12.1.6.2. Products
- 12.1.6.3. Company Financials
- 12.1.6.4. SWOT Analysis
- 12.1.7 Pejia Medical
- 12.1.7.1. Company Overview
- 12.1.7.2. Products
- 12.1.7.3. Company Financials
- 12.1.7.4. SWOT Analysis
- 12.1.1 Medtronic
- 12.2. Market Entropy
- 12.2.1 Company's Key Areas Served
- 12.2.2 Recent Developments
- 12.3. Company Market Share Analysis 2025
- 12.3.1 Top 5 Companies Market Share Analysis
- 12.3.2 Top 3 Companies Market Share Analysis
- 12.4. List of Potential Customers
- 13. Research Methodology
List of Figures
- Figure 1: Global Neuro Intervention Intermediate Catheter Revenue Breakdown (billion, %) by Region 2025 & 2033
- Figure 2: North America Neuro Intervention Intermediate Catheter Revenue (billion), by Application 2025 & 2033
- Figure 3: North America Neuro Intervention Intermediate Catheter Revenue Share (%), by Application 2025 & 2033
- Figure 4: North America Neuro Intervention Intermediate Catheter Revenue (billion), by Types 2025 & 2033
- Figure 5: North America Neuro Intervention Intermediate Catheter Revenue Share (%), by Types 2025 & 2033
- Figure 6: North America Neuro Intervention Intermediate Catheter Revenue (billion), by Country 2025 & 2033
- Figure 7: North America Neuro Intervention Intermediate Catheter Revenue Share (%), by Country 2025 & 2033
- Figure 8: South America Neuro Intervention Intermediate Catheter Revenue (billion), by Application 2025 & 2033
- Figure 9: South America Neuro Intervention Intermediate Catheter Revenue Share (%), by Application 2025 & 2033
- Figure 10: South America Neuro Intervention Intermediate Catheter Revenue (billion), by Types 2025 & 2033
- Figure 11: South America Neuro Intervention Intermediate Catheter Revenue Share (%), by Types 2025 & 2033
- Figure 12: South America Neuro Intervention Intermediate Catheter Revenue (billion), by Country 2025 & 2033
- Figure 13: South America Neuro Intervention Intermediate Catheter Revenue Share (%), by Country 2025 & 2033
- Figure 14: Europe Neuro Intervention Intermediate Catheter Revenue (billion), by Application 2025 & 2033
- Figure 15: Europe Neuro Intervention Intermediate Catheter Revenue Share (%), by Application 2025 & 2033
- Figure 16: Europe Neuro Intervention Intermediate Catheter Revenue (billion), by Types 2025 & 2033
- Figure 17: Europe Neuro Intervention Intermediate Catheter Revenue Share (%), by Types 2025 & 2033
- Figure 18: Europe Neuro Intervention Intermediate Catheter Revenue (billion), by Country 2025 & 2033
- Figure 19: Europe Neuro Intervention Intermediate Catheter Revenue Share (%), by Country 2025 & 2033
- Figure 20: Middle East & Africa Neuro Intervention Intermediate Catheter Revenue (billion), by Application 2025 & 2033
- Figure 21: Middle East & Africa Neuro Intervention Intermediate Catheter Revenue Share (%), by Application 2025 & 2033
- Figure 22: Middle East & Africa Neuro Intervention Intermediate Catheter Revenue (billion), by Types 2025 & 2033
- Figure 23: Middle East & Africa Neuro Intervention Intermediate Catheter Revenue Share (%), by Types 2025 & 2033
- Figure 24: Middle East & Africa Neuro Intervention Intermediate Catheter Revenue (billion), by Country 2025 & 2033
- Figure 25: Middle East & Africa Neuro Intervention Intermediate Catheter Revenue Share (%), by Country 2025 & 2033
- Figure 26: Asia Pacific Neuro Intervention Intermediate Catheter Revenue (billion), by Application 2025 & 2033
- Figure 27: Asia Pacific Neuro Intervention Intermediate Catheter Revenue Share (%), by Application 2025 & 2033
- Figure 28: Asia Pacific Neuro Intervention Intermediate Catheter Revenue (billion), by Types 2025 & 2033
- Figure 29: Asia Pacific Neuro Intervention Intermediate Catheter Revenue Share (%), by Types 2025 & 2033
- Figure 30: Asia Pacific Neuro Intervention Intermediate Catheter Revenue (billion), by Country 2025 & 2033
- Figure 31: Asia Pacific Neuro Intervention Intermediate Catheter Revenue Share (%), by Country 2025 & 2033
List of Tables
- Table 1: Global Neuro Intervention Intermediate Catheter Revenue billion Forecast, by Application 2020 & 2033
- Table 2: Global Neuro Intervention Intermediate Catheter Revenue billion Forecast, by Types 2020 & 2033
- Table 3: Global Neuro Intervention Intermediate Catheter Revenue billion Forecast, by Region 2020 & 2033
- Table 4: Global Neuro Intervention Intermediate Catheter Revenue billion Forecast, by Application 2020 & 2033
- Table 5: Global Neuro Intervention Intermediate Catheter Revenue billion Forecast, by Types 2020 & 2033
- Table 6: Global Neuro Intervention Intermediate Catheter Revenue billion Forecast, by Country 2020 & 2033
- Table 7: United States Neuro Intervention Intermediate Catheter Revenue (billion) Forecast, by Application 2020 & 2033
- Table 8: Canada Neuro Intervention Intermediate Catheter Revenue (billion) Forecast, by Application 2020 & 2033
- Table 9: Mexico Neuro Intervention Intermediate Catheter Revenue (billion) Forecast, by Application 2020 & 2033
- Table 10: Global Neuro Intervention Intermediate Catheter Revenue billion Forecast, by Application 2020 & 2033
- Table 11: Global Neuro Intervention Intermediate Catheter Revenue billion Forecast, by Types 2020 & 2033
- Table 12: Global Neuro Intervention Intermediate Catheter Revenue billion Forecast, by Country 2020 & 2033
- Table 13: Brazil Neuro Intervention Intermediate Catheter Revenue (billion) Forecast, by Application 2020 & 2033
- Table 14: Argentina Neuro Intervention Intermediate Catheter Revenue (billion) Forecast, by Application 2020 & 2033
- Table 15: Rest of South America Neuro Intervention Intermediate Catheter Revenue (billion) Forecast, by Application 2020 & 2033
- Table 16: Global Neuro Intervention Intermediate Catheter Revenue billion Forecast, by Application 2020 & 2033
- Table 17: Global Neuro Intervention Intermediate Catheter Revenue billion Forecast, by Types 2020 & 2033
- Table 18: Global Neuro Intervention Intermediate Catheter Revenue billion Forecast, by Country 2020 & 2033
- Table 19: United Kingdom Neuro Intervention Intermediate Catheter Revenue (billion) Forecast, by Application 2020 & 2033
- Table 20: Germany Neuro Intervention Intermediate Catheter Revenue (billion) Forecast, by Application 2020 & 2033
- Table 21: France Neuro Intervention Intermediate Catheter Revenue (billion) Forecast, by Application 2020 & 2033
- Table 22: Italy Neuro Intervention Intermediate Catheter Revenue (billion) Forecast, by Application 2020 & 2033
- Table 23: Spain Neuro Intervention Intermediate Catheter Revenue (billion) Forecast, by Application 2020 & 2033
- Table 24: Russia Neuro Intervention Intermediate Catheter Revenue (billion) Forecast, by Application 2020 & 2033
- Table 25: Benelux Neuro Intervention Intermediate Catheter Revenue (billion) Forecast, by Application 2020 & 2033
- Table 26: Nordics Neuro Intervention Intermediate Catheter Revenue (billion) Forecast, by Application 2020 & 2033
- Table 27: Rest of Europe Neuro Intervention Intermediate Catheter Revenue (billion) Forecast, by Application 2020 & 2033
- Table 28: Global Neuro Intervention Intermediate Catheter Revenue billion Forecast, by Application 2020 & 2033
- Table 29: Global Neuro Intervention Intermediate Catheter Revenue billion Forecast, by Types 2020 & 2033
- Table 30: Global Neuro Intervention Intermediate Catheter Revenue billion Forecast, by Country 2020 & 2033
- Table 31: Turkey Neuro Intervention Intermediate Catheter Revenue (billion) Forecast, by Application 2020 & 2033
- Table 32: Israel Neuro Intervention Intermediate Catheter Revenue (billion) Forecast, by Application 2020 & 2033
- Table 33: GCC Neuro Intervention Intermediate Catheter Revenue (billion) Forecast, by Application 2020 & 2033
- Table 34: North Africa Neuro Intervention Intermediate Catheter Revenue (billion) Forecast, by Application 2020 & 2033
- Table 35: South Africa Neuro Intervention Intermediate Catheter Revenue (billion) Forecast, by Application 2020 & 2033
- Table 36: Rest of Middle East & Africa Neuro Intervention Intermediate Catheter Revenue (billion) Forecast, by Application 2020 & 2033
- Table 37: Global Neuro Intervention Intermediate Catheter Revenue billion Forecast, by Application 2020 & 2033
- Table 38: Global Neuro Intervention Intermediate Catheter Revenue billion Forecast, by Types 2020 & 2033
- Table 39: Global Neuro Intervention Intermediate Catheter Revenue billion Forecast, by Country 2020 & 2033
- Table 40: China Neuro Intervention Intermediate Catheter Revenue (billion) Forecast, by Application 2020 & 2033
- Table 41: India Neuro Intervention Intermediate Catheter Revenue (billion) Forecast, by Application 2020 & 2033
- Table 42: Japan Neuro Intervention Intermediate Catheter Revenue (billion) Forecast, by Application 2020 & 2033
- Table 43: South Korea Neuro Intervention Intermediate Catheter Revenue (billion) Forecast, by Application 2020 & 2033
- Table 44: ASEAN Neuro Intervention Intermediate Catheter Revenue (billion) Forecast, by Application 2020 & 2033
- Table 45: Oceania Neuro Intervention Intermediate Catheter Revenue (billion) Forecast, by Application 2020 & 2033
- Table 46: Rest of Asia Pacific Neuro Intervention Intermediate Catheter Revenue (billion) Forecast, by Application 2020 & 2033
Frequently Asked Questions
1. What is the projected Compound Annual Growth Rate (CAGR) of the Neuro Intervention Intermediate Catheter?
The projected CAGR is approximately 4.7%.
2. Which companies are prominent players in the Neuro Intervention Intermediate Catheter?
Key companies in the market include Medtronic, Stryker, Penumbra, MicroVention, TJWY Medical, Nanjing Precision Medical, Pejia Medical.
3. What are the main segments of the Neuro Intervention Intermediate Catheter?
The market segments include Application, Types.
4. Can you provide details about the market size?
The market size is estimated to be USD 2.82 billion as of 2022.
5. What are some drivers contributing to market growth?
N/A
6. What are the notable trends driving market growth?
N/A
7. Are there any restraints impacting market growth?
N/A
8. Can you provide examples of recent developments in the market?
N/A
9. What pricing options are available for accessing the report?
Pricing options include single-user, multi-user, and enterprise licenses priced at USD 4900.00, USD 7350.00, and USD 9800.00 respectively.
10. Is the market size provided in terms of value or volume?
The market size is provided in terms of value, measured in billion.
11. Are there any specific market keywords associated with the report?
Yes, the market keyword associated with the report is "Neuro Intervention Intermediate Catheter," which aids in identifying and referencing the specific market segment covered.
12. How do I determine which pricing option suits my needs best?
The pricing options vary based on user requirements and access needs. Individual users may opt for single-user licenses, while businesses requiring broader access may choose multi-user or enterprise licenses for cost-effective access to the report.
13. Are there any additional resources or data provided in the Neuro Intervention Intermediate Catheter report?
While the report offers comprehensive insights, it's advisable to review the specific contents or supplementary materials provided to ascertain if additional resources or data are available.
14. How can I stay updated on further developments or reports in the Neuro Intervention Intermediate Catheter?
To stay informed about further developments, trends, and reports in the Neuro Intervention Intermediate Catheter, consider subscribing to industry newsletters, following relevant companies and organizations, or regularly checking reputable industry news sources and publications.
Methodology
Step 1 - Identification of Relevant Samples Size from Population Database



Step 2 - Approaches for Defining Global Market Size (Value, Volume* & Price*)

Note*: In applicable scenarios
Step 3 - Data Sources
Primary Research
- Web Analytics
- Survey Reports
- Research Institute
- Latest Research Reports
- Opinion Leaders
Secondary Research
- Annual Reports
- White Paper
- Latest Press Release
- Industry Association
- Paid Database
- Investor Presentations

Step 4 - Data Triangulation
Involves using different sources of information in order to increase the validity of a study
These sources are likely to be stakeholders in a program - participants, other researchers, program staff, other community members, and so on.
Then we put all data in single framework & apply various statistical tools to find out the dynamic on the market.
During the analysis stage, feedback from the stakeholder groups would be compared to determine areas of agreement as well as areas of divergence


