Key Insights
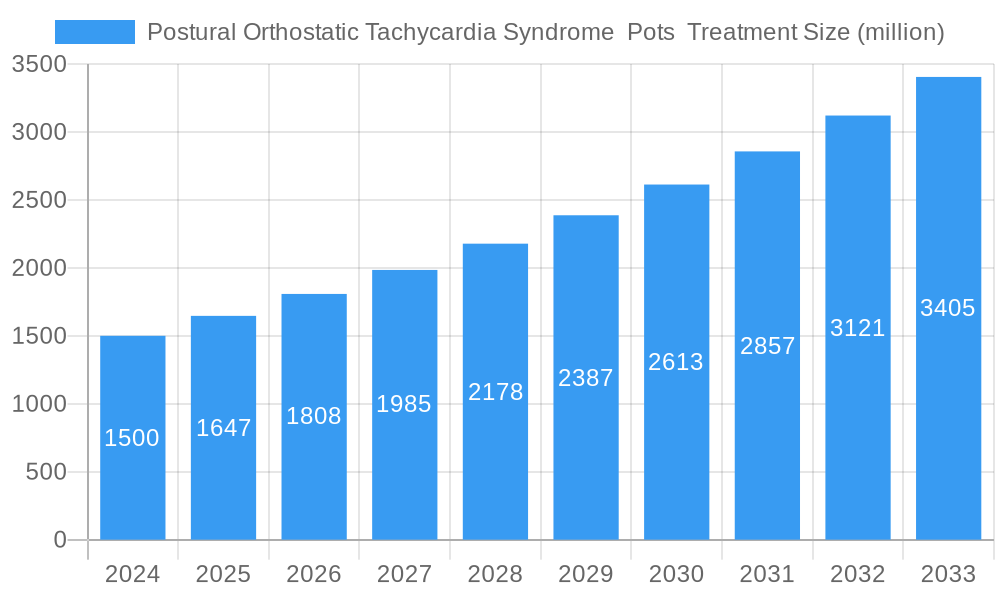
The global Postural Orthostatic Tachycardia Syndrome (POTS) treatment market is poised for substantial growth, driven by increasing awareness, improved diagnostic capabilities, and a rising incidence of the condition. Valued at an estimated $1.5 billion in 2024, the market is projected to expand at a robust Compound Annual Growth Rate (CAGR) of 9.8% through 2033. This upward trajectory is fueled by several key factors. Advances in understanding POTS, once considered rare, have led to better identification and diagnosis across various healthcare settings, including hospitals, clinics, and ambulatory surgical centers. The growing prevalence, particularly among younger demographics and post-viral syndromes, is creating a larger patient pool demanding effective therapeutic interventions. Pharmaceutical companies like Pfizer, Novartis, AstraZeneca, Merck, Sanofi, Daiichi Sankyo, and Bayer are actively involved, investing in research and development to bring innovative treatments to market. The focus is shifting towards combination therapies and personalized medicine approaches to address the complex and multifaceted nature of POTS symptoms.
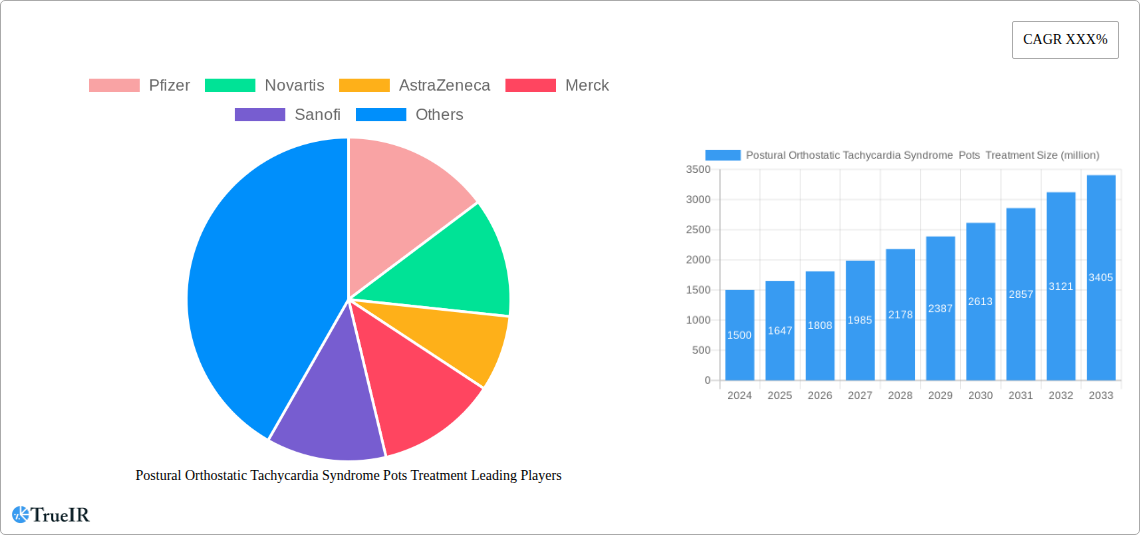
Postural Orthostatic Tachycardia Syndrome Pots Treatment Market Size (In Billion)

The market's expansion is further supported by emerging trends in treatment modalities, encompassing both pharmacological interventions and non-pharmacological approaches such as lifestyle modifications and physical therapy. Primary forms of treatment, aimed at managing core symptoms like heart rate elevation and blood pressure fluctuations, are a significant segment, while secondary forms, addressing underlying causes or associated conditions, are also gaining traction. Geographically, North America and Europe currently dominate the market due to advanced healthcare infrastructure and high patient awareness. However, the Asia Pacific region is expected to witness the fastest growth, fueled by increasing healthcare expenditure, a rising middle class, and a growing focus on rare disease research and treatment. While restraining factors such as limited disease understanding, lack of universally recognized diagnostic criteria, and the high cost of some specialized treatments exist, the overall outlook for the POTS treatment market remains exceptionally positive, promising significant advancements in patient care and market value.
Postural Orthostatic Tachycardia Syndrome Pots Treatment Company Market Share

This comprehensive report, Postural Orthostatic Tachycardia Syndrome (POTS) Treatment Market: Global Industry Analysis, Size, Share, Growth, Trends, Forecasts 2019–2033, provides an in-depth analysis of the global market for POTS treatment. Leveraging high-volume keywords such as "POTS treatment," "orthostatic tachycardia syndrome," "dysautonomia treatment," and "neurological disorder therapeutics," this report is meticulously crafted to enhance search rankings and engage a wide array of industry professionals, including researchers, pharmaceutical companies, healthcare providers, and investors. The study encompasses a vast market size, projected to reach several billion dollars, with a robust CAGR during the forecast period. We delve into the intricate dynamics, competitive strategies, and future trajectories of this critical healthcare segment, offering actionable intelligence for strategic decision-making.
Postural Orthostatic Tachycardia Syndrome Pots Treatment Market Structure & Competitive Landscape
The Postural Orthostatic Tachycardia Syndrome (POTS) Treatment market is characterized by a moderate level of concentration, with a handful of key players, including Pfizer, Novartis, AstraZeneca, Merck, Sanofi, Daiichi Sankyo, and Bayer, holding significant market share. Innovation drivers are primarily focused on developing novel pharmacological interventions and advanced diagnostic tools to improve patient outcomes and address the unmet medical needs associated with POTS. Regulatory impacts, such as evolving FDA guidelines and international drug approval processes, play a crucial role in shaping market entry and product development strategies. Product substitutes, while limited, include off-label use of existing medications and non-pharmacological management approaches.
- Market Concentration: Moderate, with key pharmaceutical giants leading the innovation pipeline.
- Innovation Drivers: Novel drug development for symptomatic relief, improved diagnostic accuracy, and understanding of POTS pathophysiology.
- Regulatory Impacts: Stringent approval processes for new POTS treatments, influencing R&D timelines and investment.
- Product Substitutes: Lifestyle modifications, physical therapy, and off-label drug utilization.
- End-User Segmentation: Dominantly Hospitals and Clinics, with growing contributions from Research Institutes.
- M&A Trends: Emerging strategic partnerships and acquisitions aimed at consolidating market presence and expanding therapeutic portfolios. The volume of M&A activities is estimated to be in the billions.
Postural Orthostatic Tachycardia Syndrome Pots Treatment Market Trends & Opportunities
The global Postural Orthostatic Tachycardia Syndrome (POTS) Treatment market is poised for substantial growth, driven by an increasing prevalence of POTS, a growing understanding of its complex pathophysiology, and advancements in diagnostic methodologies. Market size is expected to expand significantly, reaching billions of dollars by 2033, with an impressive Compound Annual Growth Rate (CAGR) during the forecast period. Technological shifts are revolutionizing POTS diagnosis and management, with innovations in wearable technology for continuous monitoring, genetic sequencing for identifying predispositions, and advanced imaging techniques providing deeper insights into autonomic nervous system dysfunction.
Consumer preferences are increasingly shifting towards personalized treatment plans and integrated care approaches that address the multifaceted nature of POTS, encompassing both physical and psychological well-being. The competitive landscape is intensifying, with pharmaceutical companies actively investing in research and development to bring novel therapeutic agents to market. This includes exploring medications targeting specific neurotransmitter pathways, anti-inflammatory agents, and therapies aimed at improving cardiovascular regulation. Market penetration rates for POTS-specific treatments are expected to rise as diagnostic capabilities improve and physician awareness increases.
The growing patient advocacy for POTS research and treatment has also played a pivotal role in driving market expansion. Numerous patient support groups and foundations are actively involved in raising awareness, funding research, and advocating for better healthcare access. This heightened patient engagement is creating a demand for more effective and accessible treatment options, presenting significant opportunities for companies that can deliver innovative solutions. Furthermore, the increasing recognition of POTS as a distinct neurological and cardiovascular disorder by leading health organizations is leading to greater investment in research and development from both public and private sectors.
Opportunities abound in the development of combination therapies that address the various symptoms of POTS, such as dizziness, fatigue, palpitations, and cognitive dysfunction. Personalized medicine approaches, tailored to an individual's genetic makeup and specific POTS subtype, are also gaining traction. The integration of digital health solutions, including telemedicine and remote patient monitoring, offers a promising avenue for improving patient care and extending the reach of POTS treatment, especially in underserved geographical areas. The market is also witnessing a growing interest in non-pharmacological interventions, such as specialized physical therapy programs and dietary modifications, presenting opportunities for integrated healthcare service providers.
Dominant Markets & Segments in Postural Orthostatic Tachycardia Syndrome Pots Treatment
The global Postural Orthostatic Tachycardia Syndrome (POTS) Treatment market demonstrates significant regional and segment-specific dominance, driven by a confluence of factors including healthcare infrastructure, research funding, regulatory frameworks, and patient demographics. North America, particularly the United States, currently holds a dominant position due to its advanced healthcare systems, high healthcare expenditure, and robust investment in neurological and cardiovascular research. The presence of leading pharmaceutical companies and research institutions fosters a conducive environment for the development and adoption of innovative POTS treatments.
Among the application segments, Hospitals emerge as the primary point of care for POTS diagnosis and treatment, owing to their comprehensive diagnostic capabilities, specialized medical expertise, and access to a wide range of therapeutic interventions. Clinics follow closely, serving as crucial hubs for outpatient management and ongoing patient care. The increasing prevalence of POTS and the growing emphasis on early diagnosis and intervention are further bolstering the demand within these segments.
In terms of POTS Type, Primary Forms of POTS, which are idiopathic or autoimmune in origin, represent a larger segment of the market. This is attributed to a higher incidence rate and the ongoing efforts to unravel the underlying mechanisms of primary POTS. However, the market for Secondary Forms of POTS, which arise as a complication of other medical conditions such as viral infections, connective tissue disorders, or genetic predispositions, is also experiencing notable growth. This expansion is linked to improved diagnostic capabilities that can identify POTS as a comorbidity.
- Leading Region: North America (driven by the US) due to advanced healthcare infrastructure and research investment.
- Dominant Application Segments:
- Hospitals: Comprehensive diagnostic and treatment facilities, specialized care.
- Clinics: Outpatient management, follow-up care, and increasing diagnostic outreach.
- Key Growth Drivers for Applications:
- Infrastructure: Advanced diagnostic equipment and specialized medical personnel.
- Policies: Favorable reimbursement policies and government initiatives for rare disease research.
- Awareness: Rising patient and physician awareness driving early diagnosis and treatment.
- Dominant Type Segment:
- Primary Forms: Higher prevalence and ongoing research into idiopathic and autoimmune POTS.
- Growth Drivers for Type Segments:
- Research: Deeper understanding of the pathogenesis of primary POTS.
- Diagnosis: Improved ability to identify POTS as a secondary complication of various conditions.
- Targeted Therapies: Development of treatments specific to the underlying causes of each POTS type.
- Ambulatory Surgical Centers: While currently a smaller segment, these centers are projected to see growth as minimally invasive diagnostic procedures and therapeutic interventions become more common.
- Research Institutes: Crucial for driving innovation, understanding disease mechanisms, and clinical trial execution, contributing significantly to the overall market knowledge base.
Postural Orthostatic Tachycardia Syndrome Pots Treatment Product Analysis
The Postural Orthostatic Tachycardia Syndrome (POTS) Treatment market is characterized by a growing pipeline of therapeutic agents aimed at managing the diverse symptoms of this complex condition. Current product innovations focus on symptomatic relief through medications that modulate heart rate, blood pressure, and autonomic nervous system function. These include beta-blockers, alpha-agonists, and fludrocortisone, with ongoing research exploring novel targets such as serotonin receptors and immune modulators. The competitive advantage lies in products that offer a favorable risk-benefit profile, address multiple POTS symptoms, and improve patients' quality of life. Technological advancements are also driving the development of advanced diagnostic tools and wearable devices for continuous patient monitoring, further enhancing the efficacy and personalization of treatment.
Key Drivers, Barriers & Challenges in Postural Orthostatic Tachycardia Syndrome Pots Treatment
The Postural Orthostatic Tachycardia Syndrome (POTS) Treatment market is propelled by several key drivers, including the increasing global prevalence of POTS, heightened awareness among healthcare professionals and the public, and significant advancements in understanding the underlying pathophysiology. Technological innovations in diagnostics, such as tilt table testing and autonomic function tests, are improving diagnostic accuracy. Furthermore, substantial investments in research and development by pharmaceutical companies and research institutions are leading to the discovery of novel therapeutic targets and drug candidates. The growing patient advocacy movement is also a powerful catalyst, driving demand for better treatments and increased research funding.
Conversely, the market faces significant challenges and barriers. The complex and heterogeneous nature of POTS often leads to diagnostic delays and misdiagnosis, hindering timely treatment initiation. The lack of a single, universally effective cure necessitates a multi-faceted treatment approach, which can be challenging to implement. Regulatory hurdles for new drug approvals, coupled with the high cost of research and development, can impede market entry. Furthermore, limited reimbursement for POTS-specific treatments in certain regions and the relatively small patient population compared to other chronic diseases can impact market profitability and investment. Supply chain disruptions for specialized medications and diagnostic supplies also pose a persistent threat.
Growth Drivers in the Postural Orthostatic Tachycardia Syndrome Pots Treatment Market
The Postural Orthostatic Tachycardia Syndrome (POTS) Treatment market is experiencing robust growth driven by critical factors. Technologically, advancements in diagnostic tools, including refined autonomic function testing and wearable biosensors for continuous monitoring, are enabling earlier and more accurate diagnoses. Economically, increased healthcare spending and favorable reimbursement policies in developed nations are supporting market expansion. Regulatory bodies are also showing a growing commitment to facilitating the development of treatments for rare and complex neurological disorders, potentially expediting the approval process for new POTS therapies. Public health initiatives and increased funding for dysautonomia research are further fueling the development of novel treatment strategies and pharmaceutical interventions. The growing understanding of POTS as a distinct condition, often post-viral, is also contributing to its recognition and subsequent market growth.
Challenges Impacting Postural Orthostatic Tachycardia Syndrome Pots Treatment Growth
Several challenges are impacting the growth trajectory of the Postural Orthostatic Tachycardia Syndrome (POTS) Treatment market. Regulatory complexities, including the stringent and lengthy approval processes for novel therapeutics, can significantly slow down market penetration and increase development costs. Supply chain issues, such as the availability and accessibility of specialized medications and diagnostic equipment, particularly in emerging markets, can hinder consistent treatment delivery. Competitive pressures from established pharmaceutical players and the potential for generic competition, once patents expire, also influence market dynamics. Furthermore, the relatively niche market size, despite growing awareness, can make it less attractive for some large-scale pharmaceutical investments compared to more prevalent diseases. The high cost associated with developing and manufacturing specialized treatments can also be a barrier to affordability for patients.
Key Players Shaping the Postural Orthostatic Tachycardia Syndrome Pots Treatment Market
The Postural Orthostatic Tachycardia Syndrome (POTS) Treatment market is shaped by the strategic initiatives and research endeavors of leading global pharmaceutical and biotechnology companies. Key players actively contributing to the development and provision of POTS treatments include:
- Pfizer
- Novartis
- AstraZeneca
- Merck
- Sanofi
- Daiichi Sankyo
- Bayer
Significant Postural Orthostatic Tachycardia Syndrome Pots Treatment Industry Milestones
The Postural Orthostatic Tachycardia Syndrome (POTS) Treatment landscape has been shaped by several significant industry milestones:
- 2019: Increased research funding allocated to dysautonomia disorders, including POTS, by governmental health organizations.
- 2020: Publication of updated diagnostic criteria for POTS, leading to improved identification and diagnosis rates.
- 2021: Launch of clinical trials for novel pharmacological agents targeting specific POTS subtypes.
- 2022: Advancements in wearable technology for continuous cardiovascular and autonomic monitoring of POTS patients.
- 2023: Formation of strategic partnerships between pharmaceutical companies and academic institutions to accelerate POTS research.
- 2024: Emergence of specialized POTS clinics and treatment centers offering integrated care approaches.
- 2025: Anticipated regulatory submissions for promising new POTS therapies.
- 2026-2033: Expected market expansion driven by increased product approvals and wider adoption of advanced diagnostic tools.
Future Outlook for Postural Orthostatic Tachycardia Syndrome Pots Treatment Market
The future outlook for the Postural Orthostatic Tachycardia Syndrome (POTS) Treatment market is exceptionally promising, characterized by significant growth catalysts. Continued investment in research and development is expected to yield novel, targeted therapies addressing the underlying mechanisms of POTS, moving beyond symptomatic management. The integration of digital health technologies, including AI-powered diagnostic tools and personalized telemedicine platforms, will enhance patient accessibility and treatment efficacy. Strategic collaborations between pharmaceutical companies, research institutions, and patient advocacy groups will further accelerate innovation and market penetration. The increasing recognition of POTS as a significant public health concern will likely lead to more favorable regulatory pathways and increased healthcare provider education, ultimately expanding treatment options and improving the quality of life for millions affected globally.
Postural Orthostatic Tachycardia Syndrome Pots Treatment Segmentation
-
1. Application
- 1.1. Hospitals
- 1.2. Clinics
- 1.3. Ambulatory Surgical Centers
- 1.4. Research Institutes
-
2. Type
- 2.1. Primary Forms
- 2.2. Secondary Forms
Postural Orthostatic Tachycardia Syndrome Pots Treatment Segmentation By Geography
-
1. North America
- 1.1. United States
- 1.2. Canada
- 1.3. Mexico
-
2. South America
- 2.1. Brazil
- 2.2. Argentina
- 2.3. Rest of South America
-
3. Europe
- 3.1. United Kingdom
- 3.2. Germany
- 3.3. France
- 3.4. Italy
- 3.5. Spain
- 3.6. Russia
- 3.7. Benelux
- 3.8. Nordics
- 3.9. Rest of Europe
-
4. Middle East & Africa
- 4.1. Turkey
- 4.2. Israel
- 4.3. GCC
- 4.4. North Africa
- 4.5. South Africa
- 4.6. Rest of Middle East & Africa
-
5. Asia Pacific
- 5.1. China
- 5.2. India
- 5.3. Japan
- 5.4. South Korea
- 5.5. ASEAN
- 5.6. Oceania
- 5.7. Rest of Asia Pacific
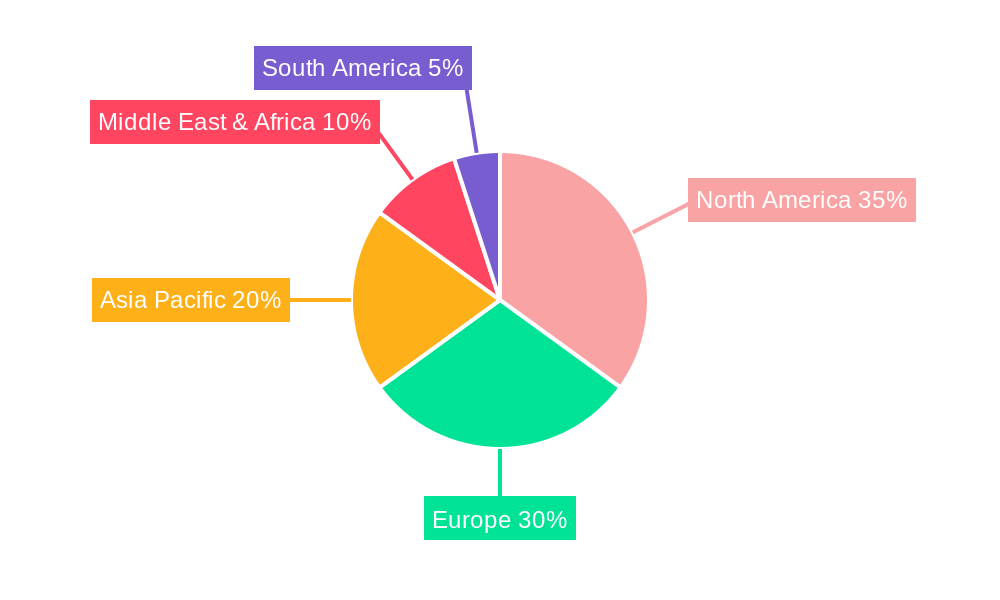
Postural Orthostatic Tachycardia Syndrome Pots Treatment Regional Market Share

Geographic Coverage of Postural Orthostatic Tachycardia Syndrome Pots Treatment
Postural Orthostatic Tachycardia Syndrome Pots Treatment REPORT HIGHLIGHTS
| Aspects | Details |
|---|---|
| Study Period | 2020-2034 |
| Base Year | 2025 |
| Estimated Year | 2026 |
| Forecast Period | 2026-2034 |
| Historical Period | 2020-2025 |
| Growth Rate | CAGR of 9.8% from 2020-2034 |
| Segmentation |
|
Table of Contents
- 1. Introduction
- 1.1. Research Scope
- 1.2. Market Segmentation
- 1.3. Research Methodology
- 1.4. Definitions and Assumptions
- 2. Executive Summary
- 2.1. Introduction
- 3. Market Dynamics
- 3.1. Introduction
- 3.2. Market Drivers
- 3.3. Market Restrains
- 3.4. Market Trends
- 4. Market Factor Analysis
- 4.1. Porters Five Forces
- 4.2. Supply/Value Chain
- 4.3. PESTEL analysis
- 4.4. Market Entropy
- 4.5. Patent/Trademark Analysis
- 5. Global Postural Orthostatic Tachycardia Syndrome Pots Treatment Analysis, Insights and Forecast, 2020-2032
- 5.1. Market Analysis, Insights and Forecast - by Application
- 5.1.1. Hospitals
- 5.1.2. Clinics
- 5.1.3. Ambulatory Surgical Centers
- 5.1.4. Research Institutes
- 5.2. Market Analysis, Insights and Forecast - by Type
- 5.2.1. Primary Forms
- 5.2.2. Secondary Forms
- 5.3. Market Analysis, Insights and Forecast - by Region
- 5.3.1. North America
- 5.3.2. South America
- 5.3.3. Europe
- 5.3.4. Middle East & Africa
- 5.3.5. Asia Pacific
- 5.1. Market Analysis, Insights and Forecast - by Application
- 6. North America Postural Orthostatic Tachycardia Syndrome Pots Treatment Analysis, Insights and Forecast, 2020-2032
- 6.1. Market Analysis, Insights and Forecast - by Application
- 6.1.1. Hospitals
- 6.1.2. Clinics
- 6.1.3. Ambulatory Surgical Centers
- 6.1.4. Research Institutes
- 6.2. Market Analysis, Insights and Forecast - by Type
- 6.2.1. Primary Forms
- 6.2.2. Secondary Forms
- 6.1. Market Analysis, Insights and Forecast - by Application
- 7. South America Postural Orthostatic Tachycardia Syndrome Pots Treatment Analysis, Insights and Forecast, 2020-2032
- 7.1. Market Analysis, Insights and Forecast - by Application
- 7.1.1. Hospitals
- 7.1.2. Clinics
- 7.1.3. Ambulatory Surgical Centers
- 7.1.4. Research Institutes
- 7.2. Market Analysis, Insights and Forecast - by Type
- 7.2.1. Primary Forms
- 7.2.2. Secondary Forms
- 7.1. Market Analysis, Insights and Forecast - by Application
- 8. Europe Postural Orthostatic Tachycardia Syndrome Pots Treatment Analysis, Insights and Forecast, 2020-2032
- 8.1. Market Analysis, Insights and Forecast - by Application
- 8.1.1. Hospitals
- 8.1.2. Clinics
- 8.1.3. Ambulatory Surgical Centers
- 8.1.4. Research Institutes
- 8.2. Market Analysis, Insights and Forecast - by Type
- 8.2.1. Primary Forms
- 8.2.2. Secondary Forms
- 8.1. Market Analysis, Insights and Forecast - by Application
- 9. Middle East & Africa Postural Orthostatic Tachycardia Syndrome Pots Treatment Analysis, Insights and Forecast, 2020-2032
- 9.1. Market Analysis, Insights and Forecast - by Application
- 9.1.1. Hospitals
- 9.1.2. Clinics
- 9.1.3. Ambulatory Surgical Centers
- 9.1.4. Research Institutes
- 9.2. Market Analysis, Insights and Forecast - by Type
- 9.2.1. Primary Forms
- 9.2.2. Secondary Forms
- 9.1. Market Analysis, Insights and Forecast - by Application
- 10. Asia Pacific Postural Orthostatic Tachycardia Syndrome Pots Treatment Analysis, Insights and Forecast, 2020-2032
- 10.1. Market Analysis, Insights and Forecast - by Application
- 10.1.1. Hospitals
- 10.1.2. Clinics
- 10.1.3. Ambulatory Surgical Centers
- 10.1.4. Research Institutes
- 10.2. Market Analysis, Insights and Forecast - by Type
- 10.2.1. Primary Forms
- 10.2.2. Secondary Forms
- 10.1. Market Analysis, Insights and Forecast - by Application
- 11. Competitive Analysis
- 11.1. Global Market Share Analysis 2025
- 11.2. Company Profiles
- 11.2.1 Pfizer
- 11.2.1.1. Overview
- 11.2.1.2. Products
- 11.2.1.3. SWOT Analysis
- 11.2.1.4. Recent Developments
- 11.2.1.5. Financials (Based on Availability)
- 11.2.2 Novartis
- 11.2.2.1. Overview
- 11.2.2.2. Products
- 11.2.2.3. SWOT Analysis
- 11.2.2.4. Recent Developments
- 11.2.2.5. Financials (Based on Availability)
- 11.2.3 AstraZeneca
- 11.2.3.1. Overview
- 11.2.3.2. Products
- 11.2.3.3. SWOT Analysis
- 11.2.3.4. Recent Developments
- 11.2.3.5. Financials (Based on Availability)
- 11.2.4 Merck
- 11.2.4.1. Overview
- 11.2.4.2. Products
- 11.2.4.3. SWOT Analysis
- 11.2.4.4. Recent Developments
- 11.2.4.5. Financials (Based on Availability)
- 11.2.5 Sanofi
- 11.2.5.1. Overview
- 11.2.5.2. Products
- 11.2.5.3. SWOT Analysis
- 11.2.5.4. Recent Developments
- 11.2.5.5. Financials (Based on Availability)
- 11.2.6 Daiichi Sankyo
- 11.2.6.1. Overview
- 11.2.6.2. Products
- 11.2.6.3. SWOT Analysis
- 11.2.6.4. Recent Developments
- 11.2.6.5. Financials (Based on Availability)
- 11.2.7 Bayer
- 11.2.7.1. Overview
- 11.2.7.2. Products
- 11.2.7.3. SWOT Analysis
- 11.2.7.4. Recent Developments
- 11.2.7.5. Financials (Based on Availability)
- 11.2.1 Pfizer
List of Figures
- Figure 1: Global Postural Orthostatic Tachycardia Syndrome Pots Treatment Revenue Breakdown (undefined, %) by Region 2025 & 2033
- Figure 2: North America Postural Orthostatic Tachycardia Syndrome Pots Treatment Revenue (undefined), by Application 2025 & 2033
- Figure 3: North America Postural Orthostatic Tachycardia Syndrome Pots Treatment Revenue Share (%), by Application 2025 & 2033
- Figure 4: North America Postural Orthostatic Tachycardia Syndrome Pots Treatment Revenue (undefined), by Type 2025 & 2033
- Figure 5: North America Postural Orthostatic Tachycardia Syndrome Pots Treatment Revenue Share (%), by Type 2025 & 2033
- Figure 6: North America Postural Orthostatic Tachycardia Syndrome Pots Treatment Revenue (undefined), by Country 2025 & 2033
- Figure 7: North America Postural Orthostatic Tachycardia Syndrome Pots Treatment Revenue Share (%), by Country 2025 & 2033
- Figure 8: South America Postural Orthostatic Tachycardia Syndrome Pots Treatment Revenue (undefined), by Application 2025 & 2033
- Figure 9: South America Postural Orthostatic Tachycardia Syndrome Pots Treatment Revenue Share (%), by Application 2025 & 2033
- Figure 10: South America Postural Orthostatic Tachycardia Syndrome Pots Treatment Revenue (undefined), by Type 2025 & 2033
- Figure 11: South America Postural Orthostatic Tachycardia Syndrome Pots Treatment Revenue Share (%), by Type 2025 & 2033
- Figure 12: South America Postural Orthostatic Tachycardia Syndrome Pots Treatment Revenue (undefined), by Country 2025 & 2033
- Figure 13: South America Postural Orthostatic Tachycardia Syndrome Pots Treatment Revenue Share (%), by Country 2025 & 2033
- Figure 14: Europe Postural Orthostatic Tachycardia Syndrome Pots Treatment Revenue (undefined), by Application 2025 & 2033
- Figure 15: Europe Postural Orthostatic Tachycardia Syndrome Pots Treatment Revenue Share (%), by Application 2025 & 2033
- Figure 16: Europe Postural Orthostatic Tachycardia Syndrome Pots Treatment Revenue (undefined), by Type 2025 & 2033
- Figure 17: Europe Postural Orthostatic Tachycardia Syndrome Pots Treatment Revenue Share (%), by Type 2025 & 2033
- Figure 18: Europe Postural Orthostatic Tachycardia Syndrome Pots Treatment Revenue (undefined), by Country 2025 & 2033
- Figure 19: Europe Postural Orthostatic Tachycardia Syndrome Pots Treatment Revenue Share (%), by Country 2025 & 2033
- Figure 20: Middle East & Africa Postural Orthostatic Tachycardia Syndrome Pots Treatment Revenue (undefined), by Application 2025 & 2033
- Figure 21: Middle East & Africa Postural Orthostatic Tachycardia Syndrome Pots Treatment Revenue Share (%), by Application 2025 & 2033
- Figure 22: Middle East & Africa Postural Orthostatic Tachycardia Syndrome Pots Treatment Revenue (undefined), by Type 2025 & 2033
- Figure 23: Middle East & Africa Postural Orthostatic Tachycardia Syndrome Pots Treatment Revenue Share (%), by Type 2025 & 2033
- Figure 24: Middle East & Africa Postural Orthostatic Tachycardia Syndrome Pots Treatment Revenue (undefined), by Country 2025 & 2033
- Figure 25: Middle East & Africa Postural Orthostatic Tachycardia Syndrome Pots Treatment Revenue Share (%), by Country 2025 & 2033
- Figure 26: Asia Pacific Postural Orthostatic Tachycardia Syndrome Pots Treatment Revenue (undefined), by Application 2025 & 2033
- Figure 27: Asia Pacific Postural Orthostatic Tachycardia Syndrome Pots Treatment Revenue Share (%), by Application 2025 & 2033
- Figure 28: Asia Pacific Postural Orthostatic Tachycardia Syndrome Pots Treatment Revenue (undefined), by Type 2025 & 2033
- Figure 29: Asia Pacific Postural Orthostatic Tachycardia Syndrome Pots Treatment Revenue Share (%), by Type 2025 & 2033
- Figure 30: Asia Pacific Postural Orthostatic Tachycardia Syndrome Pots Treatment Revenue (undefined), by Country 2025 & 2033
- Figure 31: Asia Pacific Postural Orthostatic Tachycardia Syndrome Pots Treatment Revenue Share (%), by Country 2025 & 2033
List of Tables
- Table 1: Global Postural Orthostatic Tachycardia Syndrome Pots Treatment Revenue undefined Forecast, by Application 2020 & 2033
- Table 2: Global Postural Orthostatic Tachycardia Syndrome Pots Treatment Revenue undefined Forecast, by Type 2020 & 2033
- Table 3: Global Postural Orthostatic Tachycardia Syndrome Pots Treatment Revenue undefined Forecast, by Region 2020 & 2033
- Table 4: Global Postural Orthostatic Tachycardia Syndrome Pots Treatment Revenue undefined Forecast, by Application 2020 & 2033
- Table 5: Global Postural Orthostatic Tachycardia Syndrome Pots Treatment Revenue undefined Forecast, by Type 2020 & 2033
- Table 6: Global Postural Orthostatic Tachycardia Syndrome Pots Treatment Revenue undefined Forecast, by Country 2020 & 2033
- Table 7: United States Postural Orthostatic Tachycardia Syndrome Pots Treatment Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 8: Canada Postural Orthostatic Tachycardia Syndrome Pots Treatment Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 9: Mexico Postural Orthostatic Tachycardia Syndrome Pots Treatment Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 10: Global Postural Orthostatic Tachycardia Syndrome Pots Treatment Revenue undefined Forecast, by Application 2020 & 2033
- Table 11: Global Postural Orthostatic Tachycardia Syndrome Pots Treatment Revenue undefined Forecast, by Type 2020 & 2033
- Table 12: Global Postural Orthostatic Tachycardia Syndrome Pots Treatment Revenue undefined Forecast, by Country 2020 & 2033
- Table 13: Brazil Postural Orthostatic Tachycardia Syndrome Pots Treatment Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 14: Argentina Postural Orthostatic Tachycardia Syndrome Pots Treatment Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 15: Rest of South America Postural Orthostatic Tachycardia Syndrome Pots Treatment Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 16: Global Postural Orthostatic Tachycardia Syndrome Pots Treatment Revenue undefined Forecast, by Application 2020 & 2033
- Table 17: Global Postural Orthostatic Tachycardia Syndrome Pots Treatment Revenue undefined Forecast, by Type 2020 & 2033
- Table 18: Global Postural Orthostatic Tachycardia Syndrome Pots Treatment Revenue undefined Forecast, by Country 2020 & 2033
- Table 19: United Kingdom Postural Orthostatic Tachycardia Syndrome Pots Treatment Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 20: Germany Postural Orthostatic Tachycardia Syndrome Pots Treatment Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 21: France Postural Orthostatic Tachycardia Syndrome Pots Treatment Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 22: Italy Postural Orthostatic Tachycardia Syndrome Pots Treatment Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 23: Spain Postural Orthostatic Tachycardia Syndrome Pots Treatment Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 24: Russia Postural Orthostatic Tachycardia Syndrome Pots Treatment Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 25: Benelux Postural Orthostatic Tachycardia Syndrome Pots Treatment Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 26: Nordics Postural Orthostatic Tachycardia Syndrome Pots Treatment Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 27: Rest of Europe Postural Orthostatic Tachycardia Syndrome Pots Treatment Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 28: Global Postural Orthostatic Tachycardia Syndrome Pots Treatment Revenue undefined Forecast, by Application 2020 & 2033
- Table 29: Global Postural Orthostatic Tachycardia Syndrome Pots Treatment Revenue undefined Forecast, by Type 2020 & 2033
- Table 30: Global Postural Orthostatic Tachycardia Syndrome Pots Treatment Revenue undefined Forecast, by Country 2020 & 2033
- Table 31: Turkey Postural Orthostatic Tachycardia Syndrome Pots Treatment Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 32: Israel Postural Orthostatic Tachycardia Syndrome Pots Treatment Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 33: GCC Postural Orthostatic Tachycardia Syndrome Pots Treatment Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 34: North Africa Postural Orthostatic Tachycardia Syndrome Pots Treatment Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 35: South Africa Postural Orthostatic Tachycardia Syndrome Pots Treatment Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 36: Rest of Middle East & Africa Postural Orthostatic Tachycardia Syndrome Pots Treatment Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 37: Global Postural Orthostatic Tachycardia Syndrome Pots Treatment Revenue undefined Forecast, by Application 2020 & 2033
- Table 38: Global Postural Orthostatic Tachycardia Syndrome Pots Treatment Revenue undefined Forecast, by Type 2020 & 2033
- Table 39: Global Postural Orthostatic Tachycardia Syndrome Pots Treatment Revenue undefined Forecast, by Country 2020 & 2033
- Table 40: China Postural Orthostatic Tachycardia Syndrome Pots Treatment Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 41: India Postural Orthostatic Tachycardia Syndrome Pots Treatment Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 42: Japan Postural Orthostatic Tachycardia Syndrome Pots Treatment Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 43: South Korea Postural Orthostatic Tachycardia Syndrome Pots Treatment Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 44: ASEAN Postural Orthostatic Tachycardia Syndrome Pots Treatment Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 45: Oceania Postural Orthostatic Tachycardia Syndrome Pots Treatment Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 46: Rest of Asia Pacific Postural Orthostatic Tachycardia Syndrome Pots Treatment Revenue (undefined) Forecast, by Application 2020 & 2033
Frequently Asked Questions
1. What is the projected Compound Annual Growth Rate (CAGR) of the Postural Orthostatic Tachycardia Syndrome Pots Treatment?
The projected CAGR is approximately 9.8%.
2. Which companies are prominent players in the Postural Orthostatic Tachycardia Syndrome Pots Treatment?
Key companies in the market include Pfizer, Novartis, AstraZeneca, Merck, Sanofi, Daiichi Sankyo, Bayer.
3. What are the main segments of the Postural Orthostatic Tachycardia Syndrome Pots Treatment?
The market segments include Application, Type.
4. Can you provide details about the market size?
The market size is estimated to be USD XXX N/A as of 2022.
5. What are some drivers contributing to market growth?
N/A
6. What are the notable trends driving market growth?
N/A
7. Are there any restraints impacting market growth?
N/A
8. Can you provide examples of recent developments in the market?
N/A
9. What pricing options are available for accessing the report?
Pricing options include single-user, multi-user, and enterprise licenses priced at USD 2900.00, USD 4350.00, and USD 5800.00 respectively.
10. Is the market size provided in terms of value or volume?
The market size is provided in terms of value, measured in N/A.
11. Are there any specific market keywords associated with the report?
Yes, the market keyword associated with the report is "Postural Orthostatic Tachycardia Syndrome Pots Treatment," which aids in identifying and referencing the specific market segment covered.
12. How do I determine which pricing option suits my needs best?
The pricing options vary based on user requirements and access needs. Individual users may opt for single-user licenses, while businesses requiring broader access may choose multi-user or enterprise licenses for cost-effective access to the report.
13. Are there any additional resources or data provided in the Postural Orthostatic Tachycardia Syndrome Pots Treatment report?
While the report offers comprehensive insights, it's advisable to review the specific contents or supplementary materials provided to ascertain if additional resources or data are available.
14. How can I stay updated on further developments or reports in the Postural Orthostatic Tachycardia Syndrome Pots Treatment?
To stay informed about further developments, trends, and reports in the Postural Orthostatic Tachycardia Syndrome Pots Treatment, consider subscribing to industry newsletters, following relevant companies and organizations, or regularly checking reputable industry news sources and publications.
Methodology
Step 1 - Identification of Relevant Samples Size from Population Database



Step 2 - Approaches for Defining Global Market Size (Value, Volume* & Price*)

Note*: In applicable scenarios
Step 3 - Data Sources
Primary Research
- Web Analytics
- Survey Reports
- Research Institute
- Latest Research Reports
- Opinion Leaders
Secondary Research
- Annual Reports
- White Paper
- Latest Press Release
- Industry Association
- Paid Database
- Investor Presentations

Step 4 - Data Triangulation
Involves using different sources of information in order to increase the validity of a study
These sources are likely to be stakeholders in a program - participants, other researchers, program staff, other community members, and so on.
Then we put all data in single framework & apply various statistical tools to find out the dynamic on the market.
During the analysis stage, feedback from the stakeholder groups would be compared to determine areas of agreement as well as areas of divergence


