Key Insights
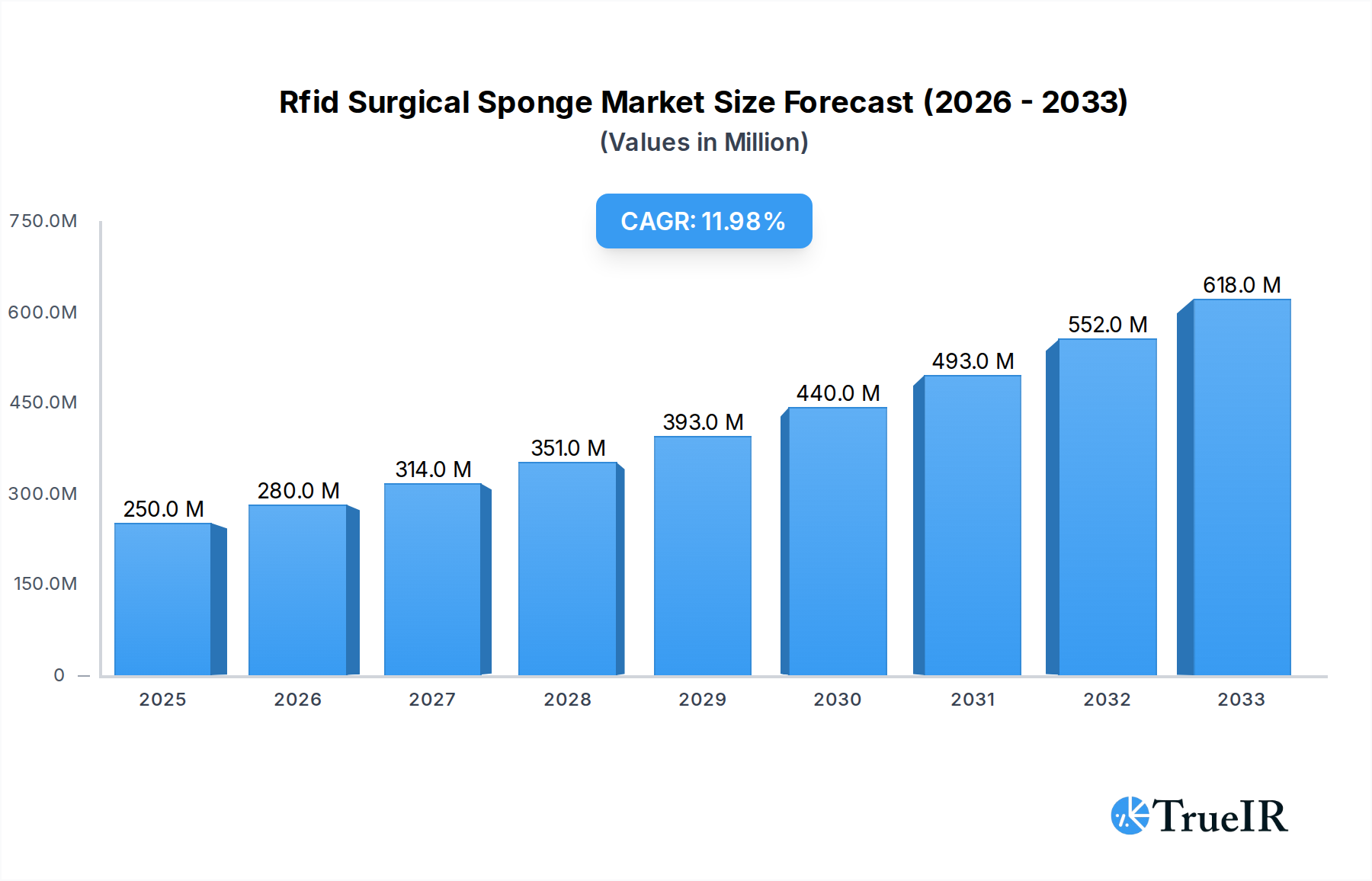
The global RFID Surgical Sponge market is experiencing robust expansion, driven primarily by an increasing emphasis on patient safety and operational efficiency within healthcare facilities. Valued at an estimated $250 million in 2025, the market is projected to grow significantly with a compound annual growth rate (CAGR) of 12% through the forecast period. A key driver for this remarkable growth is the critical need to prevent retained surgical items (RSIs), a severe medical error that can lead to adverse patient outcomes, extended hospital stays, and substantial financial and reputational damage for healthcare providers. RFID technology offers an unparalleled solution by enabling real-time tracking and accurate counting of surgical sponges, towels, and gauze, thereby minimizing human error and enhancing accountability in operating rooms. Leading companies such as Medtronic, Stryker Corporation, and STERIS are at the forefront of innovation, developing advanced RFID systems that integrate seamlessly into existing surgical workflows, further accelerating market adoption across hospitals and ambulatory surgery centers.
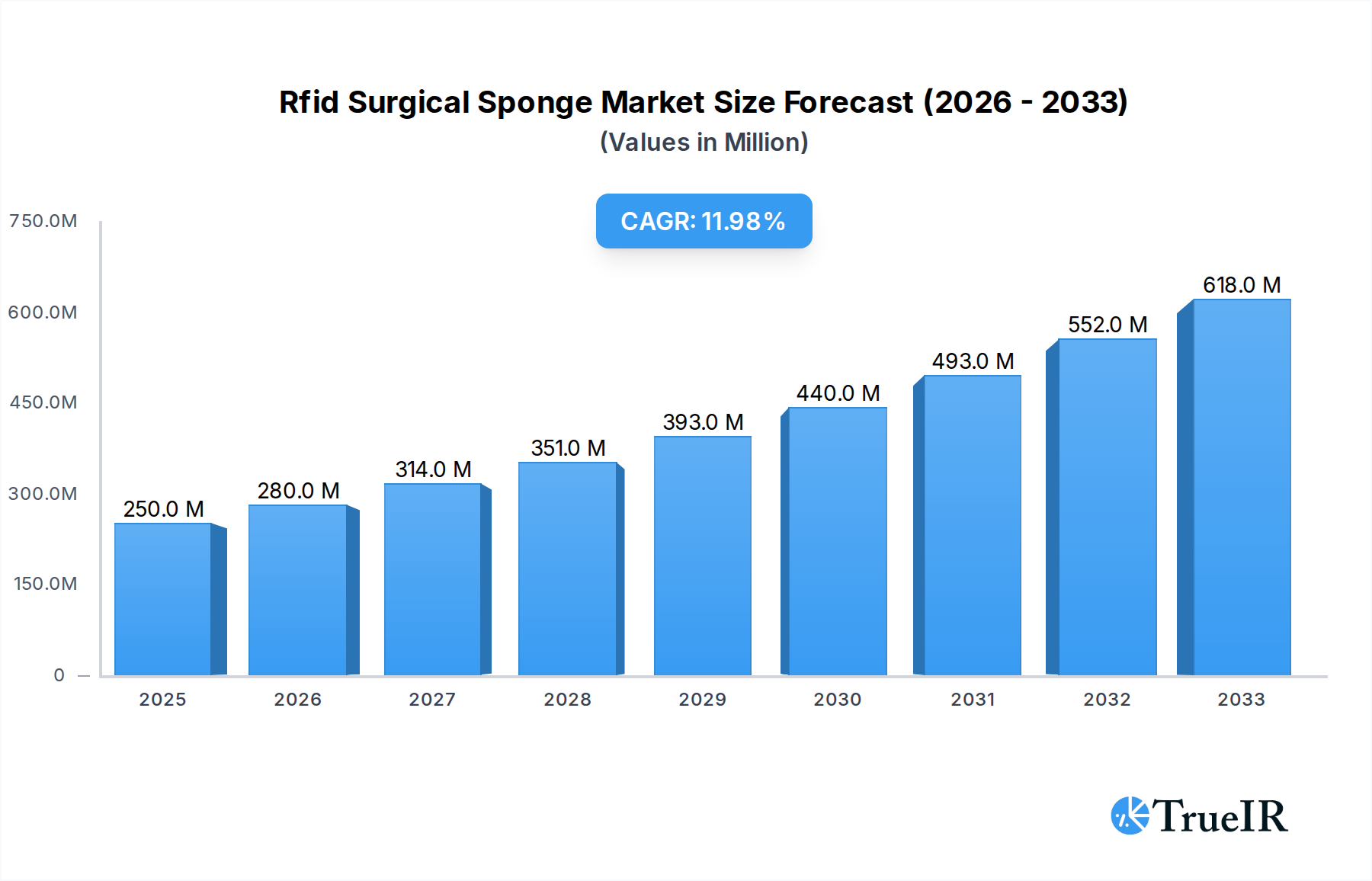
Rfid Surgical Sponge Market Size (In Million)

Furthermore, the market's trajectory is bolstered by favorable regulatory environments that advocate for enhanced patient safety protocols and the digitization of healthcare operations. The increasing volume of complex surgical procedures globally, coupled with a growing awareness of the benefits of advanced inventory management, also contributes significantly to market demand. Key trends include the miniaturization of RFID tags, allowing for integration into a wider range of surgical consumables, and the development of intelligent systems capable of interfacing with hospital information systems for comprehensive data management. While initial investment costs and the need for standardized protocols across different healthcare settings present certain challenges, the long-term benefits of improved patient safety, reduced litigation risks, and enhanced operational efficiency are expected to outweigh these hurdles. The market is poised for continued growth, with significant opportunities emerging in developed regions like North America and Europe, and rapidly expanding healthcare sectors in Asia Pacific and Latin America.
Rfid Surgical Sponge Company Market Share

This comprehensive report offers an unparalleled deep dive into the RFID Surgical Sponge Market, providing critical insights for medical device manufacturers, healthcare providers, and investors. Leveraging high-volume keywords such as patient safety, retained surgical items (RSI) prevention, operating room efficiency, surgical workflow optimization, and medical device innovation, this study analyzes the robust growth trajectory from 2019 to 2033. Discover how RFID technology is transforming surgical environments, mitigating risks, and driving a new era of accountability in healthcare. The market is projected to witness significant expansion, driven by increasing adoption in hospitals and ambulatory surgery centers aiming to enhance patient outcomes and streamline operations.
The Study Period is 2019–2033, with the Base Year and Estimated Year both being 2025, and a Forecast Period extending from 2025–2033. Our analysis incorporates Historical Period data from 2019–2024, offering a complete picture of market evolution and future opportunities. All financial values are presented in millions for clear, large-scale understanding.
Rfid Surgical Sponge Market Structure & Competitive Landscape
The global RFID Surgical Sponge market demonstrates a moderately concentrated structure, with the top five players collectively accounting for an estimated 35.xx million in revenue in 2024, indicating a dynamic competitive environment. Innovation drivers are primarily focused on enhancing detection accuracy, reducing false positives, and improving integration with existing hospital information systems, leading to a surge in R&D investments, estimated at 2.xx million annually across key players. Regulatory impacts, particularly stringent guidelines from bodies like the FDA and CE, mandate robust safety protocols, thereby accelerating the adoption of RFID solutions as a gold standard for RSI prevention. While traditional manual counting methods serve as product substitutes, their inherent fallibility drives the shift towards automated RFID systems. End-user segmentation reveals a significant preference from large hospital systems due to their higher surgical volumes and greater emphasis on advanced patient safety protocols. Mergers and acquisitions (M&A) activity has seen a steady increase, with M&A volumes reaching an estimated 5.xx million in 2023, as companies seek to consolidate market share, acquire specialized RFID technologies, and expand their geographical footprint. Recent strategic alliances underscore a collaborative effort to standardize RFID protocols and enhance interoperability within complex surgical ecosystems. The market structure is continuously evolving with new entrants introducing innovative solutions, yet established players leverage their extensive distribution networks and strong relationships with healthcare institutions. The competitive landscape is also shaped by strategic partnerships between RFID technology providers and traditional surgical sponge manufacturers, creating integrated solutions that offer superior value propositions. This evolution reflects the industry’s commitment to addressing critical patient safety concerns and improving the overall efficiency of surgical operations on a global scale.
Rfid Surgical Sponge Market Trends & Opportunities
The RFID Surgical Sponge market is experiencing significant growth, projected to reach an estimated 200.xx million by 2033, up from 75.xx million in 2024, at a robust Compound Annual Growth Rate (CAGR) of 12.xx%. This expansion is fueled by critical trends such as the escalating focus on patient safety protocols and the increasing pressure on healthcare facilities to minimize surgical errors. Technological shifts are profoundly impacting the market, with advancements in ultra-high frequency (UHF) RFID tags offering enhanced read range and accuracy, even in challenging operating room environments. Miniaturization of RFID tags and improved antenna designs are enabling seamless integration into various surgical products without compromising their primary function. The development of cloud-based RFID tracking systems provides real-time data analytics, allowing hospitals to optimize inventory management and track surgical sponges from procurement to disposal, significantly improving operational efficiency.
Consumer preferences, particularly among surgical teams and hospital administrators, are shifting towards automated, reliable solutions that reduce human error and enhance accountability. The move from manual counts to digital verification systems is seen as a crucial step in modernizing surgical workflows and ensuring a safer environment for patients. This preference is also driven by the desire to reduce the financial burden associated with retained surgical items, which can incur millions in additional healthcare costs and litigation expenses annually. Competitive dynamics are intensifying as key players invest heavily in R&D to differentiate their offerings. Companies are focusing on developing comprehensive RFID ecosystems that include not just sponges but also other surgical instruments, aiming to provide a holistic solution for surgical item management. Market penetration rates for RFID surgical sponges are currently at an estimated 30.xx% in major healthcare markets, indicating substantial room for growth as awareness increases and regulatory mandates become more widespread. The opportunity lies in expanding into emerging markets and educating smaller healthcare facilities about the benefits and cost-effectiveness of these advanced safety solutions. Furthermore, the integration of RFID technology with artificial intelligence and machine learning is opening new avenues for predictive analytics, further enhancing patient safety and operational efficiency within the surgical suite. The market is also seeing a trend towards subscription-based models for RFID tracking solutions, which lowers the upfront cost for hospitals and encourages wider adoption.
Dominant Markets & Segments in Rfid Surgical Sponge
Within the RFID Surgical Sponge market, the Hospital segment under Application and the Sponge segment under Type currently hold dominant positions, reflecting their foundational role in surgical safety. North America, particularly the United States, stands as the leading regional market due to its advanced healthcare infrastructure, stringent regulatory landscape, and high adoption rates of cutting-edge medical technologies.
Dominant Application Segment: Hospital
- Infrastructure: Hospitals, especially large university-affiliated and community hospitals, perform the vast majority of complex surgical procedures. These institutions often have dedicated operating suites, high patient volumes, and substantial budgets for investing in advanced patient safety technologies.
- Policies & Regulations: North American and European hospitals are subject to rigorous regulatory mandates and accreditation standards (ee.g., Joint Commission, NHS guidelines) that strongly encourage, and in some cases require, the implementation of technologies to prevent retained surgical items. RFID sponges directly address these compliance requirements.
- Patient Safety Imperative: The high incidence and severe consequences of retained surgical items in hospital settings drive the urgent need for infallible detection systems. Hospitals are often at the forefront of adopting solutions that enhance patient safety, reduce liability, and improve their quality ratings.
- Technological Integration: Hospitals possess the IT infrastructure and resources necessary to integrate complex RFID tracking systems into their existing electronic health records (EHR) and inventory management systems, enabling seamless workflow adoption.
- Volume & Complexity: The sheer volume and complexity of surgeries performed in hospitals, ranging from general surgery to specialized cardiovascular and neurological procedures, necessitate robust and reliable tracking solutions.
Dominant Type Segment: Sponge
- High Incidence of Retention: Surgical sponges represent the most frequently retained item during surgery, accounting for an estimated 70% of all retained surgical items. This statistic alone makes them the primary focus for RFID integration efforts.
- Ubiquitous Use: Sponges are used in virtually every surgical procedure for absorption and retraction, making their reliable tracking a universal requirement across all specialties.
- Direct Patient Contact & Risk: Sponges are directly placed within the patient's body cavity, posing immediate and severe health risks if left behind, necessitating the highest level of vigilance and technological intervention.
- Historical Challenges: Manual counting of sponges has historically been prone to human error, leading to the development of RFID solutions specifically designed to overcome these limitations.
Regional Dominance: North America
- Advanced Healthcare Infrastructure: The United States and Canada boast highly developed healthcare systems with a strong emphasis on technological innovation and patient-centered care.
- Strong Regulatory Push: Regulatory bodies like the FDA actively promote and approve technologies that enhance patient safety, creating a favorable environment for RFID surgical sponge adoption.
- High Awareness & Adoption: Healthcare providers in North America are highly aware of the risks and costs associated with retained surgical items, leading to a proactive approach in adopting preventative measures.
- Investment Capacity: Higher healthcare expenditure per capita and greater investment capacity in cutting-edge medical technologies compared to many other regions.
- Presence of Key Players: Many leading RFID surgical sponge manufacturers and technology developers are headquartered or have significant operations in North America, driving market growth and innovation. The established distribution channels and supply networks further solidify its leadership position.
Rfid Surgical Sponge Product Analysis
RFID surgical sponges represent a pivotal advancement in medical technology, integrating tiny, washable RFID tags directly into sponges to enable precise, real-time tracking within the operating room. These innovative products offer an unparalleled competitive advantage by drastically reducing the incidence of retained surgical items (RSI), a leading cause of patient harm and costly litigations. Applications span all surgical specialties, from general surgery to orthopedics and gynecology, providing an extra layer of safety beyond traditional manual counts. Technological advancements focus on developing passive RFID tags that are biocompatible, easily integrated into various sponge types, and detectable through blood, tissue, and even within the patient's body cavity using specialized scanners. This ensures a comprehensive safety net, improving surgical workflow efficiency, enhancing inventory management, and ultimately safeguarding patient lives.
Key Drivers, Barriers & Challenges in Rfid Surgical Sponge
The RFID Surgical Sponge market is propelled by a confluence of critical drivers, including the paramount focus on patient safety and the imperative to eliminate retained surgical items. Technological advancements in RFID accuracy and integration capabilities further enhance its appeal. However, the market faces significant barriers such as the initial high cost of implementation, which can be a deterrent for smaller healthcare facilities, and the need for standardized protocols across diverse surgical environments. Challenges also arise from existing infrastructure limitations, requiring substantial investment in compatible scanning equipment and IT systems.
Growth Drivers in the Rfid Surgical Sponge Market
The growth in the RFID Surgical Sponge market is significantly propelled by several key factors. The primary driver is the global emphasis on enhancing patient safety and reducing the incidence of retained surgical items (RSI), which remain a persistent concern in operating rooms worldwide. Regulatory pressures and accreditation standards from bodies like The Joint Commission and national health agencies are increasingly mandating advanced safety protocols, making RFID solutions a compelling choice for compliance. Technologically, continuous advancements in RFID tag miniaturization, improved detection accuracy, and enhanced system integration capabilities are making these solutions more reliable and user-friendly. Economically, the long-term cost savings associated with preventing RSI, including reduced re-operations, extended hospital stays, legal settlements, and reputational damage, heavily outweigh the initial investment, driving wider adoption. Furthermore, the growing awareness among healthcare providers regarding the benefits of automated tracking systems for surgical workflow efficiency and inventory management contributes substantially to market expansion.
Challenges Impacting Rfid Surgical Sponge Growth
Despite significant drivers, the RFID Surgical Sponge market faces notable challenges and restraints. A primary barrier is the high initial capital investment required for RFID infrastructure, including specialized scanners, software integration, and tag-enabled sponges. For instance, implementing a comprehensive RFID system across a medium-sized hospital could cost an estimated 1.xx million, posing a substantial financial hurdle, particularly for smaller hospitals or those with budget constraints. Another significant challenge is the lack of standardized protocols and interoperability across different RFID systems and healthcare facilities, hindering widespread adoption and seamless data exchange. Regulatory complexities and varied compliance requirements across different regions can also create market fragmentation and slow down product development and market entry. Furthermore, supply chain issues, particularly regarding the sourcing and integration of specialized RFID tags and components, can lead to production delays and increased costs, impacting overall market growth. Competitive pressures from established manual counting methods and other non-RFID alternative technologies, combined with the need for extensive staff training and resistance to change, also act as significant restraints. For example, overcoming staff reluctance to adopt new technologies can delay full system implementation by an estimated 12 to 18 months in some facilities, impacting adoption rates.
Key Players Shaping the Rfid Surgical Sponge Market
- Medtronic
- Stryker Corporation
- STERIS
- Cardinal Health
- Medline
- Integra LifeSciences
- Tally Surgical
- NHP Surgi-Pak
- Custom Hospital Products
- SDP INC.
- AllCare
- Teleflex Medical
- Amer Surg
- OWENS & MINOR INC
- BioSEAL
Significant Rfid Surgical Sponge Industry Milestones
- 2019 Q3: Introduction of next-generation UHF RFID surgical sponges by a leading player, significantly improving detection range and accuracy.
- 2020 Q1: Major regulatory body (e.g., FDA or European equivalent) issues updated guidelines emphasizing advanced technologies for RSI prevention, boosting market confidence.
- 2021 Q2: First successful large-scale implementation of integrated RFID surgical sponge tracking system across a multi-hospital network, demonstrating enhanced workflow and patient safety.
- 2022 Q4: A strategic merger between a top RFID technology firm and a traditional medical consumables manufacturer, aiming to create comprehensive smart surgical solutions.
- 2023 Q3: Launch of cloud-based RFID data analytics platform, offering real-time insights into surgical sponge inventory and usage patterns for hospitals.
- 2024 Q2: Expansion of RFID surgical sponge product lines to include specialized tags for different surgical environments and sponge types, increasing market penetration.
Future Outlook for Rfid Surgical Sponge Market
The future outlook for the RFID Surgical Sponge market is exceptionally promising, poised for sustained growth driven by persistent global efforts to enhance patient safety and improve surgical outcomes. Key growth catalysts include increasing regulatory mandates for RSI prevention, rapid technological advancements in RFID capabilities (such as improved accuracy and smaller form factors), and growing awareness among healthcare providers regarding the significant long-term cost savings associated with preventing surgical errors. Strategic opportunities lie in expanding market penetration in emerging economies, developing integrated solutions that combine RFID sponges with other surgical tracking technologies, and leveraging AI and machine learning for predictive analytics in the operating room. The market potential is vast, as healthcare systems worldwide continue to prioritize efficiency, accountability, and the ultimate well-being of patients through innovative medical device solutions.
Rfid Surgical Sponge Segmentation
-
1. Application
- 1.1. Hospital
- 1.2. Ambulatory Surgery Center
- 1.3. Others
-
2. Type
- 2.1. Sponge
- 2.2. Towels
- 2.3. Gauze
- 2.4. Others
Rfid Surgical Sponge Segmentation By Geography
-
1. North America
- 1.1. United States
- 1.2. Canada
- 1.3. Mexico
-
2. South America
- 2.1. Brazil
- 2.2. Argentina
- 2.3. Rest of South America
-
3. Europe
- 3.1. United Kingdom
- 3.2. Germany
- 3.3. France
- 3.4. Italy
- 3.5. Spain
- 3.6. Russia
- 3.7. Benelux
- 3.8. Nordics
- 3.9. Rest of Europe
-
4. Middle East & Africa
- 4.1. Turkey
- 4.2. Israel
- 4.3. GCC
- 4.4. North Africa
- 4.5. South Africa
- 4.6. Rest of Middle East & Africa
-
5. Asia Pacific
- 5.1. China
- 5.2. India
- 5.3. Japan
- 5.4. South Korea
- 5.5. ASEAN
- 5.6. Oceania
- 5.7. Rest of Asia Pacific
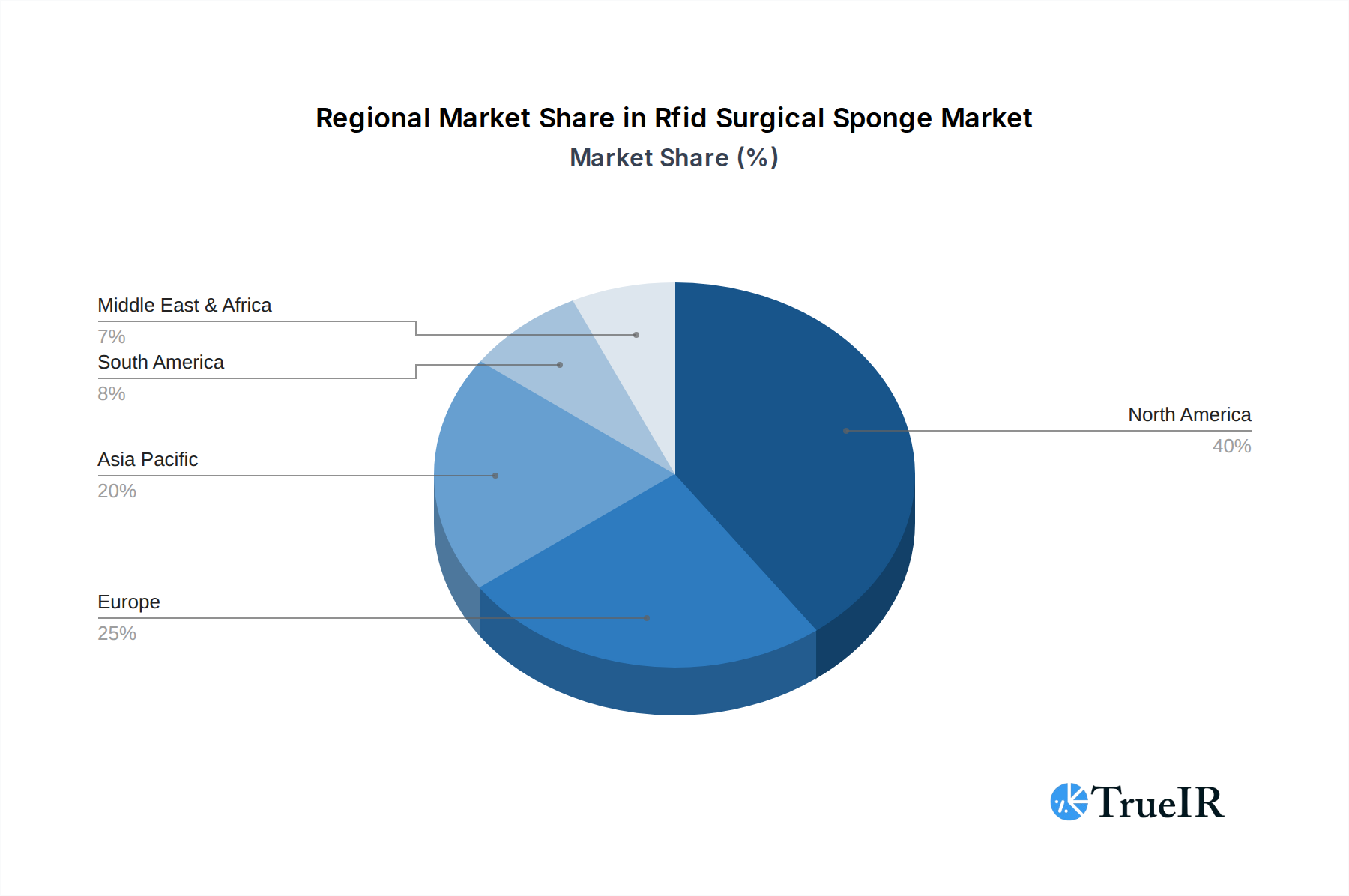
Rfid Surgical Sponge Regional Market Share

Geographic Coverage of Rfid Surgical Sponge
Rfid Surgical Sponge REPORT HIGHLIGHTS
| Aspects | Details |
|---|---|
| Study Period | 2020-2034 |
| Base Year | 2025 |
| Estimated Year | 2026 |
| Forecast Period | 2026-2034 |
| Historical Period | 2020-2025 |
| Growth Rate | CAGR of 12% from 2020-2034 |
| Segmentation |
|
Table of Contents
- 1. Introduction
- 1.1. Research Scope
- 1.2. Market Segmentation
- 1.3. Research Objective
- 1.4. Definitions and Assumptions
- 2. Executive Summary
- 2.1. Market Snapshot
- 3. Market Dynamics
- 3.1. Market Drivers
- 3.2. Market Restrains
- 3.3. Market Trends
- 3.4. Market Opportunities
- 4. Market Factor Analysis
- 4.1. Porters Five Forces
- 4.1.1. Bargaining Power of Suppliers
- 4.1.2. Bargaining Power of Buyers
- 4.1.3. Threat of New Entrants
- 4.1.4. Threat of Substitutes
- 4.1.5. Competitive Rivalry
- 4.2. PESTEL analysis
- 4.3. BCG Analysis
- 4.3.1. Stars (High Growth, High Market Share)
- 4.3.2. Cash Cows (Low Growth, High Market Share)
- 4.3.3. Question Mark (High Growth, Low Market Share)
- 4.3.4. Dogs (Low Growth, Low Market Share)
- 4.4. Ansoff Matrix Analysis
- 4.5. Supply Chain Analysis
- 4.6. Regulatory Landscape
- 4.7. Current Market Potential and Opportunity Assessment (TAM–SAM–SOM Framework)
- 4.8. TIR Analyst Note
- 4.1. Porters Five Forces
- 5. Market Analysis, Insights and Forecast 2021-2033
- 5.1. Market Analysis, Insights and Forecast - by Application
- 5.1.1. Hospital
- 5.1.2. Ambulatory Surgery Center
- 5.1.3. Others
- 5.2. Market Analysis, Insights and Forecast - by Type
- 5.2.1. Sponge
- 5.2.2. Towels
- 5.2.3. Gauze
- 5.2.4. Others
- 5.3. Market Analysis, Insights and Forecast - by Region
- 5.3.1. North America
- 5.3.2. South America
- 5.3.3. Europe
- 5.3.4. Middle East & Africa
- 5.3.5. Asia Pacific
- 5.1. Market Analysis, Insights and Forecast - by Application
- 6. Global Rfid Surgical Sponge Analysis, Insights and Forecast, 2021-2033
- 6.1. Market Analysis, Insights and Forecast - by Application
- 6.1.1. Hospital
- 6.1.2. Ambulatory Surgery Center
- 6.1.3. Others
- 6.2. Market Analysis, Insights and Forecast - by Type
- 6.2.1. Sponge
- 6.2.2. Towels
- 6.2.3. Gauze
- 6.2.4. Others
- 6.1. Market Analysis, Insights and Forecast - by Application
- 7. North America Rfid Surgical Sponge Analysis, Insights and Forecast, 2020-2032
- 7.1. Market Analysis, Insights and Forecast - by Application
- 7.1.1. Hospital
- 7.1.2. Ambulatory Surgery Center
- 7.1.3. Others
- 7.2. Market Analysis, Insights and Forecast - by Type
- 7.2.1. Sponge
- 7.2.2. Towels
- 7.2.3. Gauze
- 7.2.4. Others
- 7.1. Market Analysis, Insights and Forecast - by Application
- 8. South America Rfid Surgical Sponge Analysis, Insights and Forecast, 2020-2032
- 8.1. Market Analysis, Insights and Forecast - by Application
- 8.1.1. Hospital
- 8.1.2. Ambulatory Surgery Center
- 8.1.3. Others
- 8.2. Market Analysis, Insights and Forecast - by Type
- 8.2.1. Sponge
- 8.2.2. Towels
- 8.2.3. Gauze
- 8.2.4. Others
- 8.1. Market Analysis, Insights and Forecast - by Application
- 9. Europe Rfid Surgical Sponge Analysis, Insights and Forecast, 2020-2032
- 9.1. Market Analysis, Insights and Forecast - by Application
- 9.1.1. Hospital
- 9.1.2. Ambulatory Surgery Center
- 9.1.3. Others
- 9.2. Market Analysis, Insights and Forecast - by Type
- 9.2.1. Sponge
- 9.2.2. Towels
- 9.2.3. Gauze
- 9.2.4. Others
- 9.1. Market Analysis, Insights and Forecast - by Application
- 10. Middle East & Africa Rfid Surgical Sponge Analysis, Insights and Forecast, 2020-2032
- 10.1. Market Analysis, Insights and Forecast - by Application
- 10.1.1. Hospital
- 10.1.2. Ambulatory Surgery Center
- 10.1.3. Others
- 10.2. Market Analysis, Insights and Forecast - by Type
- 10.2.1. Sponge
- 10.2.2. Towels
- 10.2.3. Gauze
- 10.2.4. Others
- 10.1. Market Analysis, Insights and Forecast - by Application
- 11. Asia Pacific Rfid Surgical Sponge Analysis, Insights and Forecast, 2020-2032
- 11.1. Market Analysis, Insights and Forecast - by Application
- 11.1.1. Hospital
- 11.1.2. Ambulatory Surgery Center
- 11.1.3. Others
- 11.2. Market Analysis, Insights and Forecast - by Type
- 11.2.1. Sponge
- 11.2.2. Towels
- 11.2.3. Gauze
- 11.2.4. Others
- 11.1. Market Analysis, Insights and Forecast - by Application
- 12. Competitive Analysis
- 12.1. Company Profiles
- 12.1.1 Medtronic
- 12.1.1.1. Company Overview
- 12.1.1.2. Products
- 12.1.1.3. Company Financials
- 12.1.1.4. SWOT Analysis
- 12.1.2 Stryker Corporation
- 12.1.2.1. Company Overview
- 12.1.2.2. Products
- 12.1.2.3. Company Financials
- 12.1.2.4. SWOT Analysis
- 12.1.3 STERIS
- 12.1.3.1. Company Overview
- 12.1.3.2. Products
- 12.1.3.3. Company Financials
- 12.1.3.4. SWOT Analysis
- 12.1.4 Cardinal Health
- 12.1.4.1. Company Overview
- 12.1.4.2. Products
- 12.1.4.3. Company Financials
- 12.1.4.4. SWOT Analysis
- 12.1.5 Medline
- 12.1.5.1. Company Overview
- 12.1.5.2. Products
- 12.1.5.3. Company Financials
- 12.1.5.4. SWOT Analysis
- 12.1.6 Integra LifeSciences
- 12.1.6.1. Company Overview
- 12.1.6.2. Products
- 12.1.6.3. Company Financials
- 12.1.6.4. SWOT Analysis
- 12.1.7 Tally Surgical
- 12.1.7.1. Company Overview
- 12.1.7.2. Products
- 12.1.7.3. Company Financials
- 12.1.7.4. SWOT Analysis
- 12.1.8 NHP Surgi-Pak
- 12.1.8.1. Company Overview
- 12.1.8.2. Products
- 12.1.8.3. Company Financials
- 12.1.8.4. SWOT Analysis
- 12.1.9 Custom Hospital Products
- 12.1.9.1. Company Overview
- 12.1.9.2. Products
- 12.1.9.3. Company Financials
- 12.1.9.4. SWOT Analysis
- 12.1.10 SDP INC.
- 12.1.10.1. Company Overview
- 12.1.10.2. Products
- 12.1.10.3. Company Financials
- 12.1.10.4. SWOT Analysis
- 12.1.11 AllCare
- 12.1.11.1. Company Overview
- 12.1.11.2. Products
- 12.1.11.3. Company Financials
- 12.1.11.4. SWOT Analysis
- 12.1.12 Teleflex Medical
- 12.1.12.1. Company Overview
- 12.1.12.2. Products
- 12.1.12.3. Company Financials
- 12.1.12.4. SWOT Analysis
- 12.1.13 Amer Surg
- 12.1.13.1. Company Overview
- 12.1.13.2. Products
- 12.1.13.3. Company Financials
- 12.1.13.4. SWOT Analysis
- 12.1.14 OWENS & MINOR INC
- 12.1.14.1. Company Overview
- 12.1.14.2. Products
- 12.1.14.3. Company Financials
- 12.1.14.4. SWOT Analysis
- 12.1.15 BioSEAL
- 12.1.15.1. Company Overview
- 12.1.15.2. Products
- 12.1.15.3. Company Financials
- 12.1.15.4. SWOT Analysis
- 12.1.1 Medtronic
- 12.2. Market Entropy
- 12.2.1 Company's Key Areas Served
- 12.2.2 Recent Developments
- 12.3. Company Market Share Analysis 2025
- 12.3.1 Top 5 Companies Market Share Analysis
- 12.3.2 Top 3 Companies Market Share Analysis
- 12.4. List of Potential Customers
- 13. Research Methodology
List of Figures
- Figure 1: Global Rfid Surgical Sponge Revenue Breakdown (undefined, %) by Region 2025 & 2033
- Figure 2: Global Rfid Surgical Sponge Volume Breakdown (K, %) by Region 2025 & 2033
- Figure 3: North America Rfid Surgical Sponge Revenue (undefined), by Application 2025 & 2033
- Figure 4: North America Rfid Surgical Sponge Volume (K), by Application 2025 & 2033
- Figure 5: North America Rfid Surgical Sponge Revenue Share (%), by Application 2025 & 2033
- Figure 6: North America Rfid Surgical Sponge Volume Share (%), by Application 2025 & 2033
- Figure 7: North America Rfid Surgical Sponge Revenue (undefined), by Type 2025 & 2033
- Figure 8: North America Rfid Surgical Sponge Volume (K), by Type 2025 & 2033
- Figure 9: North America Rfid Surgical Sponge Revenue Share (%), by Type 2025 & 2033
- Figure 10: North America Rfid Surgical Sponge Volume Share (%), by Type 2025 & 2033
- Figure 11: North America Rfid Surgical Sponge Revenue (undefined), by Country 2025 & 2033
- Figure 12: North America Rfid Surgical Sponge Volume (K), by Country 2025 & 2033
- Figure 13: North America Rfid Surgical Sponge Revenue Share (%), by Country 2025 & 2033
- Figure 14: North America Rfid Surgical Sponge Volume Share (%), by Country 2025 & 2033
- Figure 15: South America Rfid Surgical Sponge Revenue (undefined), by Application 2025 & 2033
- Figure 16: South America Rfid Surgical Sponge Volume (K), by Application 2025 & 2033
- Figure 17: South America Rfid Surgical Sponge Revenue Share (%), by Application 2025 & 2033
- Figure 18: South America Rfid Surgical Sponge Volume Share (%), by Application 2025 & 2033
- Figure 19: South America Rfid Surgical Sponge Revenue (undefined), by Type 2025 & 2033
- Figure 20: South America Rfid Surgical Sponge Volume (K), by Type 2025 & 2033
- Figure 21: South America Rfid Surgical Sponge Revenue Share (%), by Type 2025 & 2033
- Figure 22: South America Rfid Surgical Sponge Volume Share (%), by Type 2025 & 2033
- Figure 23: South America Rfid Surgical Sponge Revenue (undefined), by Country 2025 & 2033
- Figure 24: South America Rfid Surgical Sponge Volume (K), by Country 2025 & 2033
- Figure 25: South America Rfid Surgical Sponge Revenue Share (%), by Country 2025 & 2033
- Figure 26: South America Rfid Surgical Sponge Volume Share (%), by Country 2025 & 2033
- Figure 27: Europe Rfid Surgical Sponge Revenue (undefined), by Application 2025 & 2033
- Figure 28: Europe Rfid Surgical Sponge Volume (K), by Application 2025 & 2033
- Figure 29: Europe Rfid Surgical Sponge Revenue Share (%), by Application 2025 & 2033
- Figure 30: Europe Rfid Surgical Sponge Volume Share (%), by Application 2025 & 2033
- Figure 31: Europe Rfid Surgical Sponge Revenue (undefined), by Type 2025 & 2033
- Figure 32: Europe Rfid Surgical Sponge Volume (K), by Type 2025 & 2033
- Figure 33: Europe Rfid Surgical Sponge Revenue Share (%), by Type 2025 & 2033
- Figure 34: Europe Rfid Surgical Sponge Volume Share (%), by Type 2025 & 2033
- Figure 35: Europe Rfid Surgical Sponge Revenue (undefined), by Country 2025 & 2033
- Figure 36: Europe Rfid Surgical Sponge Volume (K), by Country 2025 & 2033
- Figure 37: Europe Rfid Surgical Sponge Revenue Share (%), by Country 2025 & 2033
- Figure 38: Europe Rfid Surgical Sponge Volume Share (%), by Country 2025 & 2033
- Figure 39: Middle East & Africa Rfid Surgical Sponge Revenue (undefined), by Application 2025 & 2033
- Figure 40: Middle East & Africa Rfid Surgical Sponge Volume (K), by Application 2025 & 2033
- Figure 41: Middle East & Africa Rfid Surgical Sponge Revenue Share (%), by Application 2025 & 2033
- Figure 42: Middle East & Africa Rfid Surgical Sponge Volume Share (%), by Application 2025 & 2033
- Figure 43: Middle East & Africa Rfid Surgical Sponge Revenue (undefined), by Type 2025 & 2033
- Figure 44: Middle East & Africa Rfid Surgical Sponge Volume (K), by Type 2025 & 2033
- Figure 45: Middle East & Africa Rfid Surgical Sponge Revenue Share (%), by Type 2025 & 2033
- Figure 46: Middle East & Africa Rfid Surgical Sponge Volume Share (%), by Type 2025 & 2033
- Figure 47: Middle East & Africa Rfid Surgical Sponge Revenue (undefined), by Country 2025 & 2033
- Figure 48: Middle East & Africa Rfid Surgical Sponge Volume (K), by Country 2025 & 2033
- Figure 49: Middle East & Africa Rfid Surgical Sponge Revenue Share (%), by Country 2025 & 2033
- Figure 50: Middle East & Africa Rfid Surgical Sponge Volume Share (%), by Country 2025 & 2033
- Figure 51: Asia Pacific Rfid Surgical Sponge Revenue (undefined), by Application 2025 & 2033
- Figure 52: Asia Pacific Rfid Surgical Sponge Volume (K), by Application 2025 & 2033
- Figure 53: Asia Pacific Rfid Surgical Sponge Revenue Share (%), by Application 2025 & 2033
- Figure 54: Asia Pacific Rfid Surgical Sponge Volume Share (%), by Application 2025 & 2033
- Figure 55: Asia Pacific Rfid Surgical Sponge Revenue (undefined), by Type 2025 & 2033
- Figure 56: Asia Pacific Rfid Surgical Sponge Volume (K), by Type 2025 & 2033
- Figure 57: Asia Pacific Rfid Surgical Sponge Revenue Share (%), by Type 2025 & 2033
- Figure 58: Asia Pacific Rfid Surgical Sponge Volume Share (%), by Type 2025 & 2033
- Figure 59: Asia Pacific Rfid Surgical Sponge Revenue (undefined), by Country 2025 & 2033
- Figure 60: Asia Pacific Rfid Surgical Sponge Volume (K), by Country 2025 & 2033
- Figure 61: Asia Pacific Rfid Surgical Sponge Revenue Share (%), by Country 2025 & 2033
- Figure 62: Asia Pacific Rfid Surgical Sponge Volume Share (%), by Country 2025 & 2033
List of Tables
- Table 1: Global Rfid Surgical Sponge Revenue undefined Forecast, by Application 2020 & 2033
- Table 2: Global Rfid Surgical Sponge Volume K Forecast, by Application 2020 & 2033
- Table 3: Global Rfid Surgical Sponge Revenue undefined Forecast, by Type 2020 & 2033
- Table 4: Global Rfid Surgical Sponge Volume K Forecast, by Type 2020 & 2033
- Table 5: Global Rfid Surgical Sponge Revenue undefined Forecast, by Region 2020 & 2033
- Table 6: Global Rfid Surgical Sponge Volume K Forecast, by Region 2020 & 2033
- Table 7: Global Rfid Surgical Sponge Revenue undefined Forecast, by Application 2020 & 2033
- Table 8: Global Rfid Surgical Sponge Volume K Forecast, by Application 2020 & 2033
- Table 9: Global Rfid Surgical Sponge Revenue undefined Forecast, by Type 2020 & 2033
- Table 10: Global Rfid Surgical Sponge Volume K Forecast, by Type 2020 & 2033
- Table 11: Global Rfid Surgical Sponge Revenue undefined Forecast, by Country 2020 & 2033
- Table 12: Global Rfid Surgical Sponge Volume K Forecast, by Country 2020 & 2033
- Table 13: United States Rfid Surgical Sponge Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 14: United States Rfid Surgical Sponge Volume (K) Forecast, by Application 2020 & 2033
- Table 15: Canada Rfid Surgical Sponge Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 16: Canada Rfid Surgical Sponge Volume (K) Forecast, by Application 2020 & 2033
- Table 17: Mexico Rfid Surgical Sponge Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 18: Mexico Rfid Surgical Sponge Volume (K) Forecast, by Application 2020 & 2033
- Table 19: Global Rfid Surgical Sponge Revenue undefined Forecast, by Application 2020 & 2033
- Table 20: Global Rfid Surgical Sponge Volume K Forecast, by Application 2020 & 2033
- Table 21: Global Rfid Surgical Sponge Revenue undefined Forecast, by Type 2020 & 2033
- Table 22: Global Rfid Surgical Sponge Volume K Forecast, by Type 2020 & 2033
- Table 23: Global Rfid Surgical Sponge Revenue undefined Forecast, by Country 2020 & 2033
- Table 24: Global Rfid Surgical Sponge Volume K Forecast, by Country 2020 & 2033
- Table 25: Brazil Rfid Surgical Sponge Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 26: Brazil Rfid Surgical Sponge Volume (K) Forecast, by Application 2020 & 2033
- Table 27: Argentina Rfid Surgical Sponge Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 28: Argentina Rfid Surgical Sponge Volume (K) Forecast, by Application 2020 & 2033
- Table 29: Rest of South America Rfid Surgical Sponge Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 30: Rest of South America Rfid Surgical Sponge Volume (K) Forecast, by Application 2020 & 2033
- Table 31: Global Rfid Surgical Sponge Revenue undefined Forecast, by Application 2020 & 2033
- Table 32: Global Rfid Surgical Sponge Volume K Forecast, by Application 2020 & 2033
- Table 33: Global Rfid Surgical Sponge Revenue undefined Forecast, by Type 2020 & 2033
- Table 34: Global Rfid Surgical Sponge Volume K Forecast, by Type 2020 & 2033
- Table 35: Global Rfid Surgical Sponge Revenue undefined Forecast, by Country 2020 & 2033
- Table 36: Global Rfid Surgical Sponge Volume K Forecast, by Country 2020 & 2033
- Table 37: United Kingdom Rfid Surgical Sponge Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 38: United Kingdom Rfid Surgical Sponge Volume (K) Forecast, by Application 2020 & 2033
- Table 39: Germany Rfid Surgical Sponge Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 40: Germany Rfid Surgical Sponge Volume (K) Forecast, by Application 2020 & 2033
- Table 41: France Rfid Surgical Sponge Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 42: France Rfid Surgical Sponge Volume (K) Forecast, by Application 2020 & 2033
- Table 43: Italy Rfid Surgical Sponge Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 44: Italy Rfid Surgical Sponge Volume (K) Forecast, by Application 2020 & 2033
- Table 45: Spain Rfid Surgical Sponge Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 46: Spain Rfid Surgical Sponge Volume (K) Forecast, by Application 2020 & 2033
- Table 47: Russia Rfid Surgical Sponge Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 48: Russia Rfid Surgical Sponge Volume (K) Forecast, by Application 2020 & 2033
- Table 49: Benelux Rfid Surgical Sponge Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 50: Benelux Rfid Surgical Sponge Volume (K) Forecast, by Application 2020 & 2033
- Table 51: Nordics Rfid Surgical Sponge Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 52: Nordics Rfid Surgical Sponge Volume (K) Forecast, by Application 2020 & 2033
- Table 53: Rest of Europe Rfid Surgical Sponge Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 54: Rest of Europe Rfid Surgical Sponge Volume (K) Forecast, by Application 2020 & 2033
- Table 55: Global Rfid Surgical Sponge Revenue undefined Forecast, by Application 2020 & 2033
- Table 56: Global Rfid Surgical Sponge Volume K Forecast, by Application 2020 & 2033
- Table 57: Global Rfid Surgical Sponge Revenue undefined Forecast, by Type 2020 & 2033
- Table 58: Global Rfid Surgical Sponge Volume K Forecast, by Type 2020 & 2033
- Table 59: Global Rfid Surgical Sponge Revenue undefined Forecast, by Country 2020 & 2033
- Table 60: Global Rfid Surgical Sponge Volume K Forecast, by Country 2020 & 2033
- Table 61: Turkey Rfid Surgical Sponge Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 62: Turkey Rfid Surgical Sponge Volume (K) Forecast, by Application 2020 & 2033
- Table 63: Israel Rfid Surgical Sponge Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 64: Israel Rfid Surgical Sponge Volume (K) Forecast, by Application 2020 & 2033
- Table 65: GCC Rfid Surgical Sponge Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 66: GCC Rfid Surgical Sponge Volume (K) Forecast, by Application 2020 & 2033
- Table 67: North Africa Rfid Surgical Sponge Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 68: North Africa Rfid Surgical Sponge Volume (K) Forecast, by Application 2020 & 2033
- Table 69: South Africa Rfid Surgical Sponge Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 70: South Africa Rfid Surgical Sponge Volume (K) Forecast, by Application 2020 & 2033
- Table 71: Rest of Middle East & Africa Rfid Surgical Sponge Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 72: Rest of Middle East & Africa Rfid Surgical Sponge Volume (K) Forecast, by Application 2020 & 2033
- Table 73: Global Rfid Surgical Sponge Revenue undefined Forecast, by Application 2020 & 2033
- Table 74: Global Rfid Surgical Sponge Volume K Forecast, by Application 2020 & 2033
- Table 75: Global Rfid Surgical Sponge Revenue undefined Forecast, by Type 2020 & 2033
- Table 76: Global Rfid Surgical Sponge Volume K Forecast, by Type 2020 & 2033
- Table 77: Global Rfid Surgical Sponge Revenue undefined Forecast, by Country 2020 & 2033
- Table 78: Global Rfid Surgical Sponge Volume K Forecast, by Country 2020 & 2033
- Table 79: China Rfid Surgical Sponge Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 80: China Rfid Surgical Sponge Volume (K) Forecast, by Application 2020 & 2033
- Table 81: India Rfid Surgical Sponge Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 82: India Rfid Surgical Sponge Volume (K) Forecast, by Application 2020 & 2033
- Table 83: Japan Rfid Surgical Sponge Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 84: Japan Rfid Surgical Sponge Volume (K) Forecast, by Application 2020 & 2033
- Table 85: South Korea Rfid Surgical Sponge Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 86: South Korea Rfid Surgical Sponge Volume (K) Forecast, by Application 2020 & 2033
- Table 87: ASEAN Rfid Surgical Sponge Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 88: ASEAN Rfid Surgical Sponge Volume (K) Forecast, by Application 2020 & 2033
- Table 89: Oceania Rfid Surgical Sponge Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 90: Oceania Rfid Surgical Sponge Volume (K) Forecast, by Application 2020 & 2033
- Table 91: Rest of Asia Pacific Rfid Surgical Sponge Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 92: Rest of Asia Pacific Rfid Surgical Sponge Volume (K) Forecast, by Application 2020 & 2033
Frequently Asked Questions
1. What is the projected Compound Annual Growth Rate (CAGR) of the Rfid Surgical Sponge?
The projected CAGR is approximately 12%.
2. Which companies are prominent players in the Rfid Surgical Sponge?
Key companies in the market include Medtronic, Stryker Corporation, STERIS, Cardinal Health, Medline, Integra LifeSciences, Tally Surgical, NHP Surgi-Pak, Custom Hospital Products, SDP INC., AllCare, Teleflex Medical, Amer Surg, OWENS & MINOR INC, BioSEAL.
3. What are the main segments of the Rfid Surgical Sponge?
The market segments include Application, Type.
4. Can you provide details about the market size?
The market size is estimated to be USD XXX N/A as of 2022.
5. What are some drivers contributing to market growth?
N/A
6. What are the notable trends driving market growth?
N/A
7. Are there any restraints impacting market growth?
N/A
8. Can you provide examples of recent developments in the market?
N/A
9. What pricing options are available for accessing the report?
Pricing options include single-user, multi-user, and enterprise licenses priced at USD 3950.00, USD 5925.00, and USD 7900.00 respectively.
10. Is the market size provided in terms of value or volume?
The market size is provided in terms of value, measured in N/A and volume, measured in K.
11. Are there any specific market keywords associated with the report?
Yes, the market keyword associated with the report is "Rfid Surgical Sponge," which aids in identifying and referencing the specific market segment covered.
12. How do I determine which pricing option suits my needs best?
The pricing options vary based on user requirements and access needs. Individual users may opt for single-user licenses, while businesses requiring broader access may choose multi-user or enterprise licenses for cost-effective access to the report.
13. Are there any additional resources or data provided in the Rfid Surgical Sponge report?
While the report offers comprehensive insights, it's advisable to review the specific contents or supplementary materials provided to ascertain if additional resources or data are available.
14. How can I stay updated on further developments or reports in the Rfid Surgical Sponge?
To stay informed about further developments, trends, and reports in the Rfid Surgical Sponge, consider subscribing to industry newsletters, following relevant companies and organizations, or regularly checking reputable industry news sources and publications.
Methodology
Step 1 - Identification of Relevant Samples Size from Population Database



Step 2 - Approaches for Defining Global Market Size (Value, Volume* & Price*)

Note*: In applicable scenarios
Step 3 - Data Sources
Primary Research
- Web Analytics
- Survey Reports
- Research Institute
- Latest Research Reports
- Opinion Leaders
Secondary Research
- Annual Reports
- White Paper
- Latest Press Release
- Industry Association
- Paid Database
- Investor Presentations

Step 4 - Data Triangulation
Involves using different sources of information in order to increase the validity of a study
These sources are likely to be stakeholders in a program - participants, other researchers, program staff, other community members, and so on.
Then we put all data in single framework & apply various statistical tools to find out the dynamic on the market.
During the analysis stage, feedback from the stakeholder groups would be compared to determine areas of agreement as well as areas of divergence


