Key Insights
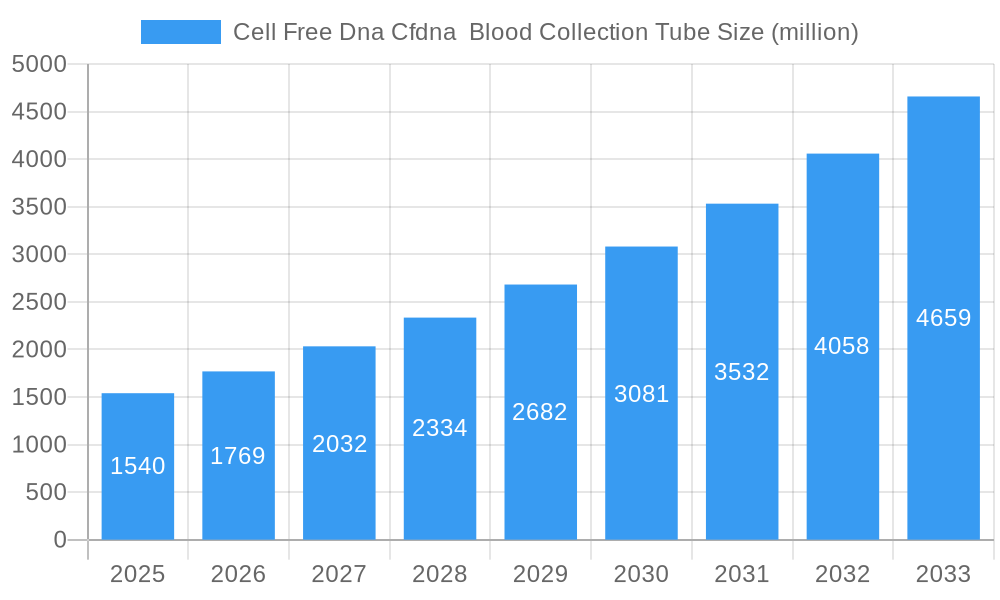
The Cell Free DNA (cfDNA) Blood Collection Tube market is poised for significant expansion, projected to reach an estimated $1.54 billion in 2025. This robust growth is fueled by a remarkable compound annual growth rate (CAGR) of 14.85% anticipated from 2025 through 2033. The increasing demand for non-invasive diagnostic methods, particularly in oncology for liquid biopsies, is a primary driver. Advancements in molecular diagnostics and the growing adoption of cfDNA analysis for prenatal testing, transplant monitoring, and infectious disease detection are further propelling market momentum. The trend towards personalized medicine and the expanding applications of cfDNA in scientific research, including genomic studies and drug development, are creating substantial opportunities for market players. The market is segmented by application, with In Vitro Diagnostics and Scientific Research being key areas, and by type, with 5ml and 10ml tubes being prevalent offerings.
Cell Free Dna Cfdna Blood Collection Tube Market Size (In Billion)

While the market benefits from strong growth drivers, certain restraints need to be considered. The high cost associated with cfDNA isolation and analysis technologies can be a barrier to widespread adoption, especially in resource-limited settings. Stringent regulatory hurdles for novel diagnostic tests and the need for extensive clinical validation also contribute to the market's challenges. However, ongoing technological innovations are aimed at reducing costs and improving efficiency, and the increasing understanding of cfDNA's diagnostic potential is likely to overcome these restraints. Key companies like Roche, Thermo Fisher Scientific, and Streck are actively investing in research and development, introducing advanced collection tubes that enhance sample stability and yield, thus supporting the market's upward trajectory. Geographically, North America and Europe currently dominate the market, driven by advanced healthcare infrastructure and high R&D spending, with Asia Pacific showing promising growth potential.
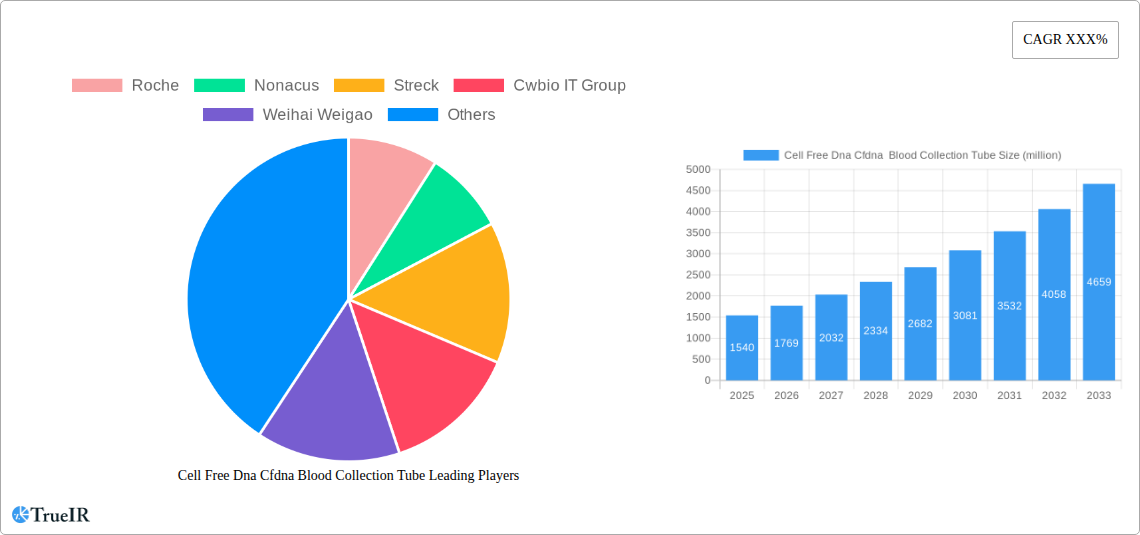
Cell Free Dna Cfdna Blood Collection Tube Company Market Share

Cell Free Dna Cfdna Blood Collection Tube Market Structure & Competitive Landscape
The global Cell Free DNA (cfDNA) blood collection tube market exhibits a dynamic and evolving competitive landscape, characterized by a blend of established giants and innovative niche players. Market concentration, while present, is not overly consolidated, with a significant number of companies vying for market share. Innovation is a primary driver, fueled by the increasing demand for advanced diagnostics and research applications. Regulatory impacts, particularly concerning the approval pathways for diagnostic kits utilizing cfDNA, also play a crucial role in shaping market entry and product development. While direct product substitutes are limited, advancements in alternative sample types or entirely different diagnostic methodologies could represent potential future threats. End-user segmentation is diverse, spanning in vitro diagnostics and scientific research, each with distinct needs and purchasing behaviors. Mergers and acquisitions (M&A) are observed as companies seek to expand their portfolios, gain access to new technologies, or strengthen their market presence. For instance, the historical period from 2019 to 2024 has witnessed approximately xx billion USD in M&A activity within this specialized sector. Concentration ratios, as measured by the combined market share of the top 5 players, are estimated to be around xx%, indicating a moderately competitive environment.
Cell Free Dna Cfdna Blood Collection Tube Market Trends & Opportunities
The Cell Free DNA (cfDNA) blood collection tube market is poised for substantial growth, driven by a confluence of technological advancements, expanding diagnostic applications, and increasing global healthcare investments. The market size is projected to surge from approximately xx billion USD in 2019 to an estimated xx billion USD by 2033, exhibiting a robust Compound Annual Growth Rate (CAGR) of xx% during the forecast period of 2025–2033. This significant expansion is underpinned by a paradigm shift in non-invasive diagnostic techniques, where cfDNA analysis plays a pivotal role in early disease detection, monitoring, and treatment efficacy assessment.
Technological shifts are a central theme, with ongoing refinements in tube formulations aimed at enhancing cfDNA yield and stability, minimizing pre-analytical variability, and ensuring superior sample integrity for downstream analyses. Innovations in inhibitor chemistry and blood stabilization matrices are crucial for accurate molecular profiling. Consumer preferences are increasingly aligned with less invasive procedures, making blood-based cfDNA testing a highly attractive alternative to traditional tissue biopsies. This growing patient and clinician demand for non-invasive diagnostics is a key market penetration driver, pushing the adoption of these advanced collection tubes across various medical specialties.
Competitive dynamics are intensifying, as evidenced by the strategic initiatives of key players. Companies are investing heavily in research and development to create tubes optimized for specific cfDNA applications, such as liquid biopsies for oncology, prenatal testing (NIPT), and transplant monitoring. Strategic partnerships between tube manufacturers and diagnostic assay developers are becoming more prevalent, creating integrated solutions that streamline the diagnostic workflow and accelerate time-to-result. The market penetration rate for cfDNA collection tubes in high-potential applications like oncology liquid biopsies is estimated to reach xx% by 2025, indicating a rapid adoption curve. Furthermore, the growing emphasis on personalized medicine and the burgeoning field of genomics are creating sustained demand for high-quality cfDNA specimens. The market is also experiencing a diversification of applications beyond traditional clinical settings, with significant growth anticipated in research laboratories focused on genomics, epigenomics, and translational research, further fueling market expansion.
Dominant Markets & Segments in Cell Free Dna Cfdna Blood Collection Tube
The Cell Free DNA (cfDNA) blood collection tube market is characterized by a clear dominance of specific applications and regional markets, driven by a combination of technological adoption, healthcare infrastructure, and unmet clinical needs.
Application: In Vitro Diagnostics (IVD)
The In Vitro Diagnostics (IVD) segment is the undisputed leader, holding a significant market share estimated at over xx billion USD in 2025. This dominance is propelled by the rapidly expanding utility of cfDNA in various diagnostic assays.
- Oncology Liquid Biopsies: This sub-segment is a major growth engine. cfDNA analysis enables non-invasive cancer detection, monitoring of treatment response, early detection of recurrence, and identification of actionable mutations for targeted therapy. The demand for accurate and sensitive cancer diagnostics is a primary driver.
- Non-Invasive Prenatal Testing (NIPT): NIPT for aneuploidies and other genetic abnormalities is a well-established and rapidly growing application. The safety and accuracy of cfDNA-based NIPT have led to its widespread adoption globally, driving substantial demand for collection tubes.
- Transplant Monitoring: cfDNA levels can indicate early signs of organ rejection, making cfDNA blood collection tubes crucial for post-transplant patient management and improved outcomes.
- Infectious Disease Detection: Emerging applications include the detection of cell-free microbial DNA for diagnosing and monitoring infectious diseases, presenting a growing opportunity.
Dominant Region: North America
North America currently leads the global market, with an estimated market size of xx billion USD in 2025. This leadership is attributed to several key factors:
- Advanced Healthcare Infrastructure: The region boasts a highly developed healthcare system with widespread access to advanced diagnostic technologies and cutting-edge medical research facilities.
- High R&D Investment: Significant investment in pharmaceutical and biotechnology research and development fuels the demand for advanced diagnostic tools and reagents, including cfDNA collection tubes.
- Early Adoption of Novel Technologies: North America has consistently been an early adopter of new diagnostic methodologies and personalized medicine approaches, including liquid biopsy.
- Favorable Regulatory Environment: While stringent, the regulatory pathways for diagnostic approvals in the US, for example, have historically supported innovation and market entry for novel IVD products.
- Presence of Key Market Players: Many leading global companies involved in cfDNA research and diagnostics have a strong presence and established distribution networks in North America.
Dominant Type: 5ml
Within product types, the 5ml collection tube segment is currently the most dominant, representing an estimated xx billion USD in market value in 2025. This is due to its versatility and suitability for a wide range of applications, particularly for routine diagnostic testing and initial research screens. However, the 10ml segment is experiencing rapid growth, driven by applications requiring higher cfDNA yields for more sensitive analyses or complex genomic studies.
The interplay of these dominant segments—IVD applications, North America as a leading region, and the 5ml tube type—creates a robust and expanding market for Cell Free DNA (cfDNA) blood collection tubes, with significant growth opportunities anticipated in emerging markets and advanced research areas.
Cell Free Dna Cfdna Blood Collection Tube Product Analysis
Cell free DNA (cfDNA) blood collection tubes represent a critical advancement in specimen stabilization for molecular diagnostics and research. Innovations focus on proprietary buffer formulations and specialized internal coatings that effectively inhibit nucleases, thereby preserving cfDNA integrity and maximizing yield from peripheral blood. These tubes are engineered to prevent the release of genomic DNA from lysed white blood cells, a common source of contamination that can skew results. Key competitive advantages include extended sample stability at room temperature, ease of use for phlebotomists and researchers, and compatibility with a broad range of downstream applications such as next-generation sequencing (NGS), PCR, and digital PCR. The continuous development of tubes optimized for specific cfDNA fractions, such as circulating tumor DNA (ctDNA), further enhances their market fit and clinical utility.
Key Drivers, Barriers & Challenges in Cell Free Dna Cfdna Blood Collection Tube
The Cell Free DNA (cfDNA) blood collection tube market is propelled by several key drivers, including the increasing demand for non-invasive diagnostic tests, particularly liquid biopsies for oncology and NIPT, and the growing global investment in genomic research. Technological advancements leading to improved cfDNA yield and stability are also significant. Policy-driven initiatives supporting personalized medicine and early disease detection further bolster market growth.
However, the market faces substantial barriers and challenges. High regulatory hurdles for new diagnostic assays utilizing these tubes can slow down market penetration. The cost of these specialized tubes, compared to standard blood collection devices, can be a restraint, especially in resource-limited settings. Supply chain complexities and the need for robust quality control to ensure consistent sample integrity are ongoing concerns. Intense competitive pressure from established and emerging players also necessitates continuous innovation and strategic pricing.
Growth Drivers in the Cell Free Dna Cfdna Blood Collection Tube Market
The growth of the Cell Free DNA (cfDNA) blood collection tube market is primarily fueled by the escalating global demand for non-invasive diagnostic solutions. Technological advancements are paramount, with continuous innovation in tube formulations that enhance cfDNA preservation and yield. Economic factors, such as increased healthcare spending and growing investments in personalized medicine and precision oncology, are significant growth catalysts. Policy initiatives aimed at promoting early disease detection and advanced diagnostics also play a crucial role. The expanding applications of cfDNA analysis in areas like infectious disease monitoring and transplant rejection detection further contribute to market expansion.
Challenges Impacting Cell Free Dna Cfdna Blood Collection Tube Growth
The growth of the Cell Free DNA (cfDNA) blood collection tube market is significantly impacted by regulatory complexities surrounding diagnostic assay approvals, which can lengthen time-to-market. Supply chain issues, including the sourcing of specialized raw materials and ensuring consistent product quality across global manufacturing sites, present ongoing challenges. Competitive pressures are intensifying, with numerous companies vying for market share, necessitating continuous product innovation and cost-effectiveness strategies. Furthermore, the perceived higher cost of these specialized tubes compared to conventional blood collection devices can be a barrier to widespread adoption in certain markets.
Key Players Shaping the Cell Free Dna Cfdna Blood Collection Tube Market
- Roche
- Nonacus
- Streck
- Cwbio IT Group
- Weihai Weigao
- Bunnyteeth
- DiaCarta
- BEAVER
- Ardent Biomed
- Hbxinle
- Thermo Fisher Scientific
Significant Cell Free Dna Cfdna Blood Collection Tube Industry Milestones
- 2019: Increased research publications highlighting the utility of cfDNA for early cancer detection.
- 2020: Expansion of NIPT usage in major global markets, driving demand for specialized collection tubes.
- 2021: Launch of novel cfDNA collection tube formulations with enhanced stabilization properties.
- 2022: Several acquisitions and partnerships in the liquid biopsy space, indirectly boosting demand for cfDNA tubes.
- 2023: Growing adoption of cfDNA monitoring for transplant patients in clinical settings.
- 2024: Emergence of new applications for cfDNA in infectious disease diagnostics.
Future Outlook for Cell Free Dna Cfdna Blood Collection Tube Market
The future outlook for the Cell Free DNA (cfDNA) blood collection tube market is exceptionally bright, driven by sustained innovation and expanding clinical utility. Strategic opportunities lie in the further development of tubes tailored for highly specific cfDNA applications, such as early-stage cancer detection and monitoring of rare mutations. The market potential is further amplified by the global push towards personalized medicine, where accurate and reliable cfDNA analysis is indispensable. Continued growth in emerging economies, coupled with increasing healthcare expenditure, will unlock new avenues for market expansion. The integration of cfDNA collection and analysis into routine clinical workflows is anticipated, solidifying the critical role of these tubes in the future of diagnostics.
Cell Free Dna Cfdna Blood Collection Tube Segmentation
-
1. Application
- 1.1. In Vitro Diagnostics
- 1.2. Scientific Research
-
2. Type
- 2.1. 5ml
- 2.2. 10ml
Cell Free Dna Cfdna Blood Collection Tube Segmentation By Geography
-
1. North America
- 1.1. United States
- 1.2. Canada
- 1.3. Mexico
-
2. South America
- 2.1. Brazil
- 2.2. Argentina
- 2.3. Rest of South America
-
3. Europe
- 3.1. United Kingdom
- 3.2. Germany
- 3.3. France
- 3.4. Italy
- 3.5. Spain
- 3.6. Russia
- 3.7. Benelux
- 3.8. Nordics
- 3.9. Rest of Europe
-
4. Middle East & Africa
- 4.1. Turkey
- 4.2. Israel
- 4.3. GCC
- 4.4. North Africa
- 4.5. South Africa
- 4.6. Rest of Middle East & Africa
-
5. Asia Pacific
- 5.1. China
- 5.2. India
- 5.3. Japan
- 5.4. South Korea
- 5.5. ASEAN
- 5.6. Oceania
- 5.7. Rest of Asia Pacific
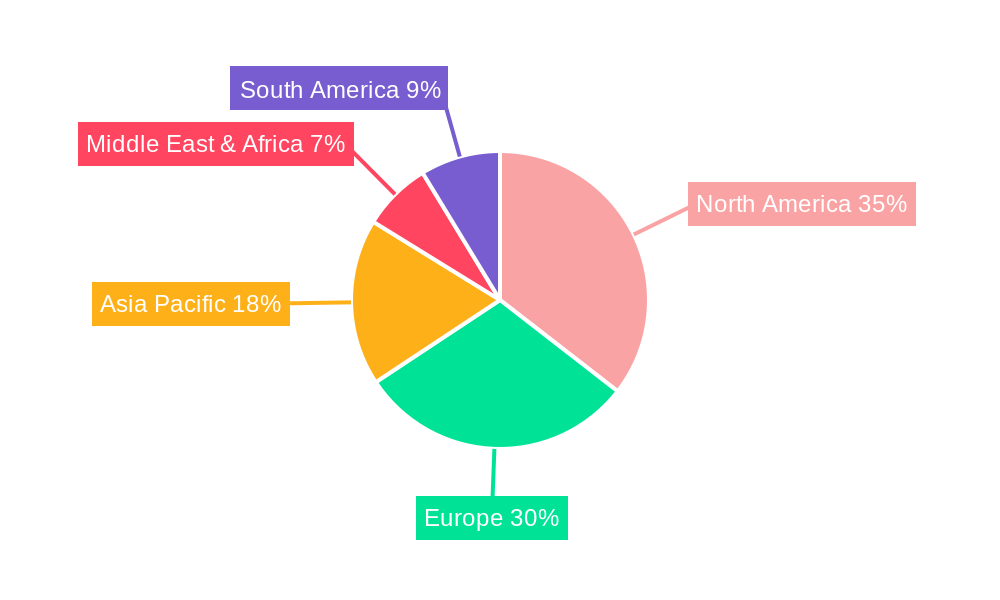
Cell Free Dna Cfdna Blood Collection Tube Regional Market Share

Geographic Coverage of Cell Free Dna Cfdna Blood Collection Tube
Cell Free Dna Cfdna Blood Collection Tube REPORT HIGHLIGHTS
| Aspects | Details |
|---|---|
| Study Period | 2020-2034 |
| Base Year | 2025 |
| Estimated Year | 2026 |
| Forecast Period | 2026-2034 |
| Historical Period | 2020-2025 |
| Growth Rate | CAGR of 14.85% from 2020-2034 |
| Segmentation |
|
Table of Contents
- 1. Introduction
- 1.1. Research Scope
- 1.2. Market Segmentation
- 1.3. Research Methodology
- 1.4. Definitions and Assumptions
- 2. Executive Summary
- 2.1. Introduction
- 3. Market Dynamics
- 3.1. Introduction
- 3.2. Market Drivers
- 3.3. Market Restrains
- 3.4. Market Trends
- 4. Market Factor Analysis
- 4.1. Porters Five Forces
- 4.2. Supply/Value Chain
- 4.3. PESTEL analysis
- 4.4. Market Entropy
- 4.5. Patent/Trademark Analysis
- 5. Global Cell Free Dna Cfdna Blood Collection Tube Analysis, Insights and Forecast, 2020-2032
- 5.1. Market Analysis, Insights and Forecast - by Application
- 5.1.1. In Vitro Diagnostics
- 5.1.2. Scientific Research
- 5.2. Market Analysis, Insights and Forecast - by Type
- 5.2.1. 5ml
- 5.2.2. 10ml
- 5.3. Market Analysis, Insights and Forecast - by Region
- 5.3.1. North America
- 5.3.2. South America
- 5.3.3. Europe
- 5.3.4. Middle East & Africa
- 5.3.5. Asia Pacific
- 5.1. Market Analysis, Insights and Forecast - by Application
- 6. North America Cell Free Dna Cfdna Blood Collection Tube Analysis, Insights and Forecast, 2020-2032
- 6.1. Market Analysis, Insights and Forecast - by Application
- 6.1.1. In Vitro Diagnostics
- 6.1.2. Scientific Research
- 6.2. Market Analysis, Insights and Forecast - by Type
- 6.2.1. 5ml
- 6.2.2. 10ml
- 6.1. Market Analysis, Insights and Forecast - by Application
- 7. South America Cell Free Dna Cfdna Blood Collection Tube Analysis, Insights and Forecast, 2020-2032
- 7.1. Market Analysis, Insights and Forecast - by Application
- 7.1.1. In Vitro Diagnostics
- 7.1.2. Scientific Research
- 7.2. Market Analysis, Insights and Forecast - by Type
- 7.2.1. 5ml
- 7.2.2. 10ml
- 7.1. Market Analysis, Insights and Forecast - by Application
- 8. Europe Cell Free Dna Cfdna Blood Collection Tube Analysis, Insights and Forecast, 2020-2032
- 8.1. Market Analysis, Insights and Forecast - by Application
- 8.1.1. In Vitro Diagnostics
- 8.1.2. Scientific Research
- 8.2. Market Analysis, Insights and Forecast - by Type
- 8.2.1. 5ml
- 8.2.2. 10ml
- 8.1. Market Analysis, Insights and Forecast - by Application
- 9. Middle East & Africa Cell Free Dna Cfdna Blood Collection Tube Analysis, Insights and Forecast, 2020-2032
- 9.1. Market Analysis, Insights and Forecast - by Application
- 9.1.1. In Vitro Diagnostics
- 9.1.2. Scientific Research
- 9.2. Market Analysis, Insights and Forecast - by Type
- 9.2.1. 5ml
- 9.2.2. 10ml
- 9.1. Market Analysis, Insights and Forecast - by Application
- 10. Asia Pacific Cell Free Dna Cfdna Blood Collection Tube Analysis, Insights and Forecast, 2020-2032
- 10.1. Market Analysis, Insights and Forecast - by Application
- 10.1.1. In Vitro Diagnostics
- 10.1.2. Scientific Research
- 10.2. Market Analysis, Insights and Forecast - by Type
- 10.2.1. 5ml
- 10.2.2. 10ml
- 10.1. Market Analysis, Insights and Forecast - by Application
- 11. Competitive Analysis
- 11.1. Global Market Share Analysis 2025
- 11.2. Company Profiles
- 11.2.1 Roche
- 11.2.1.1. Overview
- 11.2.1.2. Products
- 11.2.1.3. SWOT Analysis
- 11.2.1.4. Recent Developments
- 11.2.1.5. Financials (Based on Availability)
- 11.2.2 Nonacus
- 11.2.2.1. Overview
- 11.2.2.2. Products
- 11.2.2.3. SWOT Analysis
- 11.2.2.4. Recent Developments
- 11.2.2.5. Financials (Based on Availability)
- 11.2.3 Streck
- 11.2.3.1. Overview
- 11.2.3.2. Products
- 11.2.3.3. SWOT Analysis
- 11.2.3.4. Recent Developments
- 11.2.3.5. Financials (Based on Availability)
- 11.2.4 Cwbio IT Group
- 11.2.4.1. Overview
- 11.2.4.2. Products
- 11.2.4.3. SWOT Analysis
- 11.2.4.4. Recent Developments
- 11.2.4.5. Financials (Based on Availability)
- 11.2.5 Weihai Weigao
- 11.2.5.1. Overview
- 11.2.5.2. Products
- 11.2.5.3. SWOT Analysis
- 11.2.5.4. Recent Developments
- 11.2.5.5. Financials (Based on Availability)
- 11.2.6 Bunnyteeth
- 11.2.6.1. Overview
- 11.2.6.2. Products
- 11.2.6.3. SWOT Analysis
- 11.2.6.4. Recent Developments
- 11.2.6.5. Financials (Based on Availability)
- 11.2.7 DiaCarta
- 11.2.7.1. Overview
- 11.2.7.2. Products
- 11.2.7.3. SWOT Analysis
- 11.2.7.4. Recent Developments
- 11.2.7.5. Financials (Based on Availability)
- 11.2.8 BEAVER
- 11.2.8.1. Overview
- 11.2.8.2. Products
- 11.2.8.3. SWOT Analysis
- 11.2.8.4. Recent Developments
- 11.2.8.5. Financials (Based on Availability)
- 11.2.9 Ardent Biomed
- 11.2.9.1. Overview
- 11.2.9.2. Products
- 11.2.9.3. SWOT Analysis
- 11.2.9.4. Recent Developments
- 11.2.9.5. Financials (Based on Availability)
- 11.2.10 Hbxinle
- 11.2.10.1. Overview
- 11.2.10.2. Products
- 11.2.10.3. SWOT Analysis
- 11.2.10.4. Recent Developments
- 11.2.10.5. Financials (Based on Availability)
- 11.2.11 Thermo Fisher Scientific
- 11.2.11.1. Overview
- 11.2.11.2. Products
- 11.2.11.3. SWOT Analysis
- 11.2.11.4. Recent Developments
- 11.2.11.5. Financials (Based on Availability)
- 11.2.1 Roche
List of Figures
- Figure 1: Global Cell Free Dna Cfdna Blood Collection Tube Revenue Breakdown (undefined, %) by Region 2025 & 2033
- Figure 2: North America Cell Free Dna Cfdna Blood Collection Tube Revenue (undefined), by Application 2025 & 2033
- Figure 3: North America Cell Free Dna Cfdna Blood Collection Tube Revenue Share (%), by Application 2025 & 2033
- Figure 4: North America Cell Free Dna Cfdna Blood Collection Tube Revenue (undefined), by Type 2025 & 2033
- Figure 5: North America Cell Free Dna Cfdna Blood Collection Tube Revenue Share (%), by Type 2025 & 2033
- Figure 6: North America Cell Free Dna Cfdna Blood Collection Tube Revenue (undefined), by Country 2025 & 2033
- Figure 7: North America Cell Free Dna Cfdna Blood Collection Tube Revenue Share (%), by Country 2025 & 2033
- Figure 8: South America Cell Free Dna Cfdna Blood Collection Tube Revenue (undefined), by Application 2025 & 2033
- Figure 9: South America Cell Free Dna Cfdna Blood Collection Tube Revenue Share (%), by Application 2025 & 2033
- Figure 10: South America Cell Free Dna Cfdna Blood Collection Tube Revenue (undefined), by Type 2025 & 2033
- Figure 11: South America Cell Free Dna Cfdna Blood Collection Tube Revenue Share (%), by Type 2025 & 2033
- Figure 12: South America Cell Free Dna Cfdna Blood Collection Tube Revenue (undefined), by Country 2025 & 2033
- Figure 13: South America Cell Free Dna Cfdna Blood Collection Tube Revenue Share (%), by Country 2025 & 2033
- Figure 14: Europe Cell Free Dna Cfdna Blood Collection Tube Revenue (undefined), by Application 2025 & 2033
- Figure 15: Europe Cell Free Dna Cfdna Blood Collection Tube Revenue Share (%), by Application 2025 & 2033
- Figure 16: Europe Cell Free Dna Cfdna Blood Collection Tube Revenue (undefined), by Type 2025 & 2033
- Figure 17: Europe Cell Free Dna Cfdna Blood Collection Tube Revenue Share (%), by Type 2025 & 2033
- Figure 18: Europe Cell Free Dna Cfdna Blood Collection Tube Revenue (undefined), by Country 2025 & 2033
- Figure 19: Europe Cell Free Dna Cfdna Blood Collection Tube Revenue Share (%), by Country 2025 & 2033
- Figure 20: Middle East & Africa Cell Free Dna Cfdna Blood Collection Tube Revenue (undefined), by Application 2025 & 2033
- Figure 21: Middle East & Africa Cell Free Dna Cfdna Blood Collection Tube Revenue Share (%), by Application 2025 & 2033
- Figure 22: Middle East & Africa Cell Free Dna Cfdna Blood Collection Tube Revenue (undefined), by Type 2025 & 2033
- Figure 23: Middle East & Africa Cell Free Dna Cfdna Blood Collection Tube Revenue Share (%), by Type 2025 & 2033
- Figure 24: Middle East & Africa Cell Free Dna Cfdna Blood Collection Tube Revenue (undefined), by Country 2025 & 2033
- Figure 25: Middle East & Africa Cell Free Dna Cfdna Blood Collection Tube Revenue Share (%), by Country 2025 & 2033
- Figure 26: Asia Pacific Cell Free Dna Cfdna Blood Collection Tube Revenue (undefined), by Application 2025 & 2033
- Figure 27: Asia Pacific Cell Free Dna Cfdna Blood Collection Tube Revenue Share (%), by Application 2025 & 2033
- Figure 28: Asia Pacific Cell Free Dna Cfdna Blood Collection Tube Revenue (undefined), by Type 2025 & 2033
- Figure 29: Asia Pacific Cell Free Dna Cfdna Blood Collection Tube Revenue Share (%), by Type 2025 & 2033
- Figure 30: Asia Pacific Cell Free Dna Cfdna Blood Collection Tube Revenue (undefined), by Country 2025 & 2033
- Figure 31: Asia Pacific Cell Free Dna Cfdna Blood Collection Tube Revenue Share (%), by Country 2025 & 2033
List of Tables
- Table 1: Global Cell Free Dna Cfdna Blood Collection Tube Revenue undefined Forecast, by Application 2020 & 2033
- Table 2: Global Cell Free Dna Cfdna Blood Collection Tube Revenue undefined Forecast, by Type 2020 & 2033
- Table 3: Global Cell Free Dna Cfdna Blood Collection Tube Revenue undefined Forecast, by Region 2020 & 2033
- Table 4: Global Cell Free Dna Cfdna Blood Collection Tube Revenue undefined Forecast, by Application 2020 & 2033
- Table 5: Global Cell Free Dna Cfdna Blood Collection Tube Revenue undefined Forecast, by Type 2020 & 2033
- Table 6: Global Cell Free Dna Cfdna Blood Collection Tube Revenue undefined Forecast, by Country 2020 & 2033
- Table 7: United States Cell Free Dna Cfdna Blood Collection Tube Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 8: Canada Cell Free Dna Cfdna Blood Collection Tube Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 9: Mexico Cell Free Dna Cfdna Blood Collection Tube Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 10: Global Cell Free Dna Cfdna Blood Collection Tube Revenue undefined Forecast, by Application 2020 & 2033
- Table 11: Global Cell Free Dna Cfdna Blood Collection Tube Revenue undefined Forecast, by Type 2020 & 2033
- Table 12: Global Cell Free Dna Cfdna Blood Collection Tube Revenue undefined Forecast, by Country 2020 & 2033
- Table 13: Brazil Cell Free Dna Cfdna Blood Collection Tube Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 14: Argentina Cell Free Dna Cfdna Blood Collection Tube Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 15: Rest of South America Cell Free Dna Cfdna Blood Collection Tube Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 16: Global Cell Free Dna Cfdna Blood Collection Tube Revenue undefined Forecast, by Application 2020 & 2033
- Table 17: Global Cell Free Dna Cfdna Blood Collection Tube Revenue undefined Forecast, by Type 2020 & 2033
- Table 18: Global Cell Free Dna Cfdna Blood Collection Tube Revenue undefined Forecast, by Country 2020 & 2033
- Table 19: United Kingdom Cell Free Dna Cfdna Blood Collection Tube Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 20: Germany Cell Free Dna Cfdna Blood Collection Tube Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 21: France Cell Free Dna Cfdna Blood Collection Tube Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 22: Italy Cell Free Dna Cfdna Blood Collection Tube Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 23: Spain Cell Free Dna Cfdna Blood Collection Tube Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 24: Russia Cell Free Dna Cfdna Blood Collection Tube Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 25: Benelux Cell Free Dna Cfdna Blood Collection Tube Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 26: Nordics Cell Free Dna Cfdna Blood Collection Tube Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 27: Rest of Europe Cell Free Dna Cfdna Blood Collection Tube Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 28: Global Cell Free Dna Cfdna Blood Collection Tube Revenue undefined Forecast, by Application 2020 & 2033
- Table 29: Global Cell Free Dna Cfdna Blood Collection Tube Revenue undefined Forecast, by Type 2020 & 2033
- Table 30: Global Cell Free Dna Cfdna Blood Collection Tube Revenue undefined Forecast, by Country 2020 & 2033
- Table 31: Turkey Cell Free Dna Cfdna Blood Collection Tube Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 32: Israel Cell Free Dna Cfdna Blood Collection Tube Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 33: GCC Cell Free Dna Cfdna Blood Collection Tube Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 34: North Africa Cell Free Dna Cfdna Blood Collection Tube Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 35: South Africa Cell Free Dna Cfdna Blood Collection Tube Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 36: Rest of Middle East & Africa Cell Free Dna Cfdna Blood Collection Tube Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 37: Global Cell Free Dna Cfdna Blood Collection Tube Revenue undefined Forecast, by Application 2020 & 2033
- Table 38: Global Cell Free Dna Cfdna Blood Collection Tube Revenue undefined Forecast, by Type 2020 & 2033
- Table 39: Global Cell Free Dna Cfdna Blood Collection Tube Revenue undefined Forecast, by Country 2020 & 2033
- Table 40: China Cell Free Dna Cfdna Blood Collection Tube Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 41: India Cell Free Dna Cfdna Blood Collection Tube Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 42: Japan Cell Free Dna Cfdna Blood Collection Tube Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 43: South Korea Cell Free Dna Cfdna Blood Collection Tube Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 44: ASEAN Cell Free Dna Cfdna Blood Collection Tube Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 45: Oceania Cell Free Dna Cfdna Blood Collection Tube Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 46: Rest of Asia Pacific Cell Free Dna Cfdna Blood Collection Tube Revenue (undefined) Forecast, by Application 2020 & 2033
Frequently Asked Questions
1. What is the projected Compound Annual Growth Rate (CAGR) of the Cell Free Dna Cfdna Blood Collection Tube?
The projected CAGR is approximately 14.85%.
2. Which companies are prominent players in the Cell Free Dna Cfdna Blood Collection Tube?
Key companies in the market include Roche, Nonacus, Streck, Cwbio IT Group, Weihai Weigao, Bunnyteeth, DiaCarta, BEAVER, Ardent Biomed, Hbxinle, Thermo Fisher Scientific.
3. What are the main segments of the Cell Free Dna Cfdna Blood Collection Tube?
The market segments include Application, Type.
4. Can you provide details about the market size?
The market size is estimated to be USD XXX N/A as of 2022.
5. What are some drivers contributing to market growth?
N/A
6. What are the notable trends driving market growth?
N/A
7. Are there any restraints impacting market growth?
N/A
8. Can you provide examples of recent developments in the market?
N/A
9. What pricing options are available for accessing the report?
Pricing options include single-user, multi-user, and enterprise licenses priced at USD 4250.00, USD 6375.00, and USD 8500.00 respectively.
10. Is the market size provided in terms of value or volume?
The market size is provided in terms of value, measured in N/A.
11. Are there any specific market keywords associated with the report?
Yes, the market keyword associated with the report is "Cell Free Dna Cfdna Blood Collection Tube," which aids in identifying and referencing the specific market segment covered.
12. How do I determine which pricing option suits my needs best?
The pricing options vary based on user requirements and access needs. Individual users may opt for single-user licenses, while businesses requiring broader access may choose multi-user or enterprise licenses for cost-effective access to the report.
13. Are there any additional resources or data provided in the Cell Free Dna Cfdna Blood Collection Tube report?
While the report offers comprehensive insights, it's advisable to review the specific contents or supplementary materials provided to ascertain if additional resources or data are available.
14. How can I stay updated on further developments or reports in the Cell Free Dna Cfdna Blood Collection Tube?
To stay informed about further developments, trends, and reports in the Cell Free Dna Cfdna Blood Collection Tube, consider subscribing to industry newsletters, following relevant companies and organizations, or regularly checking reputable industry news sources and publications.
Methodology
Step 1 - Identification of Relevant Samples Size from Population Database



Step 2 - Approaches for Defining Global Market Size (Value, Volume* & Price*)

Note*: In applicable scenarios
Step 3 - Data Sources
Primary Research
- Web Analytics
- Survey Reports
- Research Institute
- Latest Research Reports
- Opinion Leaders
Secondary Research
- Annual Reports
- White Paper
- Latest Press Release
- Industry Association
- Paid Database
- Investor Presentations

Step 4 - Data Triangulation
Involves using different sources of information in order to increase the validity of a study
These sources are likely to be stakeholders in a program - participants, other researchers, program staff, other community members, and so on.
Then we put all data in single framework & apply various statistical tools to find out the dynamic on the market.
During the analysis stage, feedback from the stakeholder groups would be compared to determine areas of agreement as well as areas of divergence


