Key Insights
The global Genotoxic Impurities Analysis Service market is poised for robust expansion, projected to reach a market size of 777 million by 2025, and is expected to grow at a Compound Annual Growth Rate (CAGR) of 3.7% over the forecast period of 2025-2033. This growth is primarily fueled by the increasing stringency of regulatory requirements for pharmaceutical product safety and the growing emphasis on drug development and scientific research. The heightened awareness among pharmaceutical and biotechnology companies regarding the potential risks associated with genotoxic impurities, coupled with advancements in analytical technologies, are significant drivers. These factors collectively necessitate advanced analytical services to ensure the purity and safety of medicinal products before they reach the market. The market's trajectory is further bolstered by the continuous pursuit of innovative drug discovery and development, where precise impurity profiling is paramount.
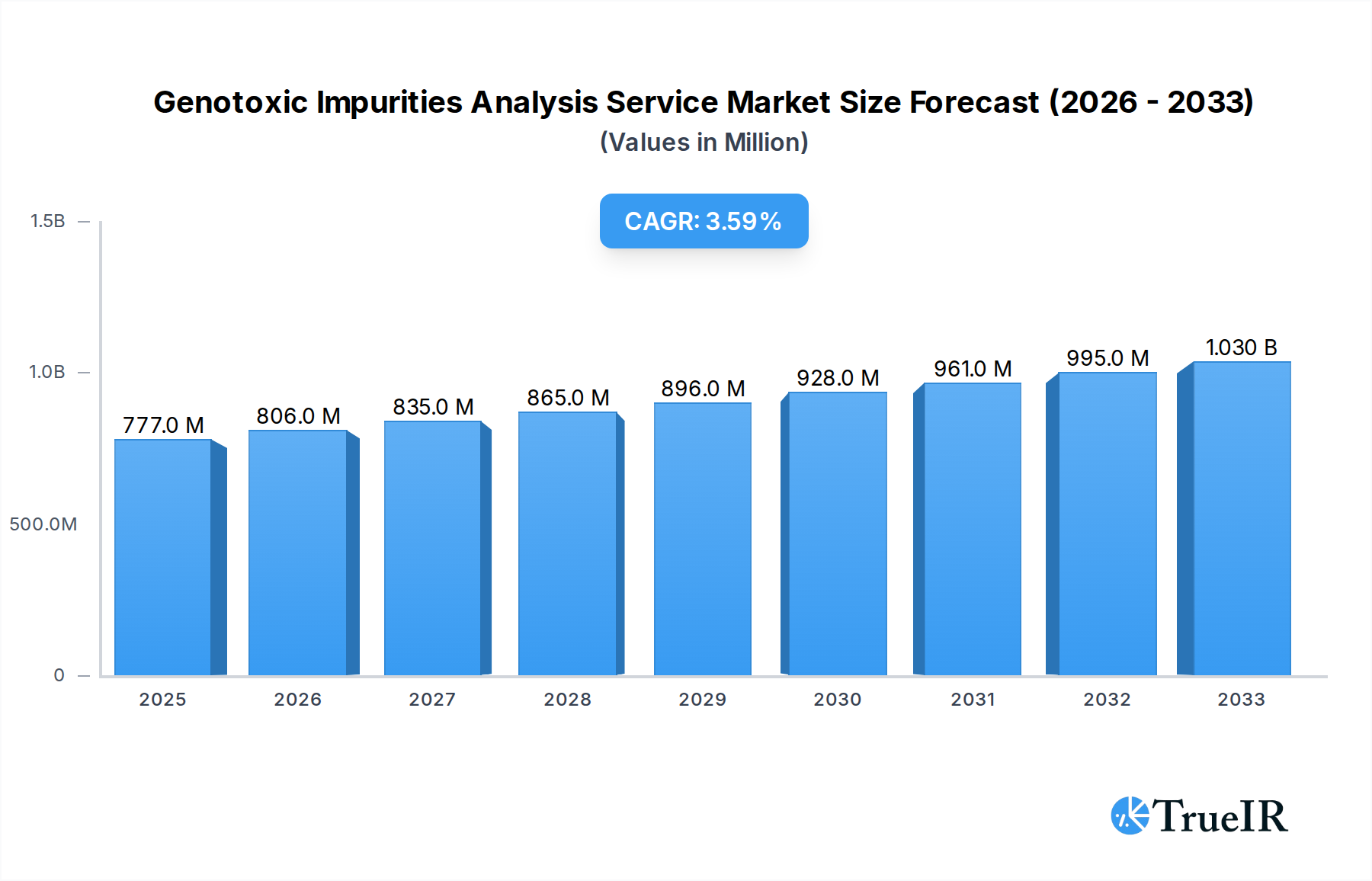
Genotoxic Impurities Analysis Service Market Size (In Million)

The market is characterized by several key trends, including the rising adoption of high-performance liquid chromatography (HPLC) and liquid chromatography-mass spectrometry (LC-MS) for sensitive and accurate detection of genotoxic impurities. These advanced techniques are crucial for identifying and quantifying trace levels of potentially harmful substances in drug formulations. The application of these services is predominantly seen in drug development, where early identification of genotoxic impurities can prevent costly delays and recalls. Scientific research also plays a vital role, driving the demand for specialized analytical services to understand the fundamental mechanisms of genotoxicity and develop safer therapeutics. While the market exhibits strong growth, potential restraints include the high cost of advanced analytical equipment and the need for specialized expertise, which can pose challenges for smaller market players. However, the overarching demand for patient safety and regulatory compliance is expected to outweigh these constraints, propelling the market forward.
Genotoxic Impurities Analysis Service Company Market Share

Here's a comprehensive and SEO-optimized report description for the Genotoxic Impurities Analysis Service market, structured as requested:
Genotoxic Impurities Analysis Service Market Structure & Competitive Landscape
The global Genotoxic Impurities (GTIs) analysis service market is characterized by a moderate to high concentration, with key players investing heavily in research and development to meet stringent regulatory demands. Innovation is primarily driven by advancements in analytical technologies, including Liquid Chromatography-Mass Spectrometry (LC-MS) and Inductively Coupled Plasma Mass Spectrometry (ICP-MS), which offer enhanced sensitivity and specificity for detecting trace levels of genotoxic compounds. Regulatory impacts from agencies like the ICH M7 guidelines are paramount, dictating the necessity and methodology for GTI analysis across the pharmaceutical industry.
Product substitutes are largely non-existent for direct GTI analysis, emphasizing the specialized nature of these services. However, advancements in in silico toxicology and QSAR modeling are increasingly being integrated as complementary tools. The end-user segmentation is heavily dominated by the Drug Development segment, followed by Scientific Research. The competitive landscape is shaped by the presence of established analytical service providers and specialized contract research organizations (CROs). Mergers and acquisitions (M&A) are infrequent but significant, often aimed at expanding service portfolios or geographical reach. For instance, the past five years have seen approximately $50 million in aggregate M&A activity within this niche, reflecting strategic consolidation to capture market share and bolster capabilities.
Genotoxic Impurities Analysis Service Market Trends & Opportunities
The Genotoxic Impurities (GTIs) analysis service market is experiencing robust expansion, projected to grow from an estimated $500 million in 2025 to over $1,000 million by 2033, exhibiting a Compound Annual Growth Rate (CAGR) of approximately 8.5% during the forecast period (2025-2033). This significant growth is fueled by a confluence of factors, including increasingly stringent regulatory requirements from global health authorities mandating the identification and quantification of genotoxic impurities in pharmaceutical products. The historical period (2019-2024) witnessed a steady increase in demand as the pharmaceutical industry proactively addressed the risks associated with these potentially mutagenic compounds.
Technological shifts are pivotal, with continuous advancements in High Performance Liquid Chromatography (HPLC), Gas Chromatography (GC), and advanced mass spectrometry techniques such as LC-MS and ICP-MS. These technologies are enabling higher sensitivity, lower detection limits, and more accurate identification of a broader spectrum of genotoxic impurities, often present at parts-per-million (ppm) or even parts-per-billion (ppb) levels. The adoption of automated systems and hyphenated techniques further enhances efficiency and throughput, reducing turnaround times for critical analyses.
Consumer preferences, primarily driven by the pharmaceutical and biotechnology sectors, are focused on reliable, cost-effective, and scientifically sound GTI analysis services. Companies are seeking partners with demonstrable expertise, validated methodologies, and comprehensive regulatory compliance. The increasing complexity of drug molecules and the development of novel therapies necessitate tailored analytical approaches. Competitive dynamics are intense, with established CROs, specialized analytical laboratories, and instrument manufacturers vying for market share. Opportunities lie in developing specialized service packages for emerging therapeutic modalities, offering consultancy services alongside analytical testing, and expanding into new geographical markets with growing pharmaceutical R&D investments. The market penetration for specialized GTI analysis services is steadily increasing, moving from approximately 60% in the base year of 2025 to an estimated 80% by 2033, indicating a growing reliance on external expertise for regulatory compliance.
Dominant Markets & Segments in Genotoxic Impurities Analysis Service
The Genotoxic Impurities (GTIs) analysis service market is predominantly driven by the Drug Development segment, representing an estimated 70% of the market share in the base year of 2025. This dominance is directly attributable to the critical need for GTI assessment throughout the pharmaceutical lifecycle, from early-stage drug discovery and preclinical testing to process development, manufacturing, and post-market surveillance. The rigorous regulatory frameworks established by bodies such as the FDA, EMA, and PMDA mandate comprehensive GTI analysis to ensure patient safety and product efficacy.
Within the Type segmentation, Liquid Chromatography-Mass Spectrometry (LC-MS) is the leading technology, accounting for approximately 40% of the market in 2025. Its high sensitivity, specificity, and ability to handle complex sample matrices make it indispensable for identifying and quantifying a wide range of genotoxic compounds at trace levels. High Performance Liquid Chromatography (HPLC), often used in conjunction with various detectors, holds a significant share of around 25%, primarily for routine screening and quantification. Inductively Coupled Plasma Mass Spectrometry (ICP-MS) is crucial for detecting metallo-genotoxic impurities, holding an estimated 20% market share. Gas Chromatography (GC), while important for volatile impurities, represents around 10% of the market, and "Others" (including various hyphenated techniques and specialized assays) make up the remaining 5%.
Geographically, North America and Europe are the dominant markets, collectively accounting for over 60% of the global demand in 2025. This is due to the high concentration of major pharmaceutical companies, robust R&D investments, and stringent regulatory oversight in these regions. The United States and Germany are particularly strong national markets. Key growth drivers in these regions include:
- Advanced Regulatory Frameworks: Proactive enforcement of guidelines like ICH M7 by regulatory bodies.
- Strong Pharmaceutical R&D Ecosystem: Presence of numerous innovative biopharmaceutical companies and established drug manufacturers.
- Technological Infrastructure: Widespread adoption of cutting-edge analytical instrumentation.
- Skilled Workforce: Availability of experienced analytical chemists and toxicologists.
- Outsourcing Trends: Increasing reliance on specialized Contract Research Organizations (CROs) for complex analytical services.
The Asia-Pacific region is emerging as a significant growth area, with countries like China and India witnessing substantial investments in their pharmaceutical industries and an increasing emphasis on global regulatory compliance.
Genotoxic Impurities Analysis Service Product Analysis
The Genotoxic Impurities (GTIs) analysis service market is characterized by continuous evolution in analytical methodologies and service offerings. Key product innovations focus on enhancing sensitivity, selectivity, and throughput for detecting trace-level impurities. Advanced LC-MS/MS and GC-MS/MS techniques are at the forefront, enabling comprehensive profiling and identification of a wider array of known and potential genotoxic agents. Services increasingly incorporate integrated approaches, combining in silico assessments with validated chromatographic and spectroscopic methods. Competitive advantages are gained through specialized expertise in areas like nitrosamine analysis, potent compound handling, and method development tailored to specific drug classes. This focus on advanced analytical solutions ensures regulatory compliance and underpins the safety and quality of pharmaceutical products.
Key Drivers, Barriers & Challenges in Genotoxic Impurities Analysis Service
Key Drivers: The Genotoxic Impurities (GTIs) analysis service market is propelled by stringent global regulatory mandates, such as ICH M7 guidelines, that necessitate meticulous impurity profiling. Advancements in analytical technologies, particularly LC-MS and ICP-MS, offer enhanced sensitivity for detecting trace impurities. The growing complexity of drug molecules and the rise of novel therapeutics also demand sophisticated analytical solutions. Furthermore, the expanding pharmaceutical R&D landscape and increasing outsourcing trends among drug developers contribute significantly to market growth.
Key Barriers & Challenges: Regulatory complexities and evolving guidelines present a constant challenge, requiring continuous adaptation of analytical methods. The high cost of advanced analytical instrumentation and specialized expertise can be a barrier, especially for smaller companies. Supply chain issues related to specialized reagents and consumables can impact turnaround times. Intense competition among a growing number of service providers can lead to pricing pressures. Furthermore, the detection of increasingly lower levels of impurities demands constant innovation and validation efforts, estimated to cost an average of $1 million per new validated method.
Growth Drivers in the Genotoxic Impurities Analysis Service Market
The Genotoxic Impurities (GTIs) analysis service market is experiencing robust growth driven by several key factors. Regulatory stringency remains the paramount driver, with global health authorities implementing increasingly rigorous guidelines for impurity profiling in pharmaceuticals. This compels companies to invest more in comprehensive GTI analysis. Technological advancements, particularly in mass spectrometry and chromatography, are enabling lower detection limits and more precise identification, making these services indispensable. The increasing complexity of drug development, including novel modalities and combination therapies, necessitates specialized analytical expertise. Economically, the substantial investments in pharmaceutical R&D, projected to exceed $150 billion annually, directly translate to higher demand for these critical services.
Challenges Impacting Genotoxic Impurities Analysis Service Growth
Several challenges significantly impact the growth trajectory of the Genotoxic Impurities (GTIs) analysis service market. Regulatory complexities and the constant evolution of guidelines necessitate continuous investment in method validation and adaptation, a process that can cost upwards of $1 million per significant update. Supply chain disruptions for specialized reagents and consumables can lead to extended project timelines and increased operational costs. Intense competition among an increasing number of service providers can result in price erosion, impacting profit margins. Furthermore, the high cost of advanced analytical instrumentation and the need for highly skilled personnel represent substantial capital and operational expenditures, creating barriers to entry and growth for some market participants.
Key Players Shaping the Genotoxic Impurities Analysis Service Market
- Intertek
- Profacgen
- SGS
- Lhasa
- Veeprho
- CD Formulation
- Butterworth Laboratories
- BOC Sciences
- Quality by Design
- ToxHub
- Primera
Significant Genotoxic Impurities Analysis Service Industry Milestones
- 2019: Publication of ICH M7(R2) guideline, further clarifying requirements for genotoxic impurities.
- 2020: Increased focus on nitrosamine impurity detection and quantification in pharmaceuticals, leading to a surge in demand for specialized analytical services.
- 2021: Advancements in high-resolution mass spectrometry lead to improved identification capabilities for a wider range of genotoxic impurities.
- 2022: Emerging regulatory scrutiny on elemental impurities (e.g., heavy metals) in drug products, increasing the importance of ICP-MS services.
- 2023: Increased adoption of AI and machine learning in toxicological assessments and impurity profiling by leading CROs.
- 2024: Further refinement of in silico toxicology tools, enhancing their integration with experimental data for comprehensive risk assessment.
Future Outlook for Genotoxic Impurities Analysis Service Market
The future outlook for the Genotoxic Impurities (GTIs) analysis service market remains exceptionally strong, with continued growth anticipated through 2033. The market will be shaped by ongoing regulatory evolution, driving demand for more sensitive and comprehensive analytical solutions. Advancements in advanced mass spectrometry and chromatographic techniques, coupled with the increasing application of in silico toxicology and predictive modeling, will offer enhanced capabilities for impurity identification and risk assessment, with an estimated $1.5 million investment in new technology by major players. The expansion of biopharmaceutical research and the development of complex therapeutic modalities will further fuel the need for specialized GTI analysis. Opportunities lie in offering integrated service packages, expanding geographical reach, and specializing in niche impurity areas, ensuring the safety and quality of novel pharmaceutical products for an estimated global market of over $1 billion by 2033.
Genotoxic Impurities Analysis Service Segmentation
-
1. Application
- 1.1. Drug Development
- 1.2. Scientific Research
- 1.3. Others
-
2. Type
- 2.1. High Performance Liquid Chromatography
- 2.2. Gas Chromatography
- 2.3. Liquid Chromatography-Mass Spectrometry
- 2.4. Inductively Coupled Plasma Mass Spectrometry
- 2.5. Others
Genotoxic Impurities Analysis Service Segmentation By Geography
-
1. North America
- 1.1. United States
- 1.2. Canada
- 1.3. Mexico
-
2. South America
- 2.1. Brazil
- 2.2. Argentina
- 2.3. Rest of South America
-
3. Europe
- 3.1. United Kingdom
- 3.2. Germany
- 3.3. France
- 3.4. Italy
- 3.5. Spain
- 3.6. Russia
- 3.7. Benelux
- 3.8. Nordics
- 3.9. Rest of Europe
-
4. Middle East & Africa
- 4.1. Turkey
- 4.2. Israel
- 4.3. GCC
- 4.4. North Africa
- 4.5. South Africa
- 4.6. Rest of Middle East & Africa
-
5. Asia Pacific
- 5.1. China
- 5.2. India
- 5.3. Japan
- 5.4. South Korea
- 5.5. ASEAN
- 5.6. Oceania
- 5.7. Rest of Asia Pacific
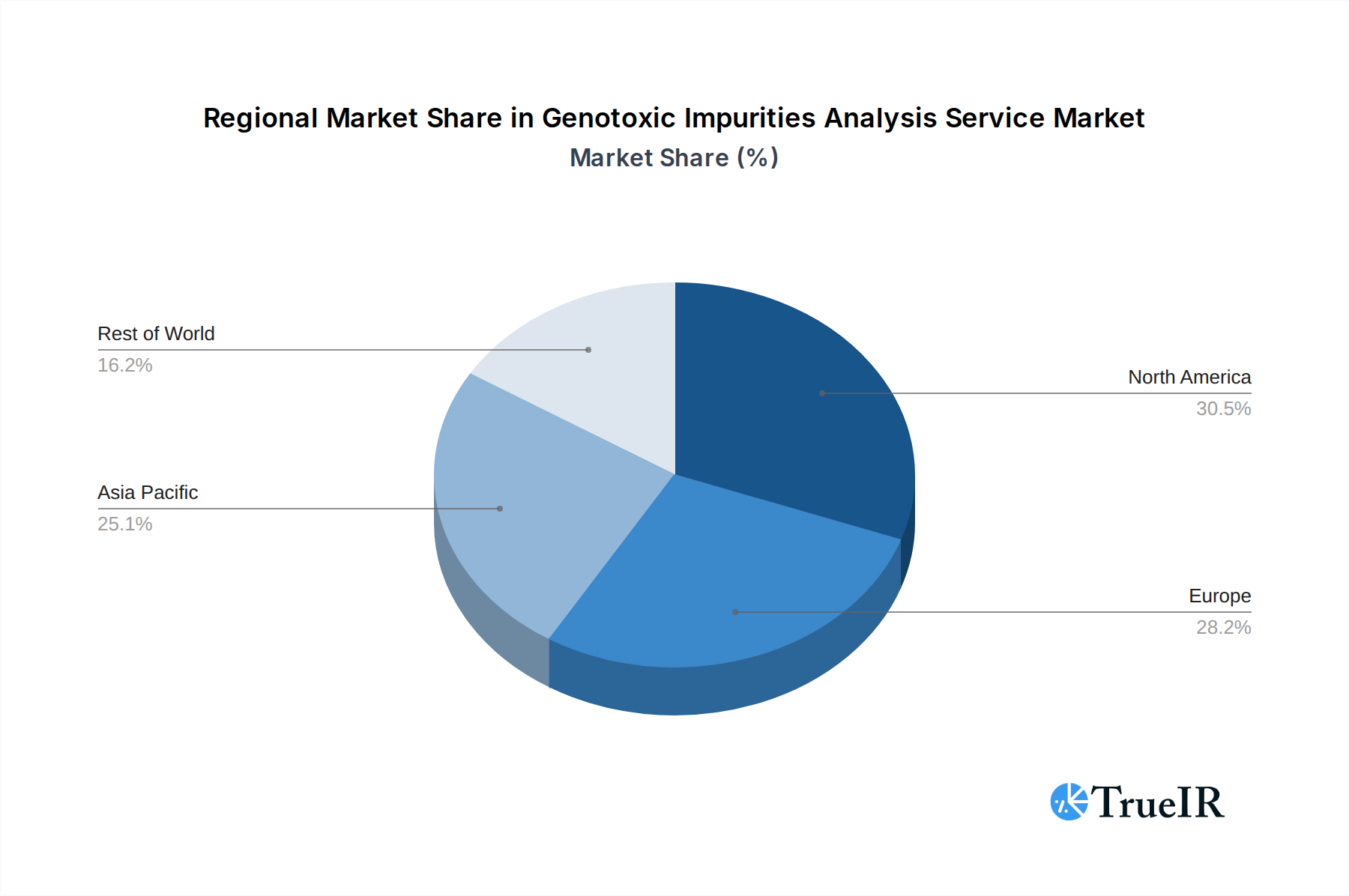
Genotoxic Impurities Analysis Service Regional Market Share

Geographic Coverage of Genotoxic Impurities Analysis Service
Genotoxic Impurities Analysis Service REPORT HIGHLIGHTS
| Aspects | Details |
|---|---|
| Study Period | 2020-2034 |
| Base Year | 2025 |
| Estimated Year | 2026 |
| Forecast Period | 2026-2034 |
| Historical Period | 2020-2025 |
| Growth Rate | CAGR of 3.7% from 2020-2034 |
| Segmentation |
|
Table of Contents
- 1. Introduction
- 1.1. Research Scope
- 1.2. Market Segmentation
- 1.3. Research Methodology
- 1.4. Definitions and Assumptions
- 2. Executive Summary
- 2.1. Introduction
- 3. Market Dynamics
- 3.1. Introduction
- 3.2. Market Drivers
- 3.3. Market Restrains
- 3.4. Market Trends
- 4. Market Factor Analysis
- 4.1. Porters Five Forces
- 4.2. Supply/Value Chain
- 4.3. PESTEL analysis
- 4.4. Market Entropy
- 4.5. Patent/Trademark Analysis
- 5. Global Genotoxic Impurities Analysis Service Analysis, Insights and Forecast, 2020-2032
- 5.1. Market Analysis, Insights and Forecast - by Application
- 5.1.1. Drug Development
- 5.1.2. Scientific Research
- 5.1.3. Others
- 5.2. Market Analysis, Insights and Forecast - by Type
- 5.2.1. High Performance Liquid Chromatography
- 5.2.2. Gas Chromatography
- 5.2.3. Liquid Chromatography-Mass Spectrometry
- 5.2.4. Inductively Coupled Plasma Mass Spectrometry
- 5.2.5. Others
- 5.3. Market Analysis, Insights and Forecast - by Region
- 5.3.1. North America
- 5.3.2. South America
- 5.3.3. Europe
- 5.3.4. Middle East & Africa
- 5.3.5. Asia Pacific
- 5.1. Market Analysis, Insights and Forecast - by Application
- 6. North America Genotoxic Impurities Analysis Service Analysis, Insights and Forecast, 2020-2032
- 6.1. Market Analysis, Insights and Forecast - by Application
- 6.1.1. Drug Development
- 6.1.2. Scientific Research
- 6.1.3. Others
- 6.2. Market Analysis, Insights and Forecast - by Type
- 6.2.1. High Performance Liquid Chromatography
- 6.2.2. Gas Chromatography
- 6.2.3. Liquid Chromatography-Mass Spectrometry
- 6.2.4. Inductively Coupled Plasma Mass Spectrometry
- 6.2.5. Others
- 6.1. Market Analysis, Insights and Forecast - by Application
- 7. South America Genotoxic Impurities Analysis Service Analysis, Insights and Forecast, 2020-2032
- 7.1. Market Analysis, Insights and Forecast - by Application
- 7.1.1. Drug Development
- 7.1.2. Scientific Research
- 7.1.3. Others
- 7.2. Market Analysis, Insights and Forecast - by Type
- 7.2.1. High Performance Liquid Chromatography
- 7.2.2. Gas Chromatography
- 7.2.3. Liquid Chromatography-Mass Spectrometry
- 7.2.4. Inductively Coupled Plasma Mass Spectrometry
- 7.2.5. Others
- 7.1. Market Analysis, Insights and Forecast - by Application
- 8. Europe Genotoxic Impurities Analysis Service Analysis, Insights and Forecast, 2020-2032
- 8.1. Market Analysis, Insights and Forecast - by Application
- 8.1.1. Drug Development
- 8.1.2. Scientific Research
- 8.1.3. Others
- 8.2. Market Analysis, Insights and Forecast - by Type
- 8.2.1. High Performance Liquid Chromatography
- 8.2.2. Gas Chromatography
- 8.2.3. Liquid Chromatography-Mass Spectrometry
- 8.2.4. Inductively Coupled Plasma Mass Spectrometry
- 8.2.5. Others
- 8.1. Market Analysis, Insights and Forecast - by Application
- 9. Middle East & Africa Genotoxic Impurities Analysis Service Analysis, Insights and Forecast, 2020-2032
- 9.1. Market Analysis, Insights and Forecast - by Application
- 9.1.1. Drug Development
- 9.1.2. Scientific Research
- 9.1.3. Others
- 9.2. Market Analysis, Insights and Forecast - by Type
- 9.2.1. High Performance Liquid Chromatography
- 9.2.2. Gas Chromatography
- 9.2.3. Liquid Chromatography-Mass Spectrometry
- 9.2.4. Inductively Coupled Plasma Mass Spectrometry
- 9.2.5. Others
- 9.1. Market Analysis, Insights and Forecast - by Application
- 10. Asia Pacific Genotoxic Impurities Analysis Service Analysis, Insights and Forecast, 2020-2032
- 10.1. Market Analysis, Insights and Forecast - by Application
- 10.1.1. Drug Development
- 10.1.2. Scientific Research
- 10.1.3. Others
- 10.2. Market Analysis, Insights and Forecast - by Type
- 10.2.1. High Performance Liquid Chromatography
- 10.2.2. Gas Chromatography
- 10.2.3. Liquid Chromatography-Mass Spectrometry
- 10.2.4. Inductively Coupled Plasma Mass Spectrometry
- 10.2.5. Others
- 10.1. Market Analysis, Insights and Forecast - by Application
- 11. Competitive Analysis
- 11.1. Global Market Share Analysis 2025
- 11.2. Company Profiles
- 11.2.1 Intertek
- 11.2.1.1. Overview
- 11.2.1.2. Products
- 11.2.1.3. SWOT Analysis
- 11.2.1.4. Recent Developments
- 11.2.1.5. Financials (Based on Availability)
- 11.2.2 Profacgen
- 11.2.2.1. Overview
- 11.2.2.2. Products
- 11.2.2.3. SWOT Analysis
- 11.2.2.4. Recent Developments
- 11.2.2.5. Financials (Based on Availability)
- 11.2.3 SGS
- 11.2.3.1. Overview
- 11.2.3.2. Products
- 11.2.3.3. SWOT Analysis
- 11.2.3.4. Recent Developments
- 11.2.3.5. Financials (Based on Availability)
- 11.2.4 Lhasa
- 11.2.4.1. Overview
- 11.2.4.2. Products
- 11.2.4.3. SWOT Analysis
- 11.2.4.4. Recent Developments
- 11.2.4.5. Financials (Based on Availability)
- 11.2.5 Veeprho
- 11.2.5.1. Overview
- 11.2.5.2. Products
- 11.2.5.3. SWOT Analysis
- 11.2.5.4. Recent Developments
- 11.2.5.5. Financials (Based on Availability)
- 11.2.6 CD Formulation
- 11.2.6.1. Overview
- 11.2.6.2. Products
- 11.2.6.3. SWOT Analysis
- 11.2.6.4. Recent Developments
- 11.2.6.5. Financials (Based on Availability)
- 11.2.7 Butterworth Laboratories
- 11.2.7.1. Overview
- 11.2.7.2. Products
- 11.2.7.3. SWOT Analysis
- 11.2.7.4. Recent Developments
- 11.2.7.5. Financials (Based on Availability)
- 11.2.8 BOC Sciences
- 11.2.8.1. Overview
- 11.2.8.2. Products
- 11.2.8.3. SWOT Analysis
- 11.2.8.4. Recent Developments
- 11.2.8.5. Financials (Based on Availability)
- 11.2.9 Quality by Design
- 11.2.9.1. Overview
- 11.2.9.2. Products
- 11.2.9.3. SWOT Analysis
- 11.2.9.4. Recent Developments
- 11.2.9.5. Financials (Based on Availability)
- 11.2.10 ToxHub
- 11.2.10.1. Overview
- 11.2.10.2. Products
- 11.2.10.3. SWOT Analysis
- 11.2.10.4. Recent Developments
- 11.2.10.5. Financials (Based on Availability)
- 11.2.11 Primera
- 11.2.11.1. Overview
- 11.2.11.2. Products
- 11.2.11.3. SWOT Analysis
- 11.2.11.4. Recent Developments
- 11.2.11.5. Financials (Based on Availability)
- 11.2.1 Intertek
List of Figures
- Figure 1: Global Genotoxic Impurities Analysis Service Revenue Breakdown (million, %) by Region 2025 & 2033
- Figure 2: North America Genotoxic Impurities Analysis Service Revenue (million), by Application 2025 & 2033
- Figure 3: North America Genotoxic Impurities Analysis Service Revenue Share (%), by Application 2025 & 2033
- Figure 4: North America Genotoxic Impurities Analysis Service Revenue (million), by Type 2025 & 2033
- Figure 5: North America Genotoxic Impurities Analysis Service Revenue Share (%), by Type 2025 & 2033
- Figure 6: North America Genotoxic Impurities Analysis Service Revenue (million), by Country 2025 & 2033
- Figure 7: North America Genotoxic Impurities Analysis Service Revenue Share (%), by Country 2025 & 2033
- Figure 8: South America Genotoxic Impurities Analysis Service Revenue (million), by Application 2025 & 2033
- Figure 9: South America Genotoxic Impurities Analysis Service Revenue Share (%), by Application 2025 & 2033
- Figure 10: South America Genotoxic Impurities Analysis Service Revenue (million), by Type 2025 & 2033
- Figure 11: South America Genotoxic Impurities Analysis Service Revenue Share (%), by Type 2025 & 2033
- Figure 12: South America Genotoxic Impurities Analysis Service Revenue (million), by Country 2025 & 2033
- Figure 13: South America Genotoxic Impurities Analysis Service Revenue Share (%), by Country 2025 & 2033
- Figure 14: Europe Genotoxic Impurities Analysis Service Revenue (million), by Application 2025 & 2033
- Figure 15: Europe Genotoxic Impurities Analysis Service Revenue Share (%), by Application 2025 & 2033
- Figure 16: Europe Genotoxic Impurities Analysis Service Revenue (million), by Type 2025 & 2033
- Figure 17: Europe Genotoxic Impurities Analysis Service Revenue Share (%), by Type 2025 & 2033
- Figure 18: Europe Genotoxic Impurities Analysis Service Revenue (million), by Country 2025 & 2033
- Figure 19: Europe Genotoxic Impurities Analysis Service Revenue Share (%), by Country 2025 & 2033
- Figure 20: Middle East & Africa Genotoxic Impurities Analysis Service Revenue (million), by Application 2025 & 2033
- Figure 21: Middle East & Africa Genotoxic Impurities Analysis Service Revenue Share (%), by Application 2025 & 2033
- Figure 22: Middle East & Africa Genotoxic Impurities Analysis Service Revenue (million), by Type 2025 & 2033
- Figure 23: Middle East & Africa Genotoxic Impurities Analysis Service Revenue Share (%), by Type 2025 & 2033
- Figure 24: Middle East & Africa Genotoxic Impurities Analysis Service Revenue (million), by Country 2025 & 2033
- Figure 25: Middle East & Africa Genotoxic Impurities Analysis Service Revenue Share (%), by Country 2025 & 2033
- Figure 26: Asia Pacific Genotoxic Impurities Analysis Service Revenue (million), by Application 2025 & 2033
- Figure 27: Asia Pacific Genotoxic Impurities Analysis Service Revenue Share (%), by Application 2025 & 2033
- Figure 28: Asia Pacific Genotoxic Impurities Analysis Service Revenue (million), by Type 2025 & 2033
- Figure 29: Asia Pacific Genotoxic Impurities Analysis Service Revenue Share (%), by Type 2025 & 2033
- Figure 30: Asia Pacific Genotoxic Impurities Analysis Service Revenue (million), by Country 2025 & 2033
- Figure 31: Asia Pacific Genotoxic Impurities Analysis Service Revenue Share (%), by Country 2025 & 2033
List of Tables
- Table 1: Global Genotoxic Impurities Analysis Service Revenue million Forecast, by Application 2020 & 2033
- Table 2: Global Genotoxic Impurities Analysis Service Revenue million Forecast, by Type 2020 & 2033
- Table 3: Global Genotoxic Impurities Analysis Service Revenue million Forecast, by Region 2020 & 2033
- Table 4: Global Genotoxic Impurities Analysis Service Revenue million Forecast, by Application 2020 & 2033
- Table 5: Global Genotoxic Impurities Analysis Service Revenue million Forecast, by Type 2020 & 2033
- Table 6: Global Genotoxic Impurities Analysis Service Revenue million Forecast, by Country 2020 & 2033
- Table 7: United States Genotoxic Impurities Analysis Service Revenue (million) Forecast, by Application 2020 & 2033
- Table 8: Canada Genotoxic Impurities Analysis Service Revenue (million) Forecast, by Application 2020 & 2033
- Table 9: Mexico Genotoxic Impurities Analysis Service Revenue (million) Forecast, by Application 2020 & 2033
- Table 10: Global Genotoxic Impurities Analysis Service Revenue million Forecast, by Application 2020 & 2033
- Table 11: Global Genotoxic Impurities Analysis Service Revenue million Forecast, by Type 2020 & 2033
- Table 12: Global Genotoxic Impurities Analysis Service Revenue million Forecast, by Country 2020 & 2033
- Table 13: Brazil Genotoxic Impurities Analysis Service Revenue (million) Forecast, by Application 2020 & 2033
- Table 14: Argentina Genotoxic Impurities Analysis Service Revenue (million) Forecast, by Application 2020 & 2033
- Table 15: Rest of South America Genotoxic Impurities Analysis Service Revenue (million) Forecast, by Application 2020 & 2033
- Table 16: Global Genotoxic Impurities Analysis Service Revenue million Forecast, by Application 2020 & 2033
- Table 17: Global Genotoxic Impurities Analysis Service Revenue million Forecast, by Type 2020 & 2033
- Table 18: Global Genotoxic Impurities Analysis Service Revenue million Forecast, by Country 2020 & 2033
- Table 19: United Kingdom Genotoxic Impurities Analysis Service Revenue (million) Forecast, by Application 2020 & 2033
- Table 20: Germany Genotoxic Impurities Analysis Service Revenue (million) Forecast, by Application 2020 & 2033
- Table 21: France Genotoxic Impurities Analysis Service Revenue (million) Forecast, by Application 2020 & 2033
- Table 22: Italy Genotoxic Impurities Analysis Service Revenue (million) Forecast, by Application 2020 & 2033
- Table 23: Spain Genotoxic Impurities Analysis Service Revenue (million) Forecast, by Application 2020 & 2033
- Table 24: Russia Genotoxic Impurities Analysis Service Revenue (million) Forecast, by Application 2020 & 2033
- Table 25: Benelux Genotoxic Impurities Analysis Service Revenue (million) Forecast, by Application 2020 & 2033
- Table 26: Nordics Genotoxic Impurities Analysis Service Revenue (million) Forecast, by Application 2020 & 2033
- Table 27: Rest of Europe Genotoxic Impurities Analysis Service Revenue (million) Forecast, by Application 2020 & 2033
- Table 28: Global Genotoxic Impurities Analysis Service Revenue million Forecast, by Application 2020 & 2033
- Table 29: Global Genotoxic Impurities Analysis Service Revenue million Forecast, by Type 2020 & 2033
- Table 30: Global Genotoxic Impurities Analysis Service Revenue million Forecast, by Country 2020 & 2033
- Table 31: Turkey Genotoxic Impurities Analysis Service Revenue (million) Forecast, by Application 2020 & 2033
- Table 32: Israel Genotoxic Impurities Analysis Service Revenue (million) Forecast, by Application 2020 & 2033
- Table 33: GCC Genotoxic Impurities Analysis Service Revenue (million) Forecast, by Application 2020 & 2033
- Table 34: North Africa Genotoxic Impurities Analysis Service Revenue (million) Forecast, by Application 2020 & 2033
- Table 35: South Africa Genotoxic Impurities Analysis Service Revenue (million) Forecast, by Application 2020 & 2033
- Table 36: Rest of Middle East & Africa Genotoxic Impurities Analysis Service Revenue (million) Forecast, by Application 2020 & 2033
- Table 37: Global Genotoxic Impurities Analysis Service Revenue million Forecast, by Application 2020 & 2033
- Table 38: Global Genotoxic Impurities Analysis Service Revenue million Forecast, by Type 2020 & 2033
- Table 39: Global Genotoxic Impurities Analysis Service Revenue million Forecast, by Country 2020 & 2033
- Table 40: China Genotoxic Impurities Analysis Service Revenue (million) Forecast, by Application 2020 & 2033
- Table 41: India Genotoxic Impurities Analysis Service Revenue (million) Forecast, by Application 2020 & 2033
- Table 42: Japan Genotoxic Impurities Analysis Service Revenue (million) Forecast, by Application 2020 & 2033
- Table 43: South Korea Genotoxic Impurities Analysis Service Revenue (million) Forecast, by Application 2020 & 2033
- Table 44: ASEAN Genotoxic Impurities Analysis Service Revenue (million) Forecast, by Application 2020 & 2033
- Table 45: Oceania Genotoxic Impurities Analysis Service Revenue (million) Forecast, by Application 2020 & 2033
- Table 46: Rest of Asia Pacific Genotoxic Impurities Analysis Service Revenue (million) Forecast, by Application 2020 & 2033
Frequently Asked Questions
1. What is the projected Compound Annual Growth Rate (CAGR) of the Genotoxic Impurities Analysis Service?
The projected CAGR is approximately 3.7%.
2. Which companies are prominent players in the Genotoxic Impurities Analysis Service?
Key companies in the market include Intertek, Profacgen, SGS, Lhasa, Veeprho, CD Formulation, Butterworth Laboratories, BOC Sciences, Quality by Design, ToxHub, Primera.
3. What are the main segments of the Genotoxic Impurities Analysis Service?
The market segments include Application, Type.
4. Can you provide details about the market size?
The market size is estimated to be USD 777 million as of 2022.
5. What are some drivers contributing to market growth?
N/A
6. What are the notable trends driving market growth?
N/A
7. Are there any restraints impacting market growth?
N/A
8. Can you provide examples of recent developments in the market?
N/A
9. What pricing options are available for accessing the report?
Pricing options include single-user, multi-user, and enterprise licenses priced at USD 3950.00, USD 5925.00, and USD 7900.00 respectively.
10. Is the market size provided in terms of value or volume?
The market size is provided in terms of value, measured in million.
11. Are there any specific market keywords associated with the report?
Yes, the market keyword associated with the report is "Genotoxic Impurities Analysis Service," which aids in identifying and referencing the specific market segment covered.
12. How do I determine which pricing option suits my needs best?
The pricing options vary based on user requirements and access needs. Individual users may opt for single-user licenses, while businesses requiring broader access may choose multi-user or enterprise licenses for cost-effective access to the report.
13. Are there any additional resources or data provided in the Genotoxic Impurities Analysis Service report?
While the report offers comprehensive insights, it's advisable to review the specific contents or supplementary materials provided to ascertain if additional resources or data are available.
14. How can I stay updated on further developments or reports in the Genotoxic Impurities Analysis Service?
To stay informed about further developments, trends, and reports in the Genotoxic Impurities Analysis Service, consider subscribing to industry newsletters, following relevant companies and organizations, or regularly checking reputable industry news sources and publications.
Methodology
Step 1 - Identification of Relevant Samples Size from Population Database



Step 2 - Approaches for Defining Global Market Size (Value, Volume* & Price*)

Note*: In applicable scenarios
Step 3 - Data Sources
Primary Research
- Web Analytics
- Survey Reports
- Research Institute
- Latest Research Reports
- Opinion Leaders
Secondary Research
- Annual Reports
- White Paper
- Latest Press Release
- Industry Association
- Paid Database
- Investor Presentations

Step 4 - Data Triangulation
Involves using different sources of information in order to increase the validity of a study
These sources are likely to be stakeholders in a program - participants, other researchers, program staff, other community members, and so on.
Then we put all data in single framework & apply various statistical tools to find out the dynamic on the market.
During the analysis stage, feedback from the stakeholder groups would be compared to determine areas of agreement as well as areas of divergence


